Advanced Manufacturing of Bactericidal Imidazole Intermediates for Global Pharma Supply Chains
Advanced Manufacturing of Bactericidal Imidazole Intermediates for Global Pharma Supply Chains
The pharmaceutical industry is constantly seeking robust and efficient pathways to synthesize nitrogen-containing heterocyclic compounds, particularly imidazole derivatives, which serve as critical scaffolds in numerous therapeutic agents ranging from antihypertensives like Losartan to novel kinase inhibitors. A significant breakthrough in this domain is detailed in patent CN110256357B, which discloses a highly effective preparation method for a specific imidazole drug molecule exhibiting potent bactericidal activity against pathogens such as Escherichia coli and Staphylococcus aureus. This technology represents a pivotal shift away from archaic synthetic routes that plagued the sector with toxicity and inefficiency, offering instead a streamlined, five-step sequence that begins with the inexpensive and readily available p-methoxybenzoic acid. By leveraging modern condensation and cyclization techniques, this methodology not only enhances the structural diversity of accessible imidazoles but also addresses the urgent need for greener, more sustainable manufacturing processes in the production of antibacterial drugs.
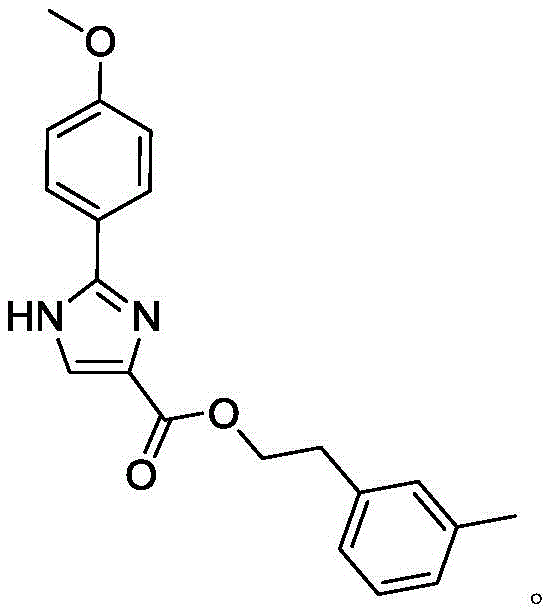
For R&D directors and process chemists, the structural integrity and purity of such intermediates are paramount, especially when considering their potential application in treating hospital sewage contamination or developing new antimicrobial therapies. The target molecule, characterized by a 2-(4-methoxyphenyl)-1H-imidazole-4-carboxylic acid core esterified with m-methyl phenethyl alcohol, demonstrates a unique pharmacological profile that warrants close attention from procurement teams looking to secure reliable pharmaceutical intermediate suppliers. The synthesis strategy outlined in the patent avoids the pitfalls of earlier methods, such as the low-yield Debus synthesis or the mercury-catalyzed routes, by employing a logical progression of amidation, thionation, and cyclization. This approach ensures that the final product meets stringent quality standards required for downstream drug development, making it an attractive candidate for companies aiming to expand their portfolio of high-value heterocyclic building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted imidazole rings has been fraught with significant technical and environmental challenges that hindered large-scale adoption. As early as 1858, the Debus method utilized ethanedione and formaldehyde but suffered from notoriously low yields, making it economically unviable for modern commercial production. Later attempts, such as the 2008 Kaila method, introduced the use of mercuric chloride as a catalyst to generate guanylguanidine intermediates; however, the high toxicity of mercury poses severe occupational health risks and creates a massive burden for waste treatment and environmental compliance. Furthermore, more recent approaches, like the 2017 Yu method, relied on expensive palladium catalysts to facilitate C-C coupling and C-N condensation tandem reactions. While effective on a small laboratory scale, the reliance on precious metals drastically inflates the cost of goods sold (COGS) and introduces supply chain vulnerabilities associated with fluctuating metal prices. These conventional methods often require harsh reaction conditions, prolonged reaction times exceeding 12 hours, and complex purification protocols to remove trace metal contaminants, all of which act as bottlenecks for cost reduction in antibacterial drug manufacturing.
The Novel Approach
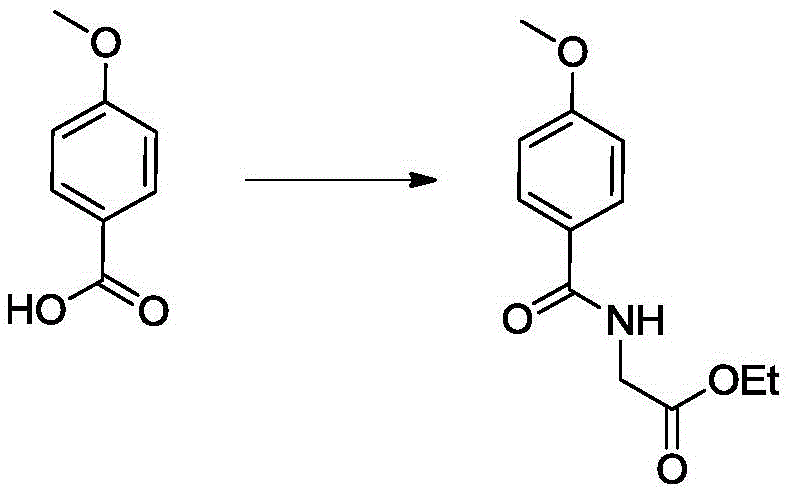
In stark contrast to these legacy techniques, the novel approach disclosed in the patent utilizes a mild, metal-free pathway that prioritizes atom economy and operational simplicity. Starting from p-methoxybenzoic acid, the process employs a condensation reagent to couple with ethyl aminoacetate, forming the initial amide backbone under nitrogen protection at room temperature. This is followed by a thionation step using Lawesson's reagent, which efficiently converts the carbonyl oxygen to sulfur, a critical transformation for the subsequent cyclization. The innovation lies in the careful control of reaction parameters, such as maintaining low temperatures during the methylation step to prevent side reactions, and utilizing formamide as a one-carbon source for ring closure. This strategy eliminates the need for toxic heavy metals entirely, thereby simplifying the downstream purification process and significantly reducing the environmental footprint. By adopting this route, manufacturers can achieve high-purity imidazole derivatives with a cleaner impurity profile, directly addressing the concerns of regulatory bodies and ensuring a safer product for eventual clinical or industrial application.
Mechanistic Insights into Formamide-Mediated Cyclization
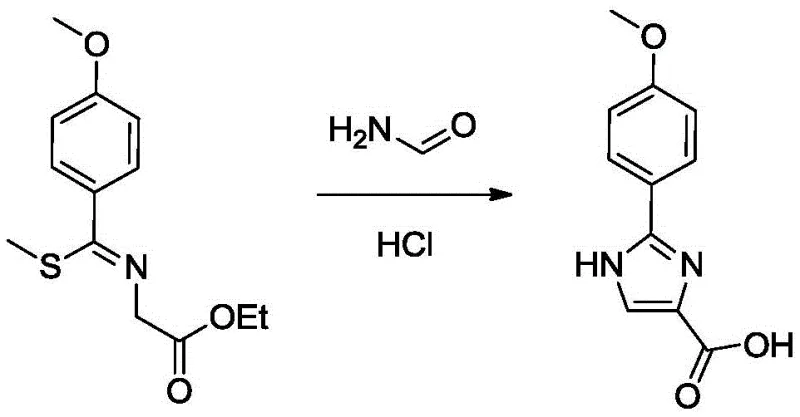
The cornerstone of this synthetic route is the fourth step, where the linear precursor undergoes intramolecular cyclization to form the imidazole ring, a transformation that requires precise mechanistic control to ensure high regioselectivity and yield. In this stage, the methylated thioimidate intermediate reacts with formamide in the presence of a strong non-nucleophilic base, such as sodium bis(trimethylsilyl)amide (NaHMDS), at cryogenic temperatures around -60°C. The mechanism likely involves the deprotonation of the formamide or the activation of the imidate carbon, facilitating a nucleophilic attack that closes the five-membered ring. The use of silyl amide bases is crucial here, as they provide the necessary basicity to drive the reaction forward without inducing unwanted hydrolysis of the sensitive ester moiety present in the molecule. Following the cyclization, a carefully controlled hydrolysis sequence is employed, where the pH is adjusted sequentially using hydrochloric acid and sodium bicarbonate to isolate the carboxylic acid form of the imidazole. This multi-stage pH manipulation is essential for precipitating the product while keeping soluble impurities in the aqueous phase, demonstrating a sophisticated understanding of solubility dynamics that is vital for achieving pharmaceutical-grade purity.
Furthermore, the impurity control mechanism inherent in this design is robust, primarily due to the specificity of the reagents chosen for each transformation. For instance, the use of Lawesson's reagent in the second step is highly selective for the amide carbonyl, minimizing the risk of reacting with the ether oxygen on the methoxy group. Similarly, the final esterification step utilizes thionyl chloride to activate the carboxylic acid, which then reacts with m-methyl phenethyl alcohol. This activation method is superior to using carbodiimides in this context because it avoids the formation of urea byproducts that can be difficult to separate from the final crystalline product. The entire sequence is designed to minimize the generation of isomeric byproducts, which is a common issue in imidazole synthesis where N-alkylation can occur at multiple positions. By strictly controlling the stoichiometry and temperature, particularly during the methylation and cyclization steps, the process ensures that the nitrogen atoms are positioned correctly within the ring, resulting in a single, well-defined isomer that exhibits the desired bactericidal activity.
How to Synthesize 2-(4-methoxyphenyl)-1H-imidazole-4-carboxylic Acid Ester Efficiently
Executing this synthesis requires strict adherence to the operational parameters defined in the patent to maximize yield and safety, particularly given the use of reactive reagents like thionyl chloride and strong bases. The process begins with the activation of p-methoxybenzoic acid, followed by a series of transformations that build complexity while maintaining functional group tolerance. Operators must ensure that moisture is excluded during the amidation and cyclization steps, typically by using dried solvents and nitrogen blankets, as water can hydrolyze the activated intermediates and reduce overall efficiency. The workup procedures, involving extractions with dichloromethane and ethyl acetate, are designed to partition the product effectively from inorganic salts and polar byproducts. Detailed standard operating procedures (SOPs) for each of the five steps are critical for reproducibility, especially when transitioning from gram-scale laboratory experiments to kilogram-scale pilot runs.
- React p-methoxybenzoic acid with ethyl aminoacetate using a condensation reagent like HBTU to form the amide intermediate.
- Convert the amide to a thioamide using Lawesson's reagent in refluxing tetrahydrofuran or toluene.
- Methylate the thioamide using iodomethane and a base like sodium ethoxide at low temperatures (-15 to -10°C).
- Cyclize the methylated intermediate with formamide using a silyl amide base (e.g., NaHMDS) followed by hydrolysis to form the imidazole acid.
- Perform final esterification with m-methyl phenethyl alcohol using thionyl chloride to obtain the target bactericidal molecule.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this synthetic route offers compelling advantages that directly impact the bottom line and supply chain resilience for pharmaceutical manufacturers. The elimination of precious metal catalysts like palladium and toxic heavy metals like mercury removes a significant cost center associated with both raw material procurement and hazardous waste disposal. Traditional methods often require specialized resin columns or complex chelation treatments to reduce metal residues to ppm levels acceptable for drug substances, adding time and expense to the production cycle. By contrast, this metal-free approach simplifies the purification train, allowing for faster batch turnover and reduced capital expenditure on specialized filtration equipment. Moreover, the starting materials, such as p-methoxybenzoic acid and ethyl glycinate, are commodity chemicals with stable global supply chains, reducing the risk of production delays caused by raw material shortages. This stability is crucial for maintaining continuous manufacturing operations and meeting the rigorous delivery schedules demanded by downstream drug developers.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of expensive catalytic systems with stoichiometric organic reagents that are significantly cheaper and easier to handle. The avoidance of palladium catalysts, which are subject to volatile market pricing and geopolitical supply constraints, stabilizes the variable cost of production. Additionally, the use of common organic solvents like ethanol, THF, and dichloromethane allows facilities to leverage existing solvent recovery infrastructure, further driving down operational expenses. The simplified workup procedures mean less labor is required for purification, and the higher selectivity of the reactions reduces the loss of valuable intermediates, leading to a substantial improvement in overall process mass intensity (PMI).
- Enhanced Supply Chain Reliability: Securing a consistent supply of critical intermediates is a top priority for supply chain heads, and this route excels in its reliance on widely available feedstocks. Unlike specialized reagents that may have single-source suppliers, the key components of this synthesis are produced by multiple chemical manufacturers globally, providing procurement teams with negotiating leverage and backup options. The robustness of the reaction conditions, which do not require extreme pressures or exotic equipment, means that the process can be easily transferred between different contract manufacturing organizations (CMOs) without significant re-validation. This flexibility ensures reducing lead time for high-purity API precursors and mitigates the risk of supply disruptions due to facility maintenance or regulatory audits at a specific vendor site.
- Scalability and Environmental Compliance: As regulatory scrutiny on pharmaceutical manufacturing intensifies, the environmental profile of a synthesis route becomes a key differentiator. This method generates waste streams that are easier to treat compared to those containing heavy metals, aligning with green chemistry principles and corporate sustainability goals. The scalability is proven by the use of standard unit operations such as stirred tank reactors and filtration units, facilitating the commercial scale-up of complex heterocyclic intermediates from pilot plants to multi-ton annual production capacities. The ability to recycle solvents and the reduced toxicity of reagents lower the compliance burden, making it easier to obtain the necessary environmental permits for expansion and ensuring long-term operational viability in strict regulatory jurisdictions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived from the specific advantages and operational details outlined in the patent documentation. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for their own production pipelines. The answers provided reflect the empirical data and procedural constraints observed during the development of this bactericidal imidazole molecule, offering a realistic view of its potential integration into existing manufacturing frameworks.
Q: Why is this synthesis method preferred over traditional Debus or Kaila methods?
A: Traditional methods often suffer from low yields or require highly toxic catalysts like mercuric chloride. This novel route utilizes mild conditions and avoids heavy metal contamination, ensuring higher purity and safer processing for pharmaceutical applications.
Q: What are the key cost drivers in this specific imidazole synthesis?
A: The primary cost drivers are the condensation reagents and the specific cyclization bases. However, by eliminating expensive palladium catalysts and toxic mercury salts, the overall waste treatment and raw material costs are significantly optimized compared to legacy processes.
Q: Is this process scalable for industrial production?
A: Yes, the process uses standard organic solvents like dichloromethane, THF, and ethanol, which are readily available on an industrial scale. The reaction conditions, such as room temperature amidation and controlled low-temperature methylation, are manageable in large-scale reactors with proper cooling systems.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imidazole Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing advanced synthetic technologies that balance performance with economic viability. Our team of expert process chemists has thoroughly analyzed the pathway described in CN110256357B and is fully prepared to support your development needs with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We understand that moving from a patent example to a commercial reality requires more than just following a recipe; it demands rigorous optimization of crystallization parameters, solvent recovery rates, and safety protocols. Our state-of-the-art facilities are equipped with stringent purity specifications and rigorous QC labs capable of detecting trace impurities at the ppm level, ensuring that every batch of imidazole intermediate we deliver meets the highest international standards for pharmaceutical use.
We invite you to collaborate with us to unlock the full potential of this bactericidal molecule for your drug discovery programs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that compares this novel route against your current supply options, highlighting specific areas where operational efficiencies can be realized. We encourage you to reach out today to discuss your specific requirements,索取 specific COA data, and review our comprehensive route feasibility assessments. Let us be your trusted partner in navigating the complexities of heterocyclic synthesis, ensuring a secure and cost-effective supply chain for your next-generation antibacterial therapies.
