Revolutionizing Aromatic Ketone Production With Novel Radical Tandem Catalysis For Commercial Scale-Up
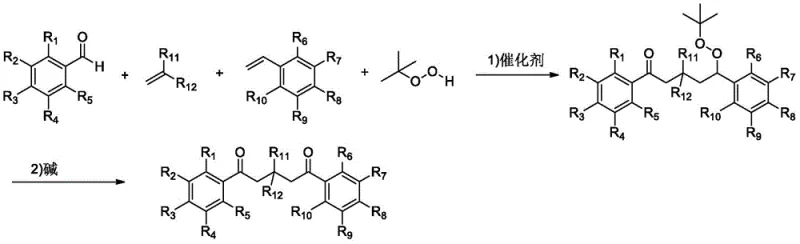
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing complex molecular architectures, particularly those containing carbonyl functionalities which serve as pivotal intermediates for further derivatization. Patent CN110256249B discloses a groundbreaking preparation method for aromatic ketone compounds substituted with different functional groups at the beta and delta positions. This technology represents a significant leap forward in synthetic organic chemistry, utilizing a transition metal-catalyzed free radical tandem reaction strategy. By employing readily available starting materials such as aromatic aldehydes, electron-deficient olefins, and styrene derivatives in the presence of tert-butyl hydroperoxide, this process efficiently generates peroxide intermediates which are subsequently converted into the target ketones via a base-mediated rearrangement. The ability to construct three new chemical bonds and introduce two distinct functional groups in a single operational sequence addresses critical bottlenecks in the synthesis of 1,5-dicarbonyl compounds, which are essential precursors for various biologically active heterocycles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of 1,5-dicarbonyl compounds and related aromatic ketones has relied heavily on Michael addition reactions. While historically significant, these conventional pathways often encounter substantial challenges when applied to complex substrate scopes. Specifically, the selectivity of Michael additions can be poor when dealing with substrates possessing other electron-withdrawing substituents, leading to mixtures of regioisomers and byproducts that complicate downstream purification. Furthermore, multi-step sequences are frequently required to install the necessary functional groups at specific positions, resulting in lower overall atom economy and increased waste generation. The harsh reaction conditions sometimes necessitated by these older methods can also limit the tolerance of sensitive functional groups, thereby restricting the chemical diversity accessible to process chemists. These limitations collectively drive up the cost of goods and extend the lead time for developing new active pharmaceutical ingredients or agrochemical candidates.
The Novel Approach
In stark contrast, the methodology described in patent CN110256249B offers a streamlined, one-pot solution that circumvents the drawbacks of stepwise synthesis. This novel approach leverages a radical cascade mechanism initiated by transition metals such as iron or nickel salts. The process begins with the generation of an acyl radical from an aromatic aldehyde using tert-butyl hydroperoxide as both a radical initiator and oxidant. This reactive species sequentially adds across an electron-deficient olefin and a styrene derivative, effectively stitching together three distinct molecular fragments with high precision. The subsequent in situ rearrangement of the peroxide intermediate under basic conditions delivers the desired beta,delta-substituted aromatic ketone directly. This strategy not only simplifies the operational workflow but also enhances the structural complexity achievable in a single transformation, providing a powerful tool for the rapid assembly of diverse chemical libraries for drug discovery programs.
Mechanistic Insights into FeCl3-Catalyzed Radical Tandem Reaction
The core of this innovative synthesis lies in its sophisticated radical mechanism, which is meticulously orchestrated by the transition metal catalyst. Initially, the transition metal, such as ferrous chloride (FeCl2), interacts with tert-butyl hydroperoxide (TBHP) to generate tert-butoxy radicals. These radicals abstract a hydrogen atom from the aromatic aldehyde substrate, producing a highly reactive acyl radical. This acyl radical then undergoes a conjugate addition to the electron-deficient olefin, such as methyl acrylate or acrylonitrile, forming a new carbon-carbon bond and a carbon-centered radical at the alpha-position relative to the electron-withdrawing group. This intermediate radical subsequently adds to the styrene derivative, extending the carbon chain and creating another radical center benzylic to the aromatic ring. Finally, this benzylic radical couples with another molecule of TBHP or a tert-butoxy radical to form the key peroxide intermediate. This cascade sequence is remarkable for its ability to control regioselectivity and construct a complex backbone from simple precursors without the need for protecting groups or harsh reagents.
Following the radical cascade, the reaction system undergoes a crucial transformation upon the addition of a base, such as 1,8-diazabicycloundec-7-ene (DBU) or sodium hydroxide. This step triggers a Kornblum-DeLaMare rearrangement, a well-known decomposition pathway for peroxides. Under basic conditions, the peroxide intermediate eliminates a molecule of tert-butyl alcohol, resulting in the formation of a carbonyl group at the delta position. This rearrangement is highly efficient and proceeds under mild thermal conditions, typically around 90°C, ensuring that sensitive functional groups installed during the radical phase remain intact. The mechanistic elegance of this two-stage process—radical construction followed by ionic rearrangement—ensures high purity of the final product by minimizing side reactions. The insensitivity of the reaction to air and moisture further underscores its robustness, making it an ideal candidate for industrial scale-up where strict anhydrous conditions can be costly and difficult to maintain.
How to Synthesize Beta,Delta-Substituted Aromatic Ketones Efficiently
The practical implementation of this synthesis route is designed for ease of execution in both laboratory and pilot plant settings. The protocol involves a sequential addition of reagents into a pressure-resistant reaction vessel, followed by controlled heating stages. The initial radical tandem reaction is conducted in a polar aprotic solvent like acetonitrile at elevated temperatures to ensure complete conversion of the starting materials into the peroxide intermediate. Once this stage is complete, the reaction mixture is cooled, and a stoichiometric amount of base is introduced to drive the rearrangement to completion. The detailed standardized synthesis steps for this procedure are outlined in the guide below, providing a clear roadmap for technical teams to replicate the high yields reported in the patent data.
- Mix aromatic aldehyde, electron-deficient olefin, styrene derivative, and tert-butyl hydroperoxide (TBHP) with a transition metal catalyst (e.g., FeCl2) in a solvent like acetonitrile.
- Heat the reaction mixture to 90°C for 12 hours under air atmosphere to facilitate the radical tandem addition and peroxide formation.
- Add a base (e.g., DBU) to the cooled reaction system and heat again at 90°C for 12 hours to induce Kornblum-DeLaMare rearrangement, yielding the final aromatic ketone.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented technology offers compelling advantages that directly address the pain points of procurement managers and supply chain directors. The primary benefit stems from the utilization of commodity chemicals as starting materials. Aromatic aldehydes, styrenes, and acrylic esters are produced on a massive global scale, ensuring a stable and continuous supply chain with minimal risk of raw material shortages. This abundance translates into significant cost stability and reduces the vulnerability of the manufacturing process to market fluctuations. Furthermore, the one-pot nature of the reaction eliminates the need for intermediate isolation and purification steps, which are traditionally resource-intensive and time-consuming. By consolidating multiple bond-forming events into a single reactor run, the process drastically reduces solvent consumption, energy usage, and labor costs associated with handling multiple batches.
- Cost Reduction in Manufacturing: The economic efficiency of this method is driven by the elimination of expensive transition metal catalysts often required in cross-coupling reactions, replacing them with abundant iron or nickel salts. Additionally, the avoidance of complex protection-deprotection sequences and the reduction in unit operations lead to substantial cost savings in the overall manufacturing budget. The high atom economy of the tandem reaction ensures that a greater proportion of the raw material mass is incorporated into the final product, minimizing waste disposal costs. These factors combine to create a highly competitive cost structure for the production of high-value aromatic ketone intermediates.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes significantly to supply chain reliability. Since the process is insensitive to air and moisture, it does not require specialized inert atmosphere equipment or rigorously dried solvents, simplifying the infrastructure requirements for production facilities. This flexibility allows for manufacturing in a wider range of facilities, reducing logistical bottlenecks. The short reaction times and straightforward workup procedures, typically involving vacuum concentration and column chromatography, enable faster turnaround times from order to delivery. This agility is crucial for meeting the dynamic demands of the pharmaceutical market, where speed to market can be a decisive competitive advantage.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram or ton quantities is facilitated by the simplicity of the reaction setup and the use of common solvents like acetonitrile and ethyl acetate. The generation of tert-butyl alcohol as the primary byproduct is environmentally favorable, as it is less toxic and easier to manage than heavy metal waste or halogenated byproducts common in other synthetic routes. The high selectivity of the reaction minimizes the formation of difficult-to-separate impurities, reducing the burden on wastewater treatment systems. This alignment with green chemistry principles not only ensures compliance with increasingly stringent environmental regulations but also enhances the sustainability profile of the final chemical products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this aromatic ketone synthesis technology. These answers are derived directly from the experimental data and specifications provided in the patent documentation, ensuring accuracy and relevance for potential partners and licensees. Understanding these details is essential for evaluating the feasibility of integrating this method into existing production workflows or new product development pipelines.
Q: What are the key advantages of this radical tandem reaction over traditional Michael addition?
A: Unlike traditional Michael additions which often suffer from poor selectivity with electron-withdrawing substrates, this method utilizes a radical tandem mechanism to construct three new chemical bonds in a single pot, offering higher yields and easier purification.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process uses cheap and easily available raw materials like styrene derivatives and aromatic aldehydes. The operation is simple, insensitive to air and moisture, and involves straightforward column chromatography for purification, making it highly scalable.
Q: What types of functional groups can be introduced using this method?
A: The method allows for the introduction of diverse functional groups at the beta and delta positions, including esters, nitriles, amides, and phosphonates, by varying the electron-deficient olefin substrate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aromatic Ketones Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the one described in patent CN110256249B for the production of high-purity aromatic ketones. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate such innovative laboratory protocols into robust commercial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop to plant floor is seamless and efficient. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of intermediate meets the exacting standards required by the global pharmaceutical industry.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. By partnering with us, you gain access to our deep reservoir of process knowledge and our dedication to quality and reliability. Please contact us today to request specific COA data and route feasibility assessments, and let us help you optimize your supply chain with superior aromatic ketone intermediates.
