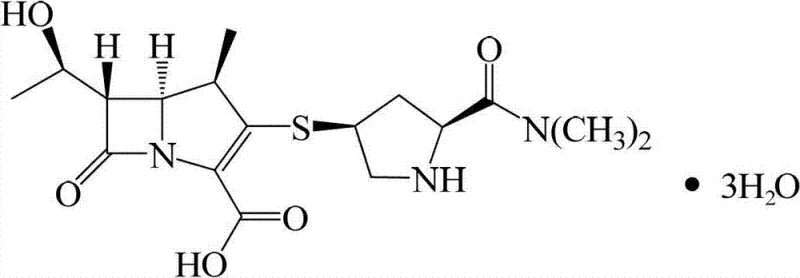
Advanced Formamide-Based Refining Technology for High-Purity Meropenem Production
The pharmaceutical landscape for broad-spectrum antibiotics demands not only potent active ingredients but also manufacturing processes that guarantee exceptional purity and stability. Patent CN102702203B introduces a transformative approach to the refining of Meropenem, a critical carbapenem antibiotic used for treating severe infections including meningitis and pneumonia. Traditional purification methods have long struggled with the inherent instability of the beta-lactam ring in Meropenem, often leading to significant yield losses and impurity profiles that fail to meet stringent pharmacopoeial standards. This new technology leverages the unique solvation properties of formamide to create a robust, high-yield refining pathway that addresses the core chemical vulnerabilities of the molecule. By shifting away from unstable protic solvents like methanol, this method offers a reliable meropenem supplier strategy that ensures consistent quality and operational efficiency.
For R&D directors and process chemists, the implications of this patent are profound, offering a solution to the chronic issue of hydrolysis during downstream processing. The ability to dissolve crude Meropenem in formamide without triggering degradation allows for a much wider operating window during crystallization. This stability is not merely a laboratory curiosity but a fundamental shift in how we approach the commercial scale-up of complex beta-lactams. The process eliminates the need for rapid, panic-induced processing speeds often required to outrun degradation kinetics in inferior solvent systems, thereby granting production teams greater control over crystal habit and particle size distribution.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of Meropenem has been plagued by the chemical fragility of the compound in common organic solvents. Prior art, such as methods utilizing acetone added to aqueous solutions or methanol-based systems, suffers from severe drawbacks that hinder industrial viability. In methanol, for instance, Meropenem exhibits poor stability, undergoing facile hydrolysis and polymerization if the operating time is not cut to an impractical minimum. This instability forces manufacturers to accept lower refining yields and higher levels of critical impurities, specifically ring-opening products and dimers. Furthermore, traditional aqueous methods often require massive volumes of anti-solvents like acetone to induce crystallization due to the low solubility of the drug in water, resulting in an unfavorable solvent-to-oil ratio that drives up waste disposal costs and energy consumption for solvent recovery.
The Novel Approach
The innovative method disclosed in the patent circumvents these issues by utilizing formamide as the primary dissolution medium. The inventors discovered that Meropenem is not only very easily dissolved in formamide but remains relatively stable within the formamide solution, resisting the hydrolysis and polymerization that characterize other solvent systems. This stability allows for a controlled crystallization process where a mixed crystallization agent—comprising organic solvents like isopropanol, acetone, or tetrahydrofuran mixed with water—is added to the formamide solution. This approach drastically reduces the consumption of dissolving and crystallizing agents compared to prior art. The result is a refined product with superior color and luster, significantly higher purity, and a yield that can be greatly improved even under original equipment conditions, making it ideal for cost reduction in API manufacturing.
Mechanistic Insights into Formamide-Mediated Stabilization
The core mechanistic advantage of this process lies in the interaction between the formamide solvent and the sensitive beta-lactam ring of the Meropenem molecule. In conventional protic solvents like methanol or water, the nucleophilic attack on the carbonyl carbon of the beta-lactam ring is facilitated, leading to ring-opening hydrolysis. Formamide, while polar and capable of hydrogen bonding, appears to provide a solvation shell that stabilizes the transition states or simply reduces the activity of water molecules enough to prevent this degradation during the dissolution phase. This kinetic stability is crucial because the refining process inherently takes time; the ability to hold the drug in solution at temperatures between 0°C and 25°C without significant degradation allows for thorough filtration and decolorization steps that would otherwise be impossible.
Furthermore, the crystallization mechanism is driven by the careful modulation of solubility through the addition of the organic solvent-water mixture. By controlling the mass ratio of the crystallization agent to the crude product (optimally between 9.0:1 and 12.0:1) and maintaining strict temperature controls (-10°C to 20°C), the process encourages the formation of a thermodynamically stable crystal lattice that excludes impurities. The slow addition of the crystallization agent, preferably via dripping, ensures that supersaturation is managed effectively, preventing the occlusion of mother liquor which often traps impurities like dimers. This precise control over the crystallization environment is what enables the final product to meet the rigorous specifications of the Japanese Pharmacopoeia (16th Edition), with ring-opening products and dimers both suppressed to levels not exceeding 0.30%.
How to Synthesize Meropenem Efficiently
The synthesis and subsequent purification of Meropenem require a delicate balance of solubility management and thermal control to preserve the integrity of the carbapenem core. The patented method simplifies this by establishing a clear, three-stage workflow that begins with the dissolution of the crude material in formamide, followed by a controlled anti-solvent crystallization, and concludes with efficient separation and drying. This route is designed to be robust against minor variations in processing time, a common source of batch failure in traditional methods. For detailed operational parameters regarding solvent ratios, temperature gradients, and agitation speeds, please refer to the standardized synthesis guide below.
- Dissolve crude Meropenem in formamide at 0-25°C with a mass ratio of 2.5-6.0: 1, optionally decolorizing with activated carbon.
- Add a crystallization agent (mixture of organic solvent like acetone/THF/isopropanol and water) to the formamide solution at -10 to 20°C.
- Separate the resulting crystals via filtration or centrifugation, wash with cold solvent, and vacuum dry to obtain high-purity Meropenem.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this formamide-based refining technology offers substantial strategic benefits that extend beyond simple yield improvements. The primary advantage lies in the drastic simplification of the solvent recovery and waste management infrastructure. Because the process utilizes significantly less solvent volume per kilogram of product compared to aqueous-acetone methods, the energy load on distillation columns is reduced, and the volume of hazardous waste requiring treatment is minimized. This directly translates to a lower cost of goods sold (COGS) and a smaller environmental footprint, aligning with modern green chemistry initiatives and reducing regulatory compliance burdens associated with solvent emissions.
- Cost Reduction in Manufacturing: The elimination of excessive solvent usage and the improvement in refining yield create a powerful economic engine for production. By avoiding the losses associated with hydrolysis and polymerization, manufacturers can extract more saleable product from the same amount of crude input. Additionally, the stability of the formamide solution removes the need for expensive, rapid-processing equipment or cryogenic conditions that might be required to stabilize the drug in other solvents. This operational simplicity reduces capital expenditure requirements and lowers the ongoing maintenance costs of the production line, ensuring a more competitive pricing structure for the final API.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by batch failures caused by process sensitivity. The robustness of the formamide method means that batches are less likely to be rejected due to out-of-specification impurity profiles, particularly regarding ring-opening products. This reliability allows for more accurate forecasting and inventory planning. Furthermore, the use of common, commercially available solvents like formamide, acetone, and isopropanol ensures that raw material sourcing remains stable and unaffected by the supply volatility often seen with more exotic or specialized reagents. This accessibility guarantees that production schedules can be maintained without interruption.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new variables that can compromise quality, but this method is inherently scalable due to its reliance on standard crystallization unit operations. The process does not require high-pressure reactors or complex catalytic systems, making it easy to transfer from pilot plant to multi-ton commercial production. From an environmental standpoint, the reduced solvent-to-oil ratio means less volatile organic compound (VOC) emission and lower energy consumption for solvent recycling. This aligns with increasingly strict global environmental regulations, future-proofing the manufacturing site against tighter emission standards and potential carbon taxes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this refining technology. They are derived from the specific experimental data and comparative examples provided in the patent documentation, focusing on the practical realities of switching from methanol-based to formamide-based systems. Understanding these nuances is critical for technical teams evaluating the feasibility of technology transfer.
Q: Why is formamide preferred over methanol for Meropenem purification?
A: Meropenem is chemically unstable in methanol solutions, prone to hydrolysis and polymerization which lowers yield and increases impurities. In contrast, Meropenem is highly soluble and remarkably stable in formamide, preventing degradation during the refining process.
Q: What are the critical impurity controls in this refining method?
A: The process specifically targets the reduction of ring-opening products, dimers, and unknown single impurities. By avoiding hydrolytic conditions, the method ensures ring-opening impurities remain below 0.30% and dimers below 0.30%, meeting strict pharmacopoeia standards.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method significantly reduces the solvent-to-oil ratio compared to traditional aqueous methods and operates under mild temperatures (0-25°C), making it highly energy-efficient and scalable for commercial manufacturing without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Meropenem Supplier
At NINGBO INNO PHARMCHEM, we recognize that the difference between a viable commercial product and a laboratory curiosity often lies in the refinement of the manufacturing process. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of advanced patents like CN102702203B are fully realized in the final product. We operate with stringent purity specifications and utilize rigorous QC labs to verify that every batch of Meropenem meets the highest international standards, including the suppression of critical impurities like dimers and ring-opening byproducts.
We invite you to collaborate with us to optimize your supply chain for high-purity carbapenem antibiotics. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that demonstrates how switching to this refined process can impact your bottom line. Contact us today to request specific COA data and route feasibility assessments tailored to your production volume requirements, and let us help you secure a stable, high-quality supply of this essential antibiotic.
