Advanced One-Step Synthesis of Sugammadex Sodium for Commercial Pharmaceutical Manufacturing
Advanced One-Step Synthesis of Sugammadex Sodium for Commercial Pharmaceutical Manufacturing
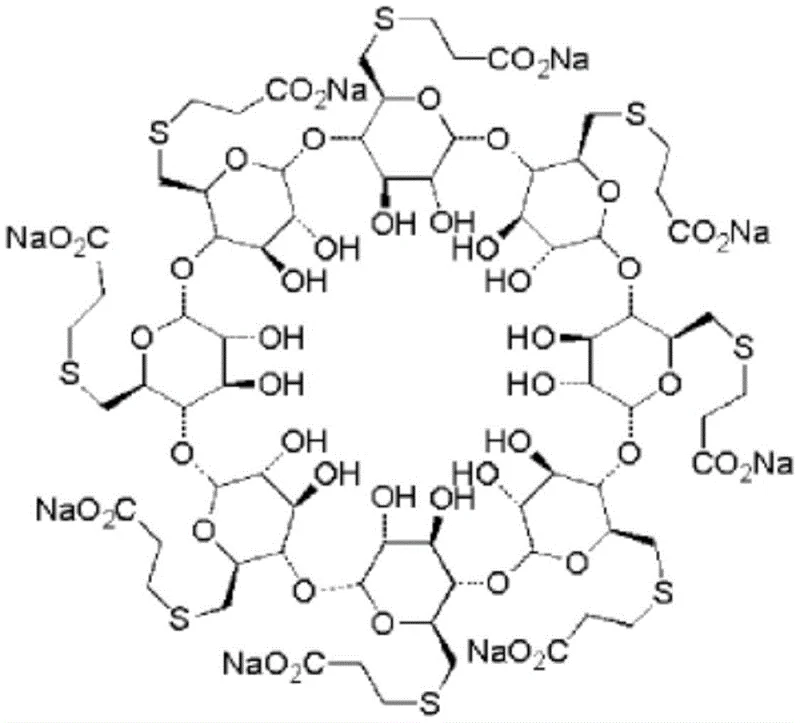
The pharmaceutical industry continuously seeks robust, scalable, and economically viable pathways for producing critical active pharmaceutical ingredients (APIs) and their intermediates. A significant breakthrough in this domain is detailed in patent CN109705237B, which discloses a novel preparation method for Sugammadex Sodium, a selective muscle relaxant antagonist widely used to reverse neuromuscular blockade induced by rocuronium bromide. This innovative protocol represents a paradigm shift from traditional multi-step halogenation processes to a streamlined, one-step synthesis involving the direct reaction of gamma-cyclodextrin with 3-mercaptopropionic acid in the presence of trifluoromethanesulfonic acid. By fundamentally altering the synthetic strategy, this method not only enhances the overall yield and purity of the final product but also addresses critical environmental and safety concerns associated with legacy manufacturing techniques. For global stakeholders, understanding the mechanistic advantages and commercial implications of this technology is essential for optimizing supply chain resilience and cost structures in the competitive landscape of anesthesia reversal agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Sugammadex Sodium has been plagued by complex, hazardous, and inefficient multi-step routes that pose significant challenges for industrial scale-up. Prominent prior art, such as the method disclosed in WO0140316, relies on a Vilsmeier-Hack reaction utilizing triphenylphosphine, iodine, and N,N-dimethylformamide to generate 6-per-deoxy-6-per-iodo-gamma-cyclodextrin, followed by substitution with 3-mercaptopropionic acid using sodium hydride. This approach introduces severe safety liabilities due to the use of pyrophoric sodium hydride and generates substantial toxic waste streams containing phosphorus and iodine residues. Furthermore, alternative routes described by Adam et al. involve bromination followed by esterification and hydrolysis, which often result in suboptimal purity levels around 80%, necessitating costly and time-consuming purification steps. Other methods employing triphosgene or oxalyl chloride for halogenation introduce additional toxicity risks and operational complexities, making them less desirable for modern green chemistry standards and large-scale commercial production where operator safety and environmental compliance are paramount.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the method outlined in patent CN109705237B offers a revolutionary one-step synthesis that dramatically simplifies the production workflow. By directly reacting gamma-cyclodextrin with 3-mercaptopropionic acid and trifluoromethanesulfonic acid, the process eliminates the need for discrete halogenation and subsequent substitution steps, thereby reducing the total number of unit operations and associated processing time. This direct thio-alkylation strategy operates under relatively mild conditions, typically between -50°C and 0°C, and utilizes common polar aprotic solvents such as DMF or DMSO, which are easily handled in standard stainless steel reactors. The absence of halogenating agents not only mitigates the risk of generating halogenated organic impurities but also significantly lowers the burden on wastewater treatment systems. Consequently, this novel approach provides a cleaner, safer, and more economically attractive pathway for manufacturing high-purity Sugammadex Sodium, aligning perfectly with the strategic goals of cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Triflic Acid-Catalyzed Thio-Alkylation
The core innovation of this synthesis lies in the utilization of trifluoromethanesulfonic acid (TfOH) as a potent activator for the cyclodextrin hydroxyl groups, facilitating a direct nucleophilic substitution by the thiol group of 3-mercaptopropionic acid. In this mechanism, the superacidic nature of TfOH likely protonates the hydroxyl groups at the C6 position of the gamma-cyclodextrin macrocycle, converting them into excellent leaving groups (water molecules) in situ. This activation allows the sulfur atom of the mercapto acid to attack the electrophilic carbon centers efficiently, forming the stable thioether linkage without the need for pre-functionalization via halides. The reaction is conducted at low temperatures, typically ranging from -40°C to -20°C, which is crucial for controlling the exothermicity of the acid-base interactions and preventing potential degradation of the sensitive cyclodextrin backbone. Following the substitution reaction, the mixture is quenched with an alkali metal solution containing sodium ions, such as sodium hydroxide or sodium methoxide, which neutralizes the acid and simultaneously converts the carboxylic acid side chains into their corresponding sodium salts, yielding the final water-soluble Sugammadex Sodium product.
From an impurity control perspective, this mechanism offers distinct advantages over halogen-based routes by minimizing the formation of poly-halogenated byproducts and partially substituted intermediates that are difficult to separate. Traditional methods often suffer from incomplete halogenation or over-halogenation, leading to a complex impurity profile that requires rigorous chromatographic purification to meet pharmacopeial standards. In the TfOH-catalyzed process, the reaction stoichiometry can be precisely tuned, with molar ratios of gamma-cyclodextrin to 3-mercaptopropionic acid to trifluoromethanesulfonic acid optimized between 1:5:5 and 1:30:30, preferably 1:8:16, to ensure complete per-substitution of the eight hydroxyl groups. The resulting crude product demonstrates high purity, often exceeding 95% to 98% as evidenced by experimental data, significantly reducing the load on downstream purification units. This enhanced selectivity ensures that the final API intermediate meets stringent quality specifications required for parenteral administration, thereby safeguarding patient safety and regulatory compliance.
How to Synthesize Sugammadex Sodium Efficiently
Implementing this advanced synthesis route requires careful attention to reaction parameters, particularly temperature control and reagent addition rates, to maximize yield and safety. The process begins by dissolving gamma-cyclodextrin in a suitable solvent like DMF under an inert nitrogen atmosphere, followed by the addition of 3-mercaptopropionic acid. The mixture is then cooled to cryogenic temperatures before the slow, dropwise addition of trifluoromethanesulfonic acid to manage the heat of reaction effectively. After the initial substitution phase is complete, the reaction is quenched with a base to adjust the pH to an alkaline range of 9-10, promoting salt formation and product precipitation. While the general workflow is straightforward, precise execution is key to reproducibility and quality assurance. For detailed standardized operating procedures and specific batch records, please refer to the technical guide below.
- React gamma-cyclodextrin with 3-mercaptopropionic acid and trifluoromethanesulfonic acid in a polar aprotic solvent at low temperature (-50°C to 0°C).
- Quench the reaction mixture using an alkali metal solution containing sodium ions, such as sodium hydroxide or sodium methoxide.
- Adjust pH to 9-10, heat to ambient temperature, and isolate the crude product via filtration and pulping.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis method translates into tangible strategic benefits that extend beyond simple chemical efficiency. By eliminating the reliance on hazardous and volatile reagents such as sodium hydride and elemental iodine, manufacturers can significantly reduce the costs associated with specialized storage, handling protocols, and hazardous waste disposal. The simplification of the process from a multi-step sequence to a one-pot reaction inherently reduces the consumption of solvents, energy, and labor hours, leading to a leaner manufacturing footprint. Furthermore, the avoidance of halogenated intermediates simplifies the regulatory filing process and reduces the risk of supply chain disruptions caused by the scarcity of specialized halogenating agents. These factors collectively contribute to a more resilient and cost-effective supply chain, ensuring consistent availability of high-quality Sugammadex Sodium for downstream formulation.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven primarily by the drastic reduction in raw material complexity and waste generation. By removing the need for expensive phosphorus and iodine reagents, as well as the pyrophoric sodium hydride, the direct material costs are substantially lowered. Additionally, the one-step nature of the reaction reduces utility consumption related to heating, cooling, and agitation across multiple stages, while the high crude purity minimizes the need for expensive recrystallization or chromatography steps. This holistic reduction in operational expenditure allows for a more competitive pricing structure without compromising on quality margins.
- Enhanced Supply Chain Reliability: Supply chain stability is greatly improved by the use of commodity chemicals that are readily available in the global market. Unlike specialized halogenating agents which may face supply bottlenecks, reagents like 3-mercaptopropionic acid and trifluoromethanesulfonic acid are produced at scale by numerous chemical suppliers, reducing the risk of single-source dependency. The simplified process flow also shortens the overall production cycle time, enabling manufacturers to respond more agilely to fluctuations in market demand and reducing the lead time for high-purity muscle relaxant antagonists.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method represents a significant advancement towards green chemistry principles. The absence of halogen usage eliminates the generation of halogenated organic waste, which is notoriously difficult and costly to treat. The process is inherently safer for commercial scale-up as it avoids the handling of pyrophoric solids in large quantities, thereby lowering insurance premiums and facility maintenance costs. This alignment with sustainable manufacturing practices not only meets current regulatory standards but also future-proofs the production facility against increasingly stringent environmental legislation.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this synthesis method, we have compiled answers to common inquiries regarding its implementation and performance. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing a transparent view of the technology's capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their own production lines or sourcing strategies.
Q: What are the primary safety advantages of this new Sugammadex Sodium synthesis method?
A: Unlike conventional methods that utilize hazardous reagents like sodium hydride (pyrophoric) or phosphorus/iodine combinations, this novel process employs trifluoromethanesulfonic acid in a controlled low-temperature environment, significantly reducing explosion risks and improving industrial safety profiles.
Q: How does the purity of Sugammadex Sodium produced via this method compare to prior art?
A: The one-step direct thio-alkylation approach minimizes the formation of intermediate byproducts common in multi-step halogenation routes, consistently achieving purity levels exceeding 95% without the need for complex chromatographic purification.
Q: Is this process scalable for commercial API production?
A: Yes, the elimination of hazardous solid reagents and the use of standard polar aprotic solvents like DMF or DMSO make this route highly amenable to large-scale reactor operations, facilitating easier heat transfer and mixing compared to heterogeneous slurry reactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sugammadex Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthetic methodologies to maintain a competitive edge in the global pharmaceutical market. Our team of expert chemists has thoroughly analyzed the potential of the triflic acid-catalyzed route for Sugammadex Sodium and is fully prepared to support its transition from laboratory scale to industrial production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel process are realized in practical, large-volume manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee that every batch meets stringent purity specifications, delivering a product that is safe, effective, and compliant with international pharmacopeial standards.
We invite pharmaceutical companies and contract manufacturing organizations to collaborate with us to leverage this technological advancement for their supply chains. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in complex cyclodextrin modification can drive value and efficiency for your business.
