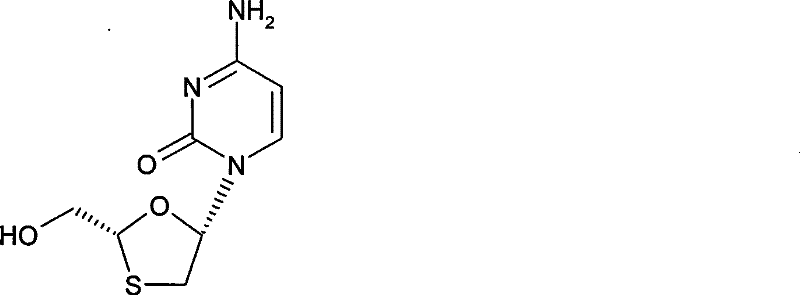
Advanced Stereoselective Manufacturing of Lamivudine Intermediates for Global Pharmaceutical Supply Chains
The pharmaceutical industry continuously seeks robust manufacturing pathways for critical antiretroviral agents, and the stereoselective preparation of Lamivudine represents a significant advancement in this domain. As disclosed in patent CN101307048B, this innovative methodology addresses the longstanding challenges associated with chirality control in nucleoside analogue synthesis. Lamivudine, chemically known as (2R-cis)-4-amino-1-(2-hydroxymethyl-1,3-oxathiolan-5-yl)-1H-pyrimidin-2-one, is a potent inhibitor of HIV and Hepatitis B virus replication. The biological activity is strictly dependent on the 2R,5S configuration, making the exclusion of unwanted enantiomers and diastereomers paramount for patient safety and efficacy. This patent introduces a strategic approach where the chiral configuration at the 2-position is established prior to the glycosylation step, fundamentally altering the purification landscape.
By employing a chiral auxiliary such as L-menthyl chloroformate, the synthesis generates diastereomeric intermediates rather than enantiomeric mixtures. This distinction is critical because diastereomers possess different physical properties, allowing for their separation through standard crystallization techniques rather than relying on expensive and low-throughput chiral chromatography. The process involves a glycosylation reaction between a specifically configured oxathiolane derivative and protected cytosine, followed by a crystallization separation to isolate the intermediate of formula (II). Subsequent deprotection yields the final high-purity Lamivudine. This route not only simplifies the operational workflow but also ensures that the final product meets stringent pharmacopeial standards for optical purity, a key requirement for regulatory approval in global markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
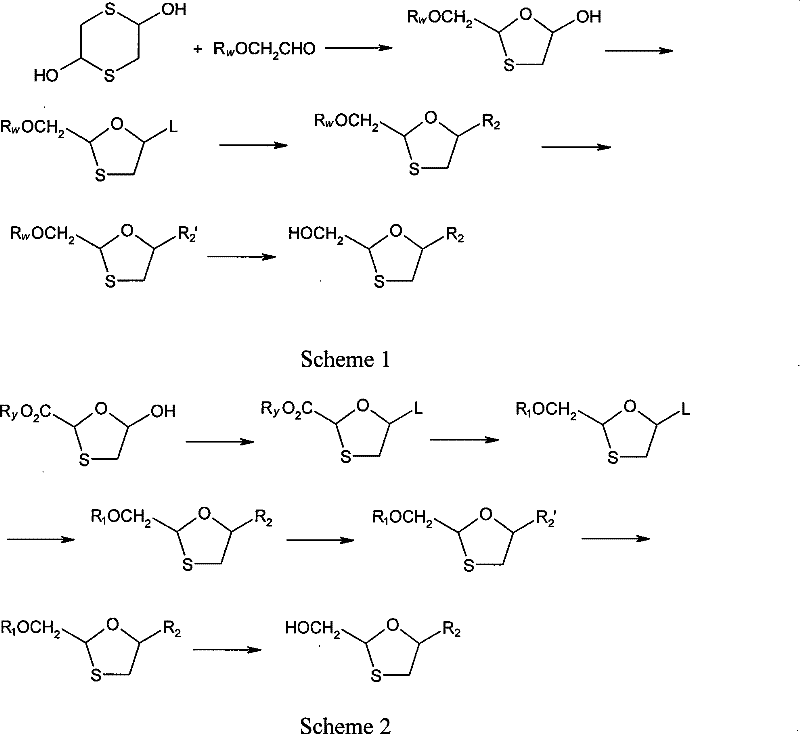
Historically, the synthesis of Lamivudine has been plagued by issues related to stereochemical control and purification complexity. Early methods, such as those referenced in WO94/14802, often involved non-stereoselective routes that produced racemic mixtures or complex blends of isomers. In these conventional pathways, the separation of the biologically active 2R,5S isomer from its inactive or toxic counterparts typically necessitated column chromatography. This reliance on chromatographic separation presents severe bottlenecks for industrial manufacturing, including high solvent consumption, low throughput, and significant difficulties in scaling up to multi-ton production levels. Furthermore, the lack of inherent chirality control in the starting materials often leads to lower overall yields, as a substantial portion of the synthesized material consists of unwanted isomers that must be discarded or recycled through energy-intensive processes.
The Novel Approach
In stark contrast to these legacy methods, the stereoselective approach detailed in CN101307048B introduces chirality at an earlier stage using a chiral auxiliary group. By attaching a bulky chiral moiety, such as an L-menthyl carbonate, to the oxathiolane ring before the glycosidic bond formation, the subsequent reaction with cytosine produces diastereomers. These diastereomers exhibit distinct solubility profiles, enabling their separation via simple recrystallization from alcoholic solvents like ethanol. This shift from chromatography to crystallization is a game-changer for process chemistry, drastically reducing processing time and solvent waste. The method ensures that the desired 2R,5S configuration is enriched efficiently, providing a streamlined path to high-purity intermediates that can be easily converted to the final API. This approach effectively decouples the complexity of chiral separation from the final purification steps, enhancing overall process robustness.
Mechanistic Insights into Stereoselective Glycosylation
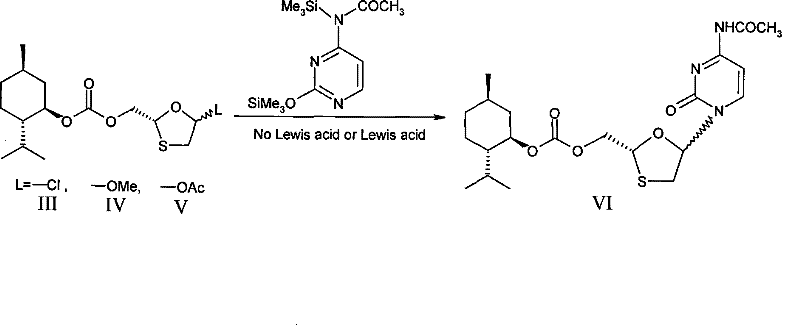
The core of this technological breakthrough lies in the precise manipulation of stereochemistry during the glycosylation event. The mechanism begins with the preparation of a chiral oxathiolane precursor, where the hydroxyl group is protected with a chiral carbonate derived from L-menthol. This bulky group exerts a steric influence that directs the approach of the silylated cytosine nucleophile during the glycosylation reaction. When the protected cytosine, activated by silylation agents like hexamethyldisilazane, reacts with the chiral oxathiolane intermediate, the transition state is biased towards the formation of specific diastereomers. The presence of the chiral auxiliary breaks the symmetry of the reaction environment, ensuring that the newly formed glycosidic bond adopts the desired orientation relative to the existing chiral center. This pre-organization minimizes the formation of unwanted alpha-anomers or incorrect stereoisomers at the source, rather than trying to fix them post-reaction.
Following the coupling reaction, the mixture contains diastereomers which differ in their three-dimensional arrangement due to the attached menthyl group. Unlike enantiomers, which have identical physical properties in an achiral environment, these diastereomers have different melting points and solubilities. The patent specifies that crystallization from ethanol allows for the selective precipitation of the desired isomer, leaving impurities in the mother liquor. This thermodynamic control is far superior to kinetic separations often required in racemic synthesis. Once the pure diastereomeric intermediate is isolated, the chiral auxiliary is removed under mild basic hydrolysis conditions, typically using potassium carbonate or macromolecular bases. This deprotection step reveals the free hydroxymethyl group and the native cytosine base, yielding Lamivudine with high optical purity and minimal contamination from the 2R,5R enantiomer or other diastereomeric impurities.
How to Synthesize Lamivudine Efficiently
The synthesis of Lamivudine via this stereoselective route involves a sequence of well-defined chemical transformations designed for scalability and purity. The process initiates with the activation of the sugar mimic using a chiral carbonate, followed by coupling with a protected nucleobase. The critical purification occurs through crystallization, leveraging the physical property differences of diastereomers. Finally, global deprotection yields the active pharmaceutical ingredient. For detailed operational parameters, stoichiometry, and specific temperature profiles required to replicate this high-yield process, please refer to the standardized synthesis guide below.
- Prepare the chiral carbonate intermediate by reacting a hydroxyl-protected oxathiolane precursor with L-menthyl chloroformate to establish stereochemistry.
- Perform glycosylation using silylated protected cytosine and the chiral intermediate under mild heating conditions to form the nucleoside bond.
- Separate the resulting diastereomeric mixture through simple crystallization in alcoholic solvents to isolate the desired 2R,5S configuration.
- Remove the protecting groups via hydrolysis using macromolecular bases and organic acid salification to yield the final high-purity Lamivudine API.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this stereoselective synthesis route offers tangible benefits that extend beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the purification train. By eliminating the need for preparative column chromatography, manufacturers can significantly reduce solvent usage and waste disposal costs, which are major components of the cost of goods sold (COGS) in fine chemical manufacturing. The ability to purify intermediates through crystallization also implies a more continuous and predictable production flow, reducing batch cycle times and increasing facility throughput. This efficiency translates directly into a more competitive pricing structure for the final API intermediate, allowing pharmaceutical companies to optimize their raw material spend without compromising on quality standards.
- Cost Reduction in Manufacturing: The replacement of expensive Lewis acid catalysts, such as trifluoromethanesulfonic acid trimethyl silicane alcohol ester, with more economical alternatives like Iodotrimethylsilane represents a significant raw material cost saving. Additionally, the avoidance of silica gel columns and the associated large volumes of elution solvents reduces both material costs and environmental compliance expenses. The process relies on standard unit operations like crystallization and filtration, which are inherently cheaper to operate than chromatographic separations, leading to substantial overall cost reductions in Lamivudine manufacturing.
- Enhanced Supply Chain Reliability: The robustness of crystallization-based purification ensures consistent batch-to-batch quality, a critical factor for maintaining uninterrupted drug supply. Since the separation of diastereomers is less sensitive to minor fluctuations in reaction conditions compared to chiral chromatography, the risk of batch failure is minimized. This reliability allows supply chain planners to forecast production timelines with greater accuracy, reducing the need for excessive safety stock and mitigating the risk of shortages in the global market for HIV and Hepatitis B treatments.
- Scalability and Environmental Compliance: The mild reaction conditions described in the patent, often operating at temperatures between 10°C and 80°C, are easily manageable in large-scale reactors without requiring specialized cryogenic or high-pressure equipment. The reduction in hazardous solvent waste and the elimination of heavy metal or complex catalyst residues simplify the wastewater treatment process. This alignment with green chemistry principles facilitates easier regulatory approval for manufacturing sites and supports corporate sustainability goals, making the supply chain more resilient to evolving environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this stereoselective Lamivudine synthesis. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and chemical mechanisms for potential partners and technical evaluators. Understanding these details is essential for assessing the feasibility of technology transfer and the potential for long-term supply agreements.
Q: How does this stereoselective method improve purity compared to traditional racemic synthesis?
A: Traditional methods often produce racemic mixtures requiring difficult column chromatography for separation. This patent introduces a chiral auxiliary (L-menthol) prior to glycosylation, creating diastereomers that can be easily separated via simple crystallization, significantly enhancing optical purity without complex equipment.
Q: What are the cost advantages of using Iodotrimethylsilane in this process?
A: The process allows for the substitution of expensive trifluoromethanesulfonic acid trimethyl silicane alcohol ester Lewis acid catalysts with the much lower-cost Iodotrimethylsilane. This substitution drastically reduces raw material costs while maintaining high reaction efficiency and stereoselectivity.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the method is specifically designed for industrial suitability. It utilizes mild reaction conditions, avoids harsh reagents, and replaces labor-intensive column chromatography with scalable crystallization steps, ensuring consistent quality and easier scale-up from pilot to commercial tonnage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lamivudine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the production of life-saving antiretroviral therapies. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated stereoselective chemistry described in CN101307048B can be successfully translated from the laboratory to the plant. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced chiral HPLC capabilities to verify the optical purity of every batch, guaranteeing that our Lamivudine intermediates meet the exacting standards required by global regulatory bodies.
We invite pharmaceutical manufacturers and procurement leaders to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized stereoselective processes, we can help you secure a stable supply of high-quality API intermediates while optimizing your overall production costs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can support your strategic supply chain objectives.
