Advanced Synthetic Route for Lamivudine: Enhancing Purity and Scalability for Global API Markets
Advanced Synthetic Route for Lamivudine: Enhancing Purity and Scalability for Global API Markets
The global demand for high-quality antiviral agents continues to surge, placing immense pressure on supply chains to deliver active pharmaceutical ingredients (APIs) with uncompromising purity and cost efficiency. In this context, the synthetic methodology disclosed in patent CN110437216B represents a significant technological leap forward for the production of Lamivudine, a critical nucleoside analogue used in the treatment of HIV and Hepatitis B. This proprietary approach fundamentally reengineers the classic synthesis by leveraging L-menthol as a robust chiral auxiliary, effectively bypassing the need for expensive enzymatic resolutions or complex sugar-based starting materials that have historically plagued the industry. By shifting the paradigm towards a more linear, chemically driven stereocontrol mechanism, this process offers a compelling value proposition for reliable pharmaceutical intermediates supplier networks seeking to optimize their portfolios. The technical depth of this invention lies not just in the final yield, but in the strategic selection of reagents that balance reactivity with environmental safety, ensuring that the resulting supply chain is both resilient and economically viable for long-term commercial partnerships.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial manufacture of Lamivudine has been hindered by several persistent technical bottlenecks that inflate costs and complicate regulatory compliance. Traditional routes often rely on the resolution of racemic mixtures using chiral enzymes or expensive chromatographic separations, which inherently cap the maximum theoretical yield at 50% unless dynamic kinetic resolution is employed, a feat that is difficult to achieve consistently on a multi-ton scale. Furthermore, many legacy processes utilize harsh reaction conditions involving strong acids or unstable intermediates that require cryogenic temperatures, leading to high energy consumption and significant safety risks in plant operations. The reliance on protected sugar derivatives as starting materials also introduces multiple protection and deprotection steps, each adding to the overall step count, reducing atom economy, and generating substantial volumes of solvent waste that require costly disposal. These factors collectively result in a high cost of goods sold (COGS) and a fragile supply chain that is vulnerable to raw material shortages, making it difficult for procurement teams to secure stable pricing for high-purity antiviral intermediates in a volatile market.
The Novel Approach
In stark contrast, the methodology outlined in the referenced patent introduces a streamlined seven-step sequence that prioritizes atom economy and operational simplicity without sacrificing stereochemical integrity. By initiating the synthesis with readily available dihaloacetic acid and L-menthol, the process establishes the chiral center early through a highly selective esterification and subsequent condensation with 2,5-dihydroxy-1,4-dithiane. This strategy eliminates the need for late-stage resolution, as the chirality is dictated by the inexpensive L-menthol auxiliary which can be easily recovered and recycled, thereby drastically reducing raw material costs. The reaction conditions are notably mild, operating within a manageable temperature range of -10°C to 130°C across the various stages, which allows for the use of standard stainless steel reactors rather than specialized cryogenic equipment. This simplification of the process infrastructure directly translates to cost reduction in API manufacturing, enabling producers to offer more competitive pricing while maintaining the rigorous quality standards required by global health authorities for antiviral therapeutics.
Mechanistic Insights into L-Menthol Mediated Chiral Induction
The core innovation of this synthetic route lies in the precise orchestration of stereochemistry during the formation of the oxathiolane ring, a critical structural motif in Lamivudine. The process begins with the condensation of dihaloacetic acid and L-menthol, catalyzed by DCC and DMAP, to form menthyl dihaloacetate, which is subsequently hydrolyzed to menthyl glyoxylate. This intermediate serves as the electrophilic partner in the pivotal condensation step with 2,5-dihydroxy-1,4-dithiane. Under the influence of the bulky chiral environment provided by the menthyl ester group, the condensation proceeds with high diastereoselectivity to favor the trans-configuration of the resulting 5-hydroxy-1,3-oxathiolane-2-carboxylic acid menthyl ester. This stereocontrol is essential, as the biological activity of Lamivudine is strictly dependent on the (-)-(2R,5S) enantiomer. Following this, the hydroxyl group is halogenated using triphosgene, creating a reactive leaving group that facilitates the subsequent nucleophilic substitution with silylated cytosine. The use of hexamethyldisilazane (HMDS) to protect the cytosine amine ensures that the coupling occurs exclusively at the N1 position, preventing the formation of regioisomeric impurities that are notoriously difficult to remove.
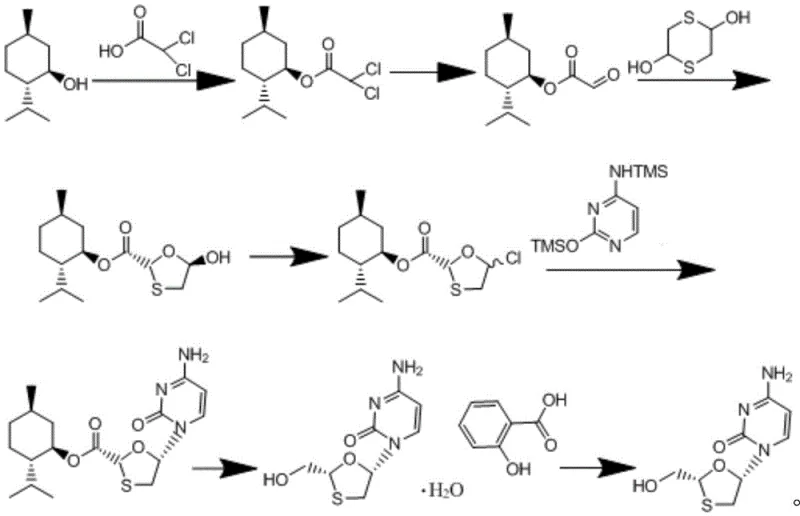
Following the coupling reaction, the menthyl ester protecting group is removed via reduction using potassium borohydride (KBH4), which simultaneously reduces the ester to the primary alcohol required for the final API structure. The crude product is then converted into a salicylate salt, a clever purification strategy that leverages the differential solubility of the salt form to exclude remaining impurities and unreacted starting materials. Finally, the free base is liberated through recrystallization in the presence of a base like triethylamine, yielding optically pure Lamivudine. Throughout this cascade, the choice of solvents such as toluene, dichloromethane, and ethanol is optimized for both solubility and ease of recovery, minimizing the environmental footprint. The mechanistic elegance of this route ensures that impurity profiles are tightly controlled, addressing the primary concerns of R&D directors regarding the stability and safety of the final drug substance.
How to Synthesize Lamivudine Efficiently
Implementing this advanced synthetic pathway requires a disciplined approach to process parameters to maximize yield and optical purity at every stage. The protocol is designed to be robust, tolerating minor variations in reagent addition rates while maintaining strict control over temperature and pH levels during the critical hydrolysis and salification steps. Operators must ensure that the silylation of cytosine is complete before introducing the halogenated oxathiolane intermediate to prevent side reactions that could compromise the coupling efficiency. Detailed standard operating procedures (SOPs) should emphasize the recycling of the L-menthol byproduct, as this is a key driver of the process's economic advantage. For a comprehensive breakdown of the specific molar ratios, reaction times, and workup procedures validated in the patent examples, please refer to the technical guide below.
- Condense dihaloacetic acid with L-menthol using DCC/DMAP catalyst to form menthyl dihaloacetate, followed by hydrolysis to generate menthyl glyoxylate.
- Perform chiral condensation with 2,5-dihydroxy-1,4-dithiane under acidic conditions to induce trans-stereochemistry in the oxathiolane ring.
- Halogenate the hydroxyl group, couple with silylated cytosine, reduce the ester, and finalize with salicylic acid salification and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this L-menthol based synthesis offers tangible strategic benefits that extend beyond simple unit price reductions. The reliance on commodity chemicals like dihaloacetic acid and L-menthol decouples the production of Lamivudine from the volatile pricing of specialized chiral sugars or enzymes, creating a more predictable cost structure. Furthermore, the high selectivity of the reaction minimizes the formation of difficult-to-remove impurities, which reduces the burden on quality control laboratories and shortens the batch release cycle time. This efficiency is crucial for maintaining continuous supply lines, especially when facing sudden spikes in demand for antiviral medications. The process is also inherently greener, generating less hazardous waste and utilizing solvents that are easier to recover and reuse, aligning with the increasingly stringent environmental regulations faced by modern chemical manufacturers.
- Cost Reduction in Manufacturing: The elimination of expensive enzymatic resolution steps and the use of low-cost starting materials significantly lower the direct material costs associated with production. By avoiding transition metal catalysts in the key coupling stages, the process also removes the need for costly metal scavenging and validation steps, further streamlining the manufacturing budget. The ability to recycle the chiral auxiliary adds another layer of economic efficiency, ensuring that the overall cost of goods is minimized without compromising on the quality of the final active ingredient.
- Enhanced Supply Chain Reliability: Sourcing raw materials for this process is straightforward, as L-menthol and dihaloacetic acids are produced globally in large volumes, reducing the risk of supply disruptions. The robustness of the chemical steps means that production can be scaled up rapidly in response to market needs without the long lead times typically associated with sourcing specialized biocatalysts. This reliability makes the supply chain more resilient to external shocks, ensuring that downstream pharmaceutical partners can maintain their own production schedules without interruption.
- Scalability and Environmental Compliance: The reaction conditions are well-suited for large-scale batch processing, utilizing standard equipment found in most multipurpose API plants. The mild temperatures and ambient pressure requirements reduce energy consumption and safety risks, facilitating easier regulatory approval for new manufacturing sites. Additionally, the reduced generation of hazardous waste simplifies waste management protocols, helping companies meet their sustainability goals and comply with evolving environmental standards in key markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic route. They are derived from the specific advantages and operational details highlighted in the patent documentation, providing clarity for stakeholders evaluating this technology for potential integration into their supply chains. Understanding these nuances is essential for making informed decisions about process adoption and vendor qualification.
Q: How does the L-menthol route improve chirality control compared to traditional methods?
A: The process utilizes L-menthol as a chiral pool auxiliary which induces high stereoselectivity during the condensation with 2,5-dihydroxy-1,4-dithiane, ensuring the formation of the desired trans-configuration without requiring complex enzymatic resolution steps.
Q: What are the primary cost drivers eliminated in this synthetic pathway?
A: By starting with inexpensive dihaloacetic acid and L-menthol instead of protected sugar derivatives, and avoiding transition metal catalysts in the key coupling steps, the raw material costs and downstream purification burdens are significantly reduced.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the reaction conditions are mild (ranging from -10°C to 130°C), utilize common organic solvents like toluene and dichloromethane, and generate minimal hazardous waste, making it highly adaptable for multi-ton commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lamivudine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to a more efficient synthetic route requires a partner with deep technical expertise and a proven track record in process development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent can be fully realized in a practical manufacturing setting. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to handle complex chiral syntheses allows us to offer a stable and high-quality supply of Lamivudine and its key intermediates, mitigating the risks associated with process variability.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic impact of switching to this L-menthol mediated route. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your volume needs, ensuring that your supply chain is both cost-effective and resilient against future market fluctuations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
