Advanced Telescoped Synthesis Strategy for High-Purity Meropenem Intermediates
Advanced Telescoped Synthesis Strategy for High-Purity Meropenem Intermediates
The pharmaceutical landscape for beta-lactam antibiotics continues to evolve, driven by the urgent need for cost-effective and scalable manufacturing processes for critical drugs like Meropenem. Patent CN100422184C introduces a sophisticated preparation method for this potent beta-methylcarbapenem antibiotic, addressing longstanding inefficiencies in prior art. The invention provides a robust chemical structure formula and concrete steps that streamline the synthesis from simple starting materials to the final active pharmaceutical ingredient. By focusing on telescoped reactions and minimizing isolation steps, this technology offers a compelling value proposition for manufacturers seeking to optimize their production lines. The strategic implementation of one-pot operations not only enhances operational efficiency but also significantly mitigates the environmental footprint associated with traditional multi-step syntheses.
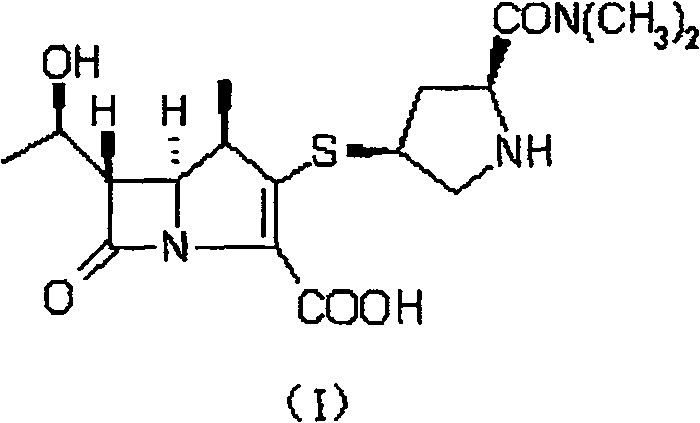
This technical breakthrough is particularly relevant for stakeholders aiming to secure a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials consistently. The patent outlines a comprehensive route that begins with readily available azetidinone derivatives and proceeds through a series of carefully orchestrated transformations. These include stereoselective alkylations, diazo transfers, and rhodium-catalyzed cyclizations, all designed to maximize atomic economy. For R&D directors and process chemists, the detailed methodology presented in CN100422184C serves as a blueprint for modernizing carbapenem production, ensuring that the final product meets stringent regulatory standards for impurity profiles and potency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of beta-methylcarbapenem antibiotics has been plagued by complex purification requirements and suboptimal yields. Prior art, such as the routes developed by Sumitomo Pharmaceuticals and Lederle Ltd., often necessitates the separation of numerous optical isomers, which drastically reduces overall throughput. For instance, conventional pathways frequently generate significant amounts of unwanted stereoisomers during the construction of the carbapenem nucleus, requiring resource-intensive chromatographic separations that are difficult to scale. Furthermore, the reliance on expensive or hard-to-obtain raw materials in older methodologies creates bottlenecks in the supply chain, leading to increased production costs and potential delays. The accumulation of solvent waste from multiple isolation steps also poses significant environmental compliance challenges, making these legacy processes less attractive for modern green chemistry initiatives.
The Novel Approach
In stark contrast, the novel approach detailed in the patent leverages a telescoped synthesis strategy that dramatically simplifies the workflow. By designing reaction sequences where the output of one step serves directly as the input for the next without intermediate purification, the process eliminates unnecessary handling and material loss. This methodology allows for the direct conversion of key precursors into the desired carbapenem core with superior stereocontrol, effectively bypassing the need for difficult chiral separations. The use of bulky inducing groups in the initial alkylation ensures that the overwhelming majority of the product forms in the correct beta-configuration, thereby enhancing yield. This streamlined logic not only accelerates the timeline from raw material to finished intermediate but also aligns perfectly with the goals of cost reduction in pharmaceutical manufacturing by minimizing solvent consumption and labor hours.
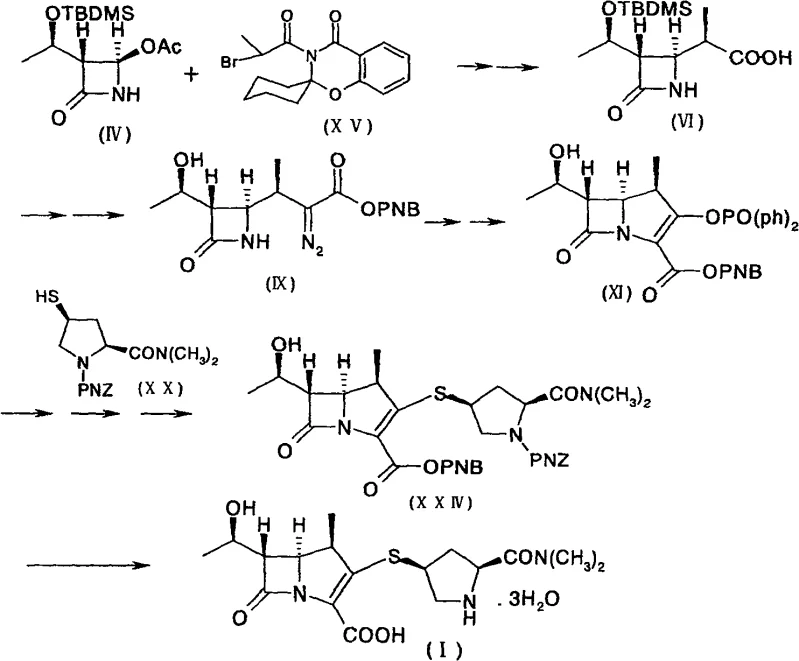
Mechanistic Insights into Rhodium-Catalyzed Cyclization and Phosphorylation
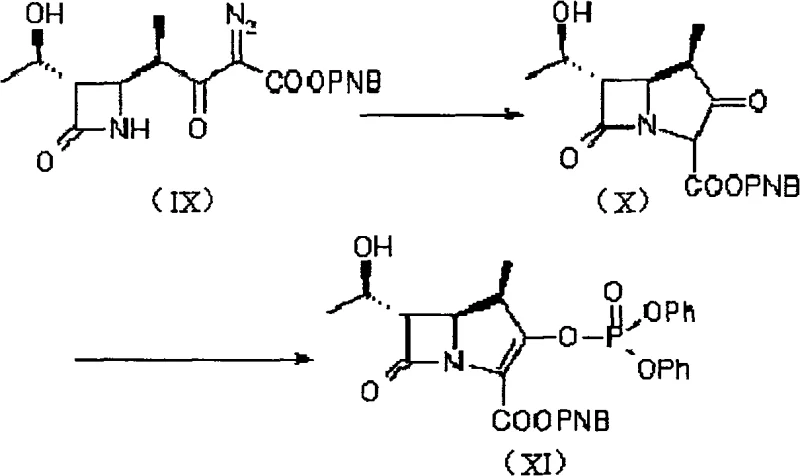
The heart of this innovative synthesis lies in the precise construction of the carbapenem ring system, specifically the transformation of the diazoketone precursor into the reactive enol phosphate. This critical sequence involves a rhodium-catalyzed intramolecular cyclization that forms the bicyclic core with high fidelity. The mechanism proceeds through the generation of a metal-carbenoid species from the diazo compound, which subsequently undergoes insertion into the adjacent C-H bond to close the five-membered ring. This step is meticulously controlled by temperature and catalyst loading, typically utilizing rhodium acetate at concentrations between 0.1% and 1% equivalent. The choice of solvent, such as ethyl acetate or tetrahydrofuran, plays a pivotal role in stabilizing the transition state and ensuring that the reaction proceeds smoothly without forming polymeric byproducts.
Following the cyclization, the resulting ketone is immediately subjected to phosphorylation to generate the enol phosphate, a highly reactive electrophile essential for the subsequent coupling with the thiol side chain. This tandem operation is executed in a single pot, preventing the degradation of the sensitive beta-lactam intermediate. The phosphorylation agent, typically diphenyl chlorophosphate, reacts with the enolate formed under basic conditions, locking the molecule into a configuration ready for nucleophilic attack. This mechanistic elegance ensures that the impurity profile remains clean, as the reactive intermediate is consumed as soon as it is formed. For technical teams, understanding this cascade is vital for troubleshooting and optimizing the process, as it highlights the importance of maintaining anhydrous conditions and precise stoichiometry to achieve the highest possible purity.
How to Synthesize Meropenem Efficiently
Implementing this synthesis requires strict adherence to the patented protocol to ensure reproducibility and safety. The process begins with the preparation of the key acid intermediate, followed by its conversion into the diazoketone, which serves as the substrate for the ring-closing reaction. Operators must be vigilant regarding temperature control during the exothermic cyclization step and the subsequent addition of the phosphorylating agent. The detailed standardized synthetic steps provided in the guide below outline the specific reagents, molar ratios, and workup procedures necessary to achieve the reported yields. By following these instructions, production teams can replicate the high-efficiency outcomes described in the patent, ensuring a consistent supply of high-quality intermediates for downstream formulation.
- Perform a stereoselective alkylation of 4-acetoxyazetidinone with a bromo-propionic amide derivative to form the key acid intermediate without isolating the spirocyclic byproduct.
- Convert the acid intermediate into a diazoketone via a three-step one-pot sequence involving activation, ketene formation, and diazo transfer.
- Execute a rhodium-catalyzed intramolecular cyclization followed immediately by phosphorylation to generate the reactive enol phosphate for final coupling.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this telescoped synthesis method offers profound benefits for procurement managers and supply chain heads looking to optimize their sourcing strategies. The primary advantage lies in the substantial reduction of operational complexity, which translates directly into lower manufacturing costs. By removing the need for multiple isolation and purification stages, the process significantly cuts down on solvent usage, energy consumption, and labor requirements. This efficiency gain allows suppliers to offer more competitive pricing without compromising on quality, making it an attractive option for large-scale API production. Furthermore, the reliance on commercially available starting materials reduces the risk of supply disruptions, ensuring a more resilient and reliable supply chain for critical antibiotic ingredients.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation steps fundamentally alters the cost structure of Meropenem production. Traditional methods often incur high expenses due to the loss of material during chromatography and crystallization, as well as the cost of disposing of large volumes of solvent waste. This new approach mitigates those costs by keeping the material in solution throughout the critical ring-forming stages. Additionally, the high stereoselectivity of the initial alkylation reduces the burden on downstream purification, further driving down the cost per kilogram. These cumulative savings create a significant margin improvement that can be passed on to customers or reinvested into capacity expansion.
- Enhanced Supply Chain Reliability: Supply chain stability is paramount in the pharmaceutical industry, where interruptions can have severe consequences. This synthesis route utilizes robust, widely available reagents such as trans-4-hydroxy-L-proline derivatives and standard azetidinones, avoiding the dependency on exotic or single-source chemicals. The simplified workflow also means that production cycles are shorter, allowing for faster turnaround times and more responsive inventory management. For procurement teams, this translates to reduced lead times and a lower risk of stockouts, ensuring that the continuous demand for beta-lactam antibiotics can be met without delay.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to industrial production often reveals hidden bottlenecks, but this method is designed with scalability in mind. The one-pot nature of the key steps reduces the equipment footprint required, as fewer reactors and separation units are needed. Moreover, the reduction in solvent waste aligns with increasingly stringent environmental regulations, minimizing the need for costly waste treatment infrastructure. This eco-friendly profile not only facilitates easier regulatory approval but also enhances the corporate sustainability credentials of the manufacturer, appealing to partners who prioritize green chemistry principles in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific advantages and procedural details outlined in the patent documentation, providing clarity for stakeholders evaluating this method. Understanding these nuances is essential for making informed decisions about process adoption and partnership opportunities. The answers reflect the practical realities of scaling this chemistry while maintaining the high standards required for pharmaceutical intermediates.
Q: How does the telescoped synthesis improve yield compared to conventional methods?
A: By eliminating intermediate isolation steps, particularly during the formation of the carbapenem core from the diazo precursor, the process minimizes material loss and degradation, significantly boosting overall yield.
Q: What are the key advantages regarding solvent usage in this patent?
A: The method utilizes common industrial solvents like ethyl acetate and acetonitrile and reduces the total volume required by carrying reaction mixtures directly into subsequent steps without concentration or extensive washing.
Q: Is the stereochemistry controlled effectively in this route?
A: Yes, the initial alkylation step employs a bulky inducing group to ensure high stereoselectivity for the beta-configuration, reducing the need for difficult chiral separations later in the process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Meropenem Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for life-saving antibiotics like Meropenem. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of the global market. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify every batch. By leveraging advanced technologies such as the telescoped synthesis described in CN100422184C, we are able to offer a level of quality and consistency that sets us apart as a leader in the fine chemical industry.
We invite potential partners to engage with our technical procurement team to discuss how our capabilities align with your specific project needs. Whether you require a Customized Cost-Saving Analysis for your existing supply chain or need to evaluate the feasibility of this novel route for your portfolio, we are ready to assist. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can drive value for your organization.
