Scalable One-Pot Synthesis of Fluralaner Intermediates for Veterinary Pharmaceutical Manufacturing
Introduction to Advanced Isoxazoline Synthesis
The global demand for effective ectoparasiticides in the veterinary sector has surged, driven by the widespread adoption of next-generation treatments like Fluralaner (commercially known as Bravecto). As the industry seeks to secure robust supply chains for these critical active pharmaceutical ingredients (APIs), the efficiency of intermediate synthesis becomes a paramount concern. Patent CN111675667A introduces a groundbreaking preparation method for isoxazoline anthelmintics that fundamentally reshapes the manufacturing landscape. This innovation transitions away from laborious, multi-step isolation processes toward a streamlined, solvent-based one-pot methodology. By integrating amidation condensation, substitution, and ring-closure reactions within a single reaction vessel, this technology not only simplifies the operational workflow but also drastically enhances the economic viability of producing high-purity veterinary drug intermediates. For procurement and technical leaders, understanding this shift is essential for evaluating long-term sourcing strategies and cost structures.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex isoxazoline derivatives relied on fragmented, multi-stage protocols that imposed significant burdens on production efficiency and cost. Traditional routes, as documented in prior art, often necessitated distinct reaction vessels for each transformation, requiring the isolation and purification of unstable intermediates after every step. A representative conventional pathway involves an initial oxidation step using composite salts like potassium iodide and potassium hydrogen persulfate, followed by extraction and drying, yielding a product with only moderate purity around 95.6%. Subsequent steps frequently demanded column chromatography for purification, a technique that is notoriously difficult to scale industrially due to high solvent consumption and low throughput. These legacy methods resulted in overall yields hovering near 50%, creating substantial material waste and extending lead times. The reliance on multiple workup procedures, including repeated extractions and evaporations, further compounded the operational complexity, making it challenging to maintain consistent quality control across large batches.
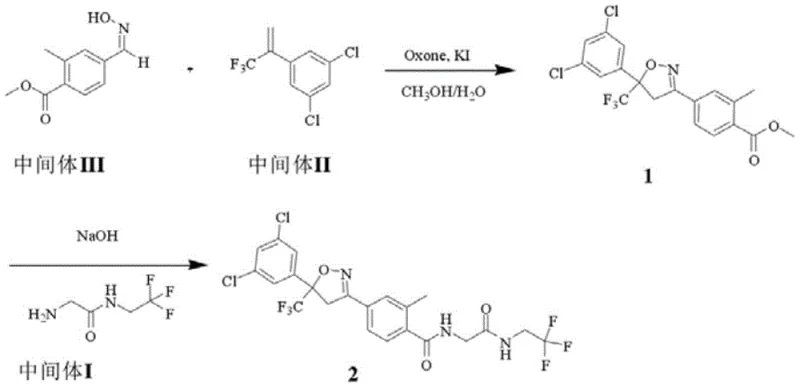
The Novel Approach
In stark contrast, the novel approach detailed in the patent leverages a sophisticated one-pot strategy that consolidates three critical chemical transformations into a unified process. This method eliminates the need for intermediate isolation, allowing the reaction mixture to proceed directly from amidation to substitution and finally to cyclization without interruption. By maintaining the reaction in a single solvent system, such as DMF or DMSO, the process minimizes solvent exchange and reduces the volume of waste generated. The elimination of column chromatography is a particularly transformative improvement, replaced instead by a simple water precipitation and recrystallization protocol that is inherently scalable. This streamlined workflow not only accelerates the production cycle but also ensures a higher retention of material, pushing molar yields to approximately 80% with purity levels exceeding 98.0%. The ability to execute these reactions at mild temperatures between 20-30°C further underscores the energy efficiency and safety profile of this modern synthetic route.
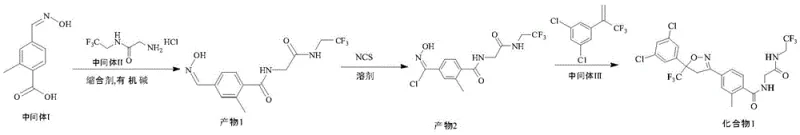
Mechanistic Insights into One-Pot Cascade Reaction
The success of this one-pot methodology relies on the precise orchestration of reaction kinetics and compatibility between reagents. The process initiates with an amidation condensation reaction where Intermediate I reacts with Intermediate II in the presence of a condensing agent like EDC hydrochloride and a catalyst such as HOBt. This step forms the foundational amide bond under mild conditions, facilitated by an organic base like triethylamine. Once the amidation is complete, monitored via TLC, the system transitions seamlessly into a substitution phase. Here, N-chlorosuccinimide (NCS) is introduced to chlorinate the oxime moiety, generating a reactive nitrile oxide precursor in situ. This intermediate is highly transient, which traditionally would require immediate capture; however, the one-pot design allows for the immediate addition of Intermediate III. The final ring-closing reaction involves a 1,3-dipolar cycloaddition, where the nitrile oxide reacts with the alkene group of Intermediate III to form the stable isoxazoline core. This cascade mechanism ensures that reactive species are consumed as soon as they are formed, minimizing side reactions and impurity formation.
Impurity control is intrinsically managed through the continuous nature of the reaction and the specific choice of reagents. By avoiding the exposure of intermediates to air and moisture during isolation, the potential for hydrolysis or degradation is significantly reduced. The use of specific developing solvents for TLC monitoring, such as a 1:1 ratio of petroleum ether to ethyl acetate, allows for real-time assessment of reaction progress, ensuring that each stage reaches completion before the next reagent is added. Furthermore, the final purification via recrystallization from solvents like petroleum ether and ethyl acetate effectively removes residual reagents and byproducts, such as succinimide from the NCS reaction. This rigorous control over the reaction environment and workup procedure results in a final product with a clean impurity profile, suitable for direct use in subsequent API synthesis without further extensive purification.
How to Synthesize Fluralaner Intermediate Efficiently
Implementing this advanced synthesis route requires careful attention to reagent stoichiometry and addition sequences to maximize yield and purity. The process begins by dissolving the starting materials in a polar aprotic solvent, followed by the sequential addition of coupling agents and bases to drive the amidation. Once the initial condensation is verified, the chlorinating agent is added portion-wise to manage exotherms and ensure complete conversion. Finally, the cyclization partner is introduced to close the heterocyclic ring. The detailed standardized synthesis steps, including specific molar ratios and temperature controls, are outlined in the guide below.
- Perform amidation condensation by reacting Intermediate I with Intermediate II using a condensing agent and organic base in a solvent like DMF.
- Execute a substitution reaction by adding N-chlorosuccinimide (NCS) directly to the mixture to chlorinate the oxime intermediate.
- Complete the ring-closing reaction by adding Intermediate III to form the isoxazoline core, followed by water precipitation and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this one-pot synthesis method offers compelling strategic advantages that extend beyond mere technical novelty. The consolidation of multiple reaction steps into a single vessel fundamentally alters the cost structure of manufacturing these high-value intermediates. By removing the need for intermediate isolation and column chromatography, the process drastically reduces the consumption of solvents and stationary phases, which are often significant cost drivers in fine chemical production. This reduction in material usage translates directly into lower variable costs per kilogram of product. Additionally, the simplified workflow decreases the demand for labor and equipment time, as fewer unit operations are required to bring the batch to completion. These efficiencies collectively contribute to a more competitive pricing model for the final intermediate, allowing buyers to negotiate better terms and secure more stable supply agreements.
- Cost Reduction in Manufacturing: The elimination of column chromatography and intermediate isolation steps removes some of the most expensive and time-consuming operations from the production line. Without the need for large volumes of elution solvents and silica gel, the direct material costs are significantly lowered. Furthermore, the reduced number of filtration and evaporation steps decreases energy consumption and equipment wear, leading to substantial operational savings. This lean manufacturing approach ensures that the cost of goods sold (COGS) is optimized, providing a buffer against raw material price fluctuations and enhancing overall margin stability for the supply chain.
- Enhanced Supply Chain Reliability: The simplified nature of the one-pot process inherently reduces the risk of production delays and bottlenecks. With fewer handoffs between different reaction stages and no dependence on slow purification techniques like chromatography, the cycle time for each batch is considerably shortened. This agility allows manufacturers to respond more rapidly to changes in demand, ensuring a consistent flow of material to downstream API producers. The robustness of the method, which tolerates mild reaction conditions and uses readily available reagents, further minimizes the risk of batch failures, thereby securing a reliable source of high-quality intermediates for long-term contracts.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces unforeseen challenges, but this one-pot method is designed with scalability in mind. The absence of complex purification steps makes it easier to transfer the process from pilot scale to commercial production without losing efficiency. Moreover, the significant reduction in solvent waste aligns with increasingly stringent environmental regulations and sustainability goals. By generating less hazardous waste and consuming fewer resources, manufacturers can operate with a smaller environmental footprint, reducing disposal costs and mitigating regulatory risks. This compliance advantage is crucial for maintaining uninterrupted operations in a highly regulated global market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this novel synthesis technology. These insights are derived directly from the patent specifications and practical considerations for industrial application, providing clarity for stakeholders evaluating this supply option.
Q: What is the primary advantage of the one-pot synthesis method for Fluralaner intermediates?
A: The primary advantage is the elimination of intermediate isolation and purification steps, specifically avoiding column chromatography, which significantly reduces solvent consumption, processing time, and overall production costs while improving yield.
Q: What represent the critical reaction conditions for this synthesis?
A: The process operates under mild conditions, typically between 20-30°C, utilizing common solvents like DMF or DMSO, and employs standard reagents such as EDC hydrochloride, HOBt, and NCS, making it highly adaptable for industrial scale-up.
Q: How does this method impact the purity profile of the final veterinary drug intermediate?
A: By minimizing handling and exposure to external contaminants through a closed one-pot system, and utilizing efficient recrystallization protocols, the method consistently achieves purity levels exceeding 98.0%, meeting stringent pharmaceutical standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluralaner Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of veterinary pharmaceuticals depends on the availability of high-quality, cost-effective intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global supply chains. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of Fluralaner intermediate adheres to the highest standards of quality and consistency. Our capability to implement advanced one-pot synthesis technologies allows us to offer a superior value proposition to our partners.
We invite you to collaborate with us to optimize your supply chain for isoxazoline anthelmintics. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your product development and commercialization goals efficiently.
