Advanced Metalloporphyrin Catalysis for High-Purity Aromatic Ketone Manufacturing
The chemical industry is currently witnessing a paradigm shift in the synthesis of high-value oxygenated intermediates, driven by the urgent need for greener and more efficient processes. Patent CN111646906A introduces a groundbreaking methodology for the preparation of aromatic ketones through the catalytic oxidation of benzylic secondary C-H bonds in aromatic hydrocarbons. This technology leverages the unique electronic properties of metalloporphyrin complexes to achieve exceptional selectivity and conversion rates under remarkably mild conditions. Unlike traditional methods that rely on harsh environments, this approach utilizes molecular oxygen as the terminal oxidant in a solvent-free system, aligning perfectly with modern principles of atom economy and environmental sustainability. For R&D directors and procurement specialists in the pharmaceutical and agrochemical sectors, this patent represents a significant opportunity to optimize supply chains for critical intermediates such as acetophenones and tetralones, which serve as foundational building blocks for a vast array of bioactive molecules and functional materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of aromatic ketones has been dominated by liquid-phase oxidation processes utilizing transition metal salts, such as cobalt or manganese acetates, often dissolved in corrosive organic solvents like acetic acid. These conventional routes suffer from inherent thermodynamic and kinetic limitations that necessitate high reaction temperatures, frequently exceeding 150°C, to achieve acceptable conversion rates. Such aggressive conditions inevitably lead to poor selectivity profiles, where the desired ketone product is prone to further deep oxidation into carboxylic acids or complete combustion into carbon dioxide and water. Furthermore, the use of corrosive solvents imposes severe constraints on reactor material selection, requiring expensive Hastelloy or lined vessels, and generates substantial volumes of acidic wastewater that demand costly treatment protocols. The difficulty in separating the homogeneous metal salt catalyst from the product stream also results in heavy metal contamination risks, complicating the purification process for sensitive pharmaceutical applications and increasing the overall cost of goods sold.
The Novel Approach
In stark contrast, the novel methodology described in the patent employs biomimetic metalloporphyrin catalysts that mimic the activity of cytochrome P450 enzymes, enabling highly selective C-H bond activation at significantly lower temperatures ranging from 80°C to 150°C. This process operates under solvent-free conditions, where the aromatic hydrocarbon substrate itself acts as the reaction medium, thereby eliminating the need for solvent recovery systems and reducing the environmental footprint of the manufacturing process. The versatility of this catalytic system is demonstrated by its applicability to a wide range of substrates, including ethylbenzene derivatives, indanes, and tetralins, as illustrated by the general substrate structures below.
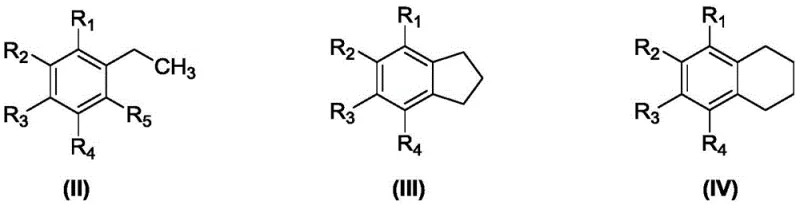
By tuning the peripheral substituents on the porphyrin ring and selecting appropriate central metal ions such as Cobalt (II) or Manganese (II), the catalyst can be optimized to suppress over-oxidation pathways, ensuring that the reaction stops selectively at the ketone stage with minimal formation of alcohol or acid byproducts. This precision in chemical transformation translates directly to simplified downstream processing and higher overall yields for the target aromatic ketone compounds.
Mechanistic Insights into Metalloporphyrin-Catalyzed C-H Oxidation
The core of this technological advancement lies in the unique electronic structure of the metalloporphyrin catalyst, depicted in Formula (I), which facilitates the homolytic or heterolytic cleavage of the dioxygen molecule to generate high-valent metal-oxo species. These reactive intermediates are capable of abstracting a hydrogen atom from the relatively inert benzylic secondary C-H bond, forming a carbon-centered radical that subsequently recombines with the hydroxyl group to form the ketone functionality. The steric bulk provided by the substituents (R1-R5) on the phenyl rings of the porphyrin macrocycle plays a critical role in protecting the active metal center from destructive dimerization or self-oxidation, thereby extending the catalyst lifetime and turnover number. This structural robustness allows the catalyst to function effectively at loadings as low as 1×10⁻⁴% (mol/mol), which is orders of magnitude lower than traditional metal salt catalysts.
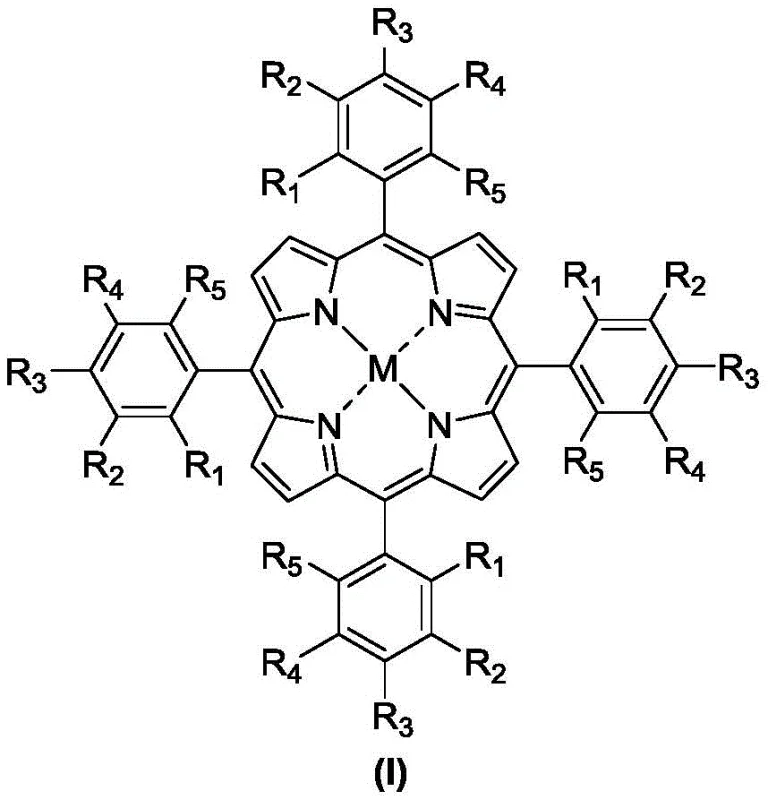
Furthermore, the mechanism inherently favors the formation of ketones over alcohols due to the rapid oxidation of the intermediate alcohol species under the reaction conditions, a phenomenon known as consecutive oxidation. To manage the inevitable formation of trace organic peroxides, which pose safety hazards and stability issues, the process incorporates a dedicated quenching step using triphenylphosphine (PPh3). This reductant efficiently converts unstable hydroperoxides into stable alcohols or ketones at room temperature, ensuring that the final product stream meets stringent safety specifications for storage and transport. This dual strategy of selective catalysis followed by chemical reduction of impurities provides a comprehensive solution for producing high-purity aromatic ketones suitable for sensitive downstream applications.
How to Synthesize Aromatic Ketones Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize the benefits of the metalloporphyrin system. The process begins by dispersing the selected metalloporphyrin catalyst directly into the aromatic hydrocarbon substrate, ensuring a homogeneous mixture without the addition of external solvents. The reaction vessel is then sealed and pressurized with oxygen or air to maintain a partial pressure between 0.20 MPa and 2.0 MPa, while the temperature is carefully regulated within the 80°C to 150°C window. Stirring rates are maintained between 600 rpm and 1200 rpm to ensure adequate mass transfer of the gaseous oxidant into the liquid phase. Following the reaction period of 3.0 to 24.0 hours, the mixture is cooled, and the specific post-treatment protocol involving triphenylphosphine is executed to guarantee product safety. For detailed standardized operating procedures and specific parameter optimization for different substrates, please refer to the guide below.
- Disperse metalloporphyrin catalyst (1×10⁻⁴% to 1% mol/mol) directly into the aromatic hydrocarbon substrate without additional solvents.
- Seal the reaction system, heat to 80-150°C with stirring, and introduce oxygen or air to a pressure of 0.20-2.0 MPa.
- Maintain reaction for 3.0-24.0 hours, then treat with triphenylphosphine to reduce peroxides before distillation and purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this metalloporphyrin-catalyzed oxidation process offers transformative advantages for supply chain resilience and cost management. The elimination of corrosive solvents and the drastic reduction in catalyst loading fundamentally alter the cost structure of aromatic ketone manufacturing. By removing the need for solvent recovery distillation columns and acidic wastewater treatment facilities, capital expenditure for new production lines can be significantly reduced, while existing infrastructure can be repurposed with minimal modification. The simplicity of the workup procedure, which primarily involves distillation and recrystallization after peroxide reduction, shortens the production cycle time and increases the throughput capacity of manufacturing plants. These operational efficiencies translate into a more competitive pricing structure for end-users without compromising on the quality or purity of the chemical intermediates supplied.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven primarily by the ultra-low catalyst dosage, which ranges from 1:1,000,000 to 1:1,000 mol/mol relative to the substrate. At such minute concentrations, the cost contribution of the expensive metalloporphyrin catalyst becomes negligible, and in many cases, the catalyst does not require recovery or recycling, effectively turning a homogeneous process into a pseudo-heterogeneous one in terms of cost accounting. Additionally, the solvent-free nature of the reaction eliminates the procurement costs associated with large volumes of organic solvents and the energy costs required for their evaporation and condensation. The reduction in energy consumption is further amplified by the lower operating temperatures, which decrease the steam or heating utility requirements compared to traditional high-temperature oxidation processes. Collectively, these factors result in a substantially lower variable cost per kilogram of product, enhancing margin potential for manufacturers and offering price stability for buyers.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of readily available and stable raw materials, specifically simple aromatic hydrocarbons and molecular oxygen or air, which are commoditized chemicals with robust global supply networks. The process does not rely on specialized or hazardous oxidants like chromic acid or permanganate, which are subject to strict regulatory controls and fluctuating availability. Furthermore, the high selectivity of the reaction minimizes the formation of difficult-to-separate byproducts, reducing the risk of production batches failing quality control specifications due to impurity profiles. This reliability ensures consistent on-time delivery performance, as production schedules are less likely to be disrupted by extended purification steps or batch rejections. The scalability of the process, demonstrated successfully from laboratory scale up to multi-liter reactors, confirms that supply volumes can be ramped up quickly to meet surging market demand without extensive process re-engineering.
- Scalability and Environmental Compliance: As regulatory pressures regarding industrial emissions and waste disposal intensify globally, this green chemistry approach positions manufacturers favorably for long-term compliance. The absence of halogenated or acidic solvents means that volatile organic compound (VOC) emissions are drastically curtailed, and the generation of hazardous liquid waste is virtually eliminated. The low peroxide content in the final product, achieved through the phosphine treatment, reduces safety risks during storage and transportation, lowering insurance premiums and logistics costs. The process is inherently scalable, as evidenced by the successful execution of experiments in 1.00L and 3.00L reactors with consistent conversion and selectivity metrics, indicating a smooth path to ton-scale commercial production. This environmental and safety profile not only meets current regulatory standards but also anticipates future tightening of environmental laws, safeguarding the supply chain against potential regulatory shutdowns or fines.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this metalloporphyrin catalytic system. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for evaluating the technology's fit within your specific manufacturing context. Understanding these nuances is crucial for R&D teams planning pilot studies and procurement officers negotiating supply agreements. The answers reflect the proven capabilities of the system regarding substrate scope, catalyst stability, and product purity.
Q: What are the advantages of using metalloporphyrins over traditional metal salt catalysts?
A: Metalloporphyrins offer significantly higher selectivity for aromatic ketones, preventing deep oxidation to carboxylic acids. They operate under milder temperatures (80-150°C) and do not require corrosive solvents like acetic acid, simplifying downstream processing and equipment maintenance.
Q: Is catalyst recovery required for this process?
A: Due to the extremely low catalyst loading (as low as 1:1,000,000 mol/mol), the metalloporphyrin often does not require complex separation or recovery steps, which drastically reduces production costs and waste generation compared to traditional homogeneous catalysis.
Q: How are hazardous peroxide byproducts managed in this synthesis?
A: The process includes a specific post-treatment step where triphenylphosphine (PPh3) is added to the reaction mixture at room temperature. This effectively reduces any generated organic peroxides to safe alcohols or ketones, ensuring high product safety and stability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aromatic Ketones Supplier
The technological potential of metalloporphyrin-catalyzed oxidation represents a significant leap forward in the synthesis of fine chemical intermediates, offering a pathway to cleaner, cheaper, and more efficient production. NINGBO INNO PHARMCHEM stands at the forefront of adopting such innovative methodologies, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of this solvent-free, high-pressure oxidation process, ensuring that every batch meets stringent purity specifications required by the global pharmaceutical and agrochemical industries. With our rigorous QC labs and commitment to continuous process improvement, we guarantee a supply of high-purity aromatic ketones that are free from heavy metal contaminants and hazardous peroxide residues, providing our partners with peace of mind and a competitive edge in their own downstream syntheses.
We invite you to collaborate with us to explore how this advanced catalytic technology can be integrated into your supply chain to drive value and efficiency. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. Please contact us today to request specific COA data for our aromatic ketone portfolio and to discuss route feasibility assessments for your custom synthesis projects. Let us partner with you to transform the way high-value intermediates are manufactured, ensuring a sustainable and profitable future for your chemical operations.
