Advanced Synthesis of Diether Diphthalimide for High-Performance Polyetherimide Manufacturing
The chemical industry is constantly seeking more efficient pathways to synthesize high-performance polymer intermediates, and the preparation of diether diphthalimide stands as a critical bottleneck in the production of polyetherimide (PEI) resins. Patent CN109734646B introduces a transformative methodology that addresses long-standing inefficiencies in this synthetic route by leveraging the unique properties of ionic liquids within a non-polar solvent system. This innovation represents a significant leap forward for manufacturers aiming to produce high-purity electronic chemical intermediates with reduced environmental impact and operational complexity. By shifting away from traditional polar aprotic solvents, this technology enables a streamlined one-pot process that maintains high reaction kinetics while simplifying downstream purification. For R&D directors and process engineers, understanding the nuances of this catalytic system is essential for evaluating its potential integration into existing commercial scale-up of complex polymer additives pipelines.
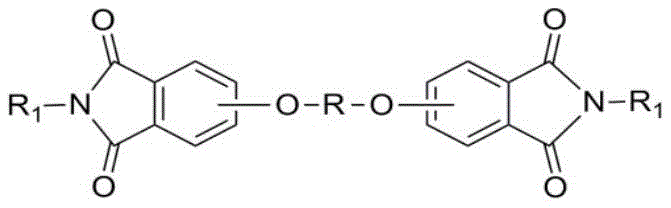
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of diether diphthalimide has relied heavily on the use of polar aprotic solvents such as dimethyl sulfoxide (DMSO), N,N'-dimethylformamide (DMF), or N,N'-dimethylacetamide (DMAC) to facilitate the nucleophilic substitution reaction. While these solvents are effective at solvating the phenolate salts and accelerating the reaction rate, they introduce severe downstream processing challenges that negatively impact overall manufacturing efficiency. These solvents possess high boiling points and poor thermal stability, making their removal energy-intensive and often resulting in product degradation if temperatures are not strictly controlled. Furthermore, their complete miscibility with water complicates the separation of inorganic salt byproducts, typically requiring large volumes of water to precipitate the product, which generates substantial wastewater and complicates solvent recovery efforts. Additionally, the conventional two-step process often requires an initial formation of phenolate in a non-polar solvent followed by a solvent swap, a procedure prone to issues such as phenolate agglomeration, wall-hanging, and uneven heating, all of which detrimentally affect reaction yield and reproducibility.
The Novel Approach
The methodology disclosed in the patent fundamentally reimagines this process by conducting the entire reaction sequence within a single non-polar solvent system, such as toluene or o-xylene, augmented by the addition of a specialized ionic liquid catalyst. This approach eliminates the cumbersome solvent exchange step, thereby preventing the physical handling issues associated with drying phenolate salts and ensuring a homogeneous reaction environment throughout the process. The ionic liquid functions as a highly effective phase transfer catalyst, bridging the solubility gap between the inorganic phenolate species and the organic phthalimide substrate within the non-polar medium. This results in a dramatic improvement in reaction conversion rates, with yields consistently exceeding 90%, while simultaneously simplifying the workup procedure. Post-reaction, the mixture can be treated with water to extract both the inorganic salts and the ionic liquid catalyst, leaving the desired product in the organic phase for easy isolation via solvent evaporation, which significantly reduces waste generation and enhances the overall sustainability profile of the manufacturing operation.
Mechanistic Insights into Ionic Liquid-Catalyzed Nucleophilic Substitution
The core of this technological advancement lies in the synergistic interaction between the ionic liquid catalyst and the reactants within the non-polar solvent matrix. In traditional systems, the phenolate salt exhibits poor solubility in non-polar media, leading to heterogeneous reaction conditions that limit mass transfer and reaction rates. The introduction of ionic liquids, such as 1-butyl-3-methylimidazolium hexafluorophosphate ([BMIM][PF6]) or tetrafluoroborate ([BMIM][BF4]), alters this dynamic by forming a micro-environment that stabilizes the phenolate anion and facilitates its transport to the organic phase where the nucleophilic attack occurs. This phase transfer mechanism ensures that the concentration of reactive species at the interface remains high, driving the substitution reaction forward with exceptional efficiency even at moderate temperatures ranging from 100°C to 150°C. The stability of the ionic liquid under these thermal conditions ensures that it remains active throughout the reaction duration without decomposing, thereby maintaining consistent catalytic activity.
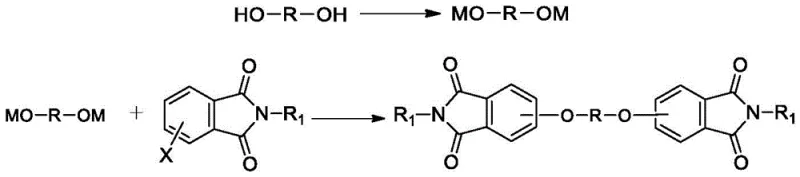
From an impurity control perspective, this mechanism offers distinct advantages by minimizing side reactions that are often promoted by harsh polar solvents or excessive thermal stress. The mild yet effective catalytic environment reduces the likelihood of hydrolysis or other degradation pathways that could compromise the structural integrity of the sensitive phthalimide moiety. Furthermore, the ability to remove the catalyst simply through aqueous extraction means that there is no risk of metal contamination or residual catalyst carryover, which is a critical quality attribute for intermediates destined for high-performance electronic materials or medical applications. The result is a product with purity levels surpassing 99%, achieved through a physically simple separation process that relies on the differential solubility of the components rather than complex chromatographic techniques, ensuring batch-to-batch consistency and reliability.
How to Synthesize Diether Diphthalimide Efficiently
The practical implementation of this synthesis route involves a straightforward protocol that begins with the formation of the phenolate salt in a non-polar solvent like o-xylene or toluene under an inert nitrogen atmosphere. Once the water generated during salt formation is removed via azeotropic distillation, the ionic liquid catalyst and the substituted phthalimide are introduced directly into the same reactor, avoiding any intermediate isolation steps. The reaction mixture is then heated to reflux conditions, typically between 110°C and 140°C, and maintained for a period of 4 to 10 hours to ensure complete conversion. Following the reaction, the crude mixture undergoes a filtration step to remove any insoluble particulates, after which the organic filtrate is washed with hot water to extract the ionic liquid and inorganic salts. The final isolation is achieved by evaporating the non-polar solvent under reduced pressure, yielding the target diether diphthalimide as a high-purity solid powder ready for subsequent polymerization steps.
- React diphenol raw materials with strong bases (NaOH/KOH) in a non-polar solvent like toluene or o-xylene under inert atmosphere to form the phenolate salt, removing water via azeotropic distillation.
- Add the phthalimide compound and an ionic liquid catalyst (e.g., [BMIM][PF6]) directly to the reaction mixture without changing solvents, and heat to 100-150°C to drive the nucleophilic substitution.
- Upon completion, filter the mixture, wash the organic layer with water to remove salts and the ionic liquid catalyst, and evaporate the solvent to obtain high-purity solid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this ionic liquid-catalyzed process presents a compelling value proposition centered around operational efficiency and cost optimization. By eliminating the need for expensive and difficult-to-recycle polar aprotic solvents, manufacturers can significantly reduce raw material costs and the associated expenses of solvent recovery infrastructure. The simplified workflow, which removes the solvent swap and drying steps, translates directly into reduced cycle times and lower energy consumption per kilogram of product produced. Moreover, the drastic reduction in wastewater generation aligns with increasingly stringent environmental regulations, mitigating the risk of compliance penalties and reducing the burden on waste treatment facilities. These factors collectively contribute to a more resilient and cost-effective supply chain capable of meeting the growing demand for high-performance polymer precursors.
- Cost Reduction in Manufacturing: The elimination of polar solvents like DMF and DMSO removes the significant overhead associated with their purchase, storage, and complex recovery processes, leading to substantial operational savings. Additionally, the ability to recycle the non-polar solvent (toluene or o-xylene) further drives down variable costs, while the high reaction yield minimizes raw material waste, ensuring that every kilogram of input contributes maximally to the final output.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as bisphenols and simple phthalimides, combined with a robust catalytic system, ensures a stable supply of raw materials that is less susceptible to market volatility. The simplified process flow reduces the number of unit operations required, decreasing the likelihood of equipment failure or process upsets that could disrupt production schedules and delay deliveries to downstream customers.
- Scalability and Environmental Compliance: The one-pot nature of this synthesis makes it inherently scalable, allowing for seamless transition from pilot plant to full commercial production without the need for major equipment reconfiguration. The significant reduction in hazardous waste and wastewater generation simplifies environmental permitting and reporting, positioning manufacturers as responsible stewards of sustainability while avoiding the logistical complexities of hazardous waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology. These insights are derived directly from the experimental data and process descriptions provided in the patent literature, offering a clear understanding of the method's capabilities and limitations for potential adopters.
Q: What is the primary advantage of using ionic liquids in this synthesis?
A: Ionic liquids act as highly efficient phase transfer catalysts in non-polar solvents, significantly increasing reaction yields to over 90% while eliminating the need for difficult-to-remove polar aprotic solvents like DMF or DMSO.
Q: How does this method improve product purity compared to traditional routes?
A: By utilizing a simple water extraction workup, both the inorganic salt byproducts and the ionic liquid catalyst are effectively removed from the organic phase, allowing the final product to achieve purity levels exceeding 99% without complex chromatography.
Q: Can this process accommodate different bisphenol structures?
A: Yes, the methodology is robust and adaptable to various diphenol substrates, including Bisphenol A, Bisphenol S, and biphenyl derivatives, making it versatile for synthesizing different grades of polyetherimide precursors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diether Diphthalimide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation polyetherimide materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of diether diphthalimide performs consistently in your polymerization processes. Our expertise in green chemistry and process optimization allows us to offer solutions that not only meet technical requirements but also align with your sustainability goals.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific production needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits inherent in this technology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is equipped with the most advanced and reliable chemical solutions available in the market.
