Scalable Manufacturing of Combretastatin A-4 Precursors Using Optimized Perkin Condensation
Scalable Manufacturing of Combretastatin A-4 Precursors Using Optimized Perkin Condensation
The pharmaceutical industry's relentless pursuit of potent anti-tumor agents has placed significant focus on tubulin-binding compounds, specifically Combretastatin A-4 and its analogues. Patent CN1266104C introduces a groundbreaking synthetic methodology for producing (Z)-3'-hydroxy-3,4,4',5-tetramethoxy diphenyl ethylene, a critical intermediate in this class of vascular targeting agents. This intellectual property outlines a robust, four-step chemical sequence that fundamentally reimagines the construction of the diphenylethylene skeleton. By shifting away from costly and toxic reagents traditionally associated with Wittig or Suzuki couplings, this patent presents a pathway that is not only chemically elegant but also commercially viable for large-scale production. The innovation lies in the strategic use of inexpensive starting materials and the elimination of complex purification bottlenecks, directly addressing the pain points of modern API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of combretastatin analogues has been plagued by significant economic and operational hurdles. Traditional Wittig reaction pathways necessitate the use of triphenylphosphine, a reagent known for its toxicity and the generation of difficult-to-remove triphenylphosphine oxide byproducts. Furthermore, these reactions often demand cryogenic conditions, such as minus 78 degrees Celsius, requiring specialized equipment and driving up energy costs. Alternatively, Suzuki coupling methods, while effective, rely on precious metal catalysts like palladium and expensive boronic acid derivatives, which severely impact the cost of goods sold (COGS). Even earlier iterations of the Perkin reaction were flawed, relying on 3,4,5-trimethoxyphenylacetic acid as a starting block—a material that is notoriously difficult to source commercially and expensive to synthesize in-house due to the involvement of hazardous cyanide chemistry.
The Novel Approach
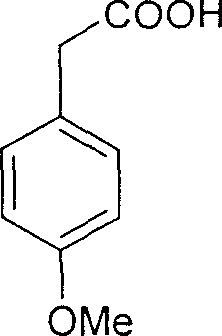
The methodology disclosed in CN1266104C circumvents these legacy issues by employing p-methoxyphenylacetic acid (homoanisic acid) as the foundational building block. This commodity chemical is abundant, stable, and significantly cheaper than its trimethoxy counterparts. The process ingeniously introduces the necessary functionality through a controlled bromination step followed by a modified Perkin condensation. This strategic pivot allows manufacturers to bypass the supply chain volatility associated with specialized aromatic acids. Moreover, the downstream processing is drastically simplified; the final product can be isolated through straightforward recrystallization rather than labor-intensive column chromatography, resulting in a cleaner process mass intensity (PMI) profile and reduced solvent waste.
Mechanistic Insights into Modified Perkin Condensation and Decarboxylation
The core of this synthetic strategy relies on a precise sequence of electrophilic substitution and condensation reactions. The process initiates with the regioselective bromination of homoanisic acid. Under controlled temperatures ranging from 0 to 30 degrees Celsius, bromine reacts with the electron-rich aromatic ring to yield 3-bromo-4-methoxyphenylacetic acid with high fidelity. This bromine atom serves a dual purpose: it acts as a blocking group to direct subsequent substitution and is later displaced to reveal the crucial phenolic hydroxyl group. Following this, the acid undergoes condensation with 3,4,5-trimethoxybenzaldehyde in the presence of acetic anhydride and triethylamine. Heating this mixture to between 90 and 150 degrees Celsius facilitates the formation of the carbon-carbon double bond, generating the (E)-acrylic acid intermediate.
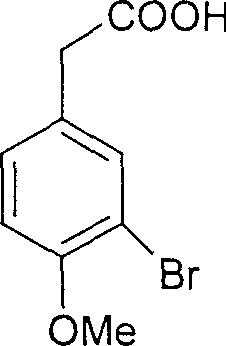
Subsequent transformation involves a sophisticated hydrolytic debromination. The bromo-intermediate is treated with sodium hydroxide solution in the presence of a copper catalyst, such as copper sulfate. This step effectively replaces the bromine atom with a hydroxyl group while maintaining the integrity of the sensitive alkene linkage. The final and perhaps most critical step is the decarboxylation. Conducted in quinoline with metallic copper at elevated temperatures of 180 to 220 degrees Celsius, this reaction removes the carboxylic acid moiety. Crucially, these specific thermal and catalytic conditions favor the thermodynamic equilibration towards the biologically active (Z)-isomer, overcoming the typical preference for the (E)-isomer seen in many stilbene syntheses. This mechanistic control ensures high stereochemical purity without the need for difficult photo-isomerization post-treatments.
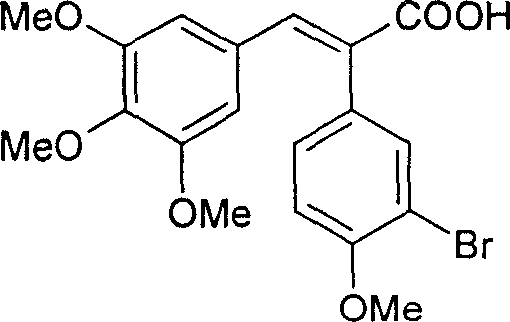
How to Synthesize (Z)-3'-hydroxy-3,4,4',5-tetramethoxy diphenyl ethylene Efficiently
Implementing this synthesis requires careful attention to reaction parameters, particularly temperature control during the bromination and decarboxylation phases to maximize yield and selectivity. The patent provides a clear roadmap for transitioning from gram-scale laboratory experiments to multi-kilogram production batches. The detailed standardized synthesis steps below outline the precise stoichiometry, solvent choices, and workup procedures necessary to replicate the high yields reported in the intellectual property. Adhering to these protocols ensures consistent quality and minimizes the formation of impurities that could complicate downstream crystallization.
- Brominate p-methoxyphenylacetic acid to obtain 3-bromo-4-methoxyphenylacetic acid.
- Perform Perkin condensation with 3,4,5-trimethoxy benzaldehyde to form the acrylic acid derivative.
- Execute phenolic hydroxyl substitution and debromination followed by decarboxylation to yield the final Z-isomer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift to this synthetic route represents a substantial opportunity for cost optimization and risk mitigation. The reliance on homoanisic acid eliminates the dependency on niche suppliers of trimethoxy-substituted phenylacetic acids, thereby stabilizing the raw material supply chain against market fluctuations. Furthermore, the avoidance of palladium catalysts removes the need for expensive metal scavenging steps and rigorous heavy metal testing, which are significant cost drivers in API manufacturing. The simplification of the purification process from column chromatography to recrystallization not only reduces solvent consumption but also shortens the overall production cycle time, enhancing facility throughput.
- Cost Reduction in Manufacturing: The replacement of expensive starting materials and catalysts with commodity chemicals leads to a drastic reduction in direct material costs. By eliminating the need for column chromatography, the process significantly lowers solvent usage and waste disposal expenses, contributing to a leaner manufacturing budget.
- Enhanced Supply Chain Reliability: Sourcing p-methoxyphenylacetic acid is far more reliable than procuring specialized boronic acids or phosphonium salts. This stability ensures continuous production schedules and reduces the risk of delays caused by raw material shortages, securing the supply of this critical oncology intermediate.
- Scalability and Environmental Compliance: The use of recyclable solvents like ethyl acetate and petroleum ether, combined with the absence of toxic phosphine waste, aligns with modern green chemistry principles. This environmental compatibility simplifies regulatory compliance and facilitates easier scale-up from pilot plants to commercial tonnage production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on yield expectations, purity profiles, and operational safety. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for commercial manufacturing.
Q: What is the primary cost advantage of this synthesis route?
A: The process utilizes p-methoxyphenylacetic acid (homoanisic acid), a cheap and readily available commodity chemical, replacing the expensive and difficult-to-source 3,4,5-trimethoxyphenylacetic acid required in traditional Perkin methods.
Q: How does this method improve purification efficiency?
A: Unlike prior art methods that often require complex column chromatography, this optimized route allows for purification via simple recrystallization using environmentally friendly solvents like ethyl acetate and petroleum ether.
Q: What is the stereochemical outcome of the decarboxylation step?
A: The decarboxylation step, conducted in the presence of quinoline and copper powder, selectively yields the biologically active (Z)-isomer with improved cis-trans selectivity compared to literature values.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (Z)-3'-hydroxy-3,4,4',5-tetramethoxy diphenyl ethylene Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes for high-value oncology intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN1266104C are fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of (Z)-3'-hydroxy-3,4,4',5-tetramethoxy diphenyl ethylene meets the exacting standards required for clinical and commercial applications.
We invite global partners to leverage our technical expertise to optimize their supply chains. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to support your next project milestone.
