Advanced Telmisartan Manufacturing: Overcoming Stability Barriers for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic pathways that balance high purity with economic viability, particularly for blockbuster antihypertensive agents like Telmisartan. Patent CN1344712A introduces a pivotal modification to the established synthesis of Telmisartan (III), addressing critical stability issues inherent in previous methodologies. This innovation replaces the traditionally used tert-butyl protecting group with methyl or ethyl esters, fundamentally altering the reaction landscape to favor stability and control. By shifting the protection strategy on the biphenyl carboxylic acid moiety, the process mitigates the risks associated with acid-labile groups, thereby enhancing the overall reliability of the manufacturing sequence. This technical evolution is not merely a chemical curiosity but a strategic imperative for any reliable telmisartan intermediate supplier aiming to secure long-term production contracts. The implications of this patent extend beyond the laboratory, offering a blueprint for cost reduction in API manufacturing through simplified raw material handling and improved yield consistency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
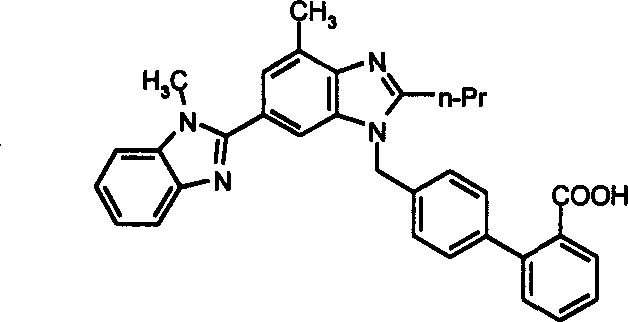
Historically, the synthesis of Telmisartan has relied heavily on the use of 4'-bromomethylbiphenyl-2-carboxylic acid tert-butyl ester as a key coupling partner. While conceptually sound, this reagent presents significant operational challenges during the nucleophilic substitution phase. The tert-butyl group is notoriously susceptible to cleavage under mildly acidic conditions, which can inadvertently occur during reaction workups or due to trace acidic impurities in solvents. This instability leads to premature deprotection, generating free carboxylic acid species that complicate the reaction matrix and foster the formation of difficult-to-remove impurities. Consequently, the reaction pathway becomes wayward and difficult to control, often resulting in suboptimal yields that erode profit margins. For a commercial scale-up of complex pharmaceutical intermediates, such unpredictability is unacceptable, as it necessitates extensive purification steps and increases the risk of batch failure, ultimately compromising the supply chain continuity for downstream drug product manufacturers.
The Novel Approach
In stark contrast, the methodology disclosed in CN1344712A utilizes 4'-bromomethylbiphenyl-2-carboxylate methyl or ethyl esters, which exhibit superior chemical stability compared to their tert-butyl counterparts. These alkyl esters are not only easier to prepare and handle but also remain inert under the reaction conditions required for the coupling with the benzimidazole intermediate (I). This stability ensures that the protecting group remains intact throughout the nucleophilic substitution, preventing the generation of acidic byproducts that could derail the synthesis. Furthermore, the subsequent deprotection step via hydrolysis is straightforward and efficient, allowing for the clean liberation of the final carboxylic acid functionality without the harsh conditions often required to remove bulky tert-butyl groups. This streamlined approach directly translates to high-purity telmisartan with reduced impurity profiles, satisfying the stringent quality requirements of global regulatory bodies while simplifying the overall process flow.
Mechanistic Insights into Nucleophilic Substitution and Ester Hydrolysis
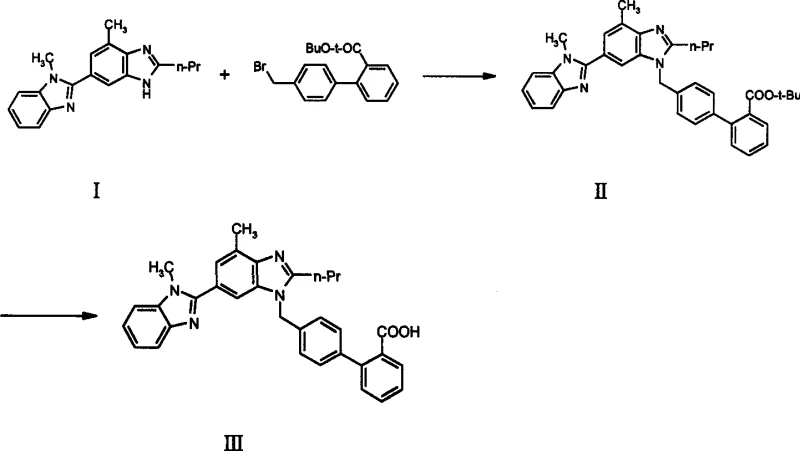
The core of this improved synthesis lies in the nucleophilic displacement of the bromide in the biphenyl derivative by the nitrogen of the benzimidazole ring. In the novel process, the use of methyl or ethyl esters allows for a wider selection of bases, ranging from mild carbonates like potassium carbonate to stronger alkoxides like sodium ethoxide, without risking ester hydrolysis or deprotection during the alkylation step. The reaction proceeds smoothly in polar aprotic solvents such as DMF or DMSO, where the nucleophile is sufficiently activated to attack the benzylic carbon. The absence of acid-sensitive groups means that the reaction environment can be optimized purely for kinetics and solubility, rather than being constrained by the need to preserve a fragile protecting group. This flexibility is crucial for reducing lead time for high-purity antihypertensive agents, as it allows process chemists to push reaction rates higher without fear of degradation, thereby shortening cycle times and increasing throughput capacity in multi-purpose reactors.
Following the coupling, the conversion of the intermediate ester (IV) to the final acid (III) is achieved through standard hydrolysis protocols that are highly scalable. Whether performed under acidic conditions using hydrochloric acid in acetic acid or under basic conditions with sodium hydroxethoxide in alcohol-water mixtures, the cleavage of the methyl or ethyl ester is rapid and quantitative. Unlike the tert-butyl group, which requires strong acids for removal and can generate isobutylene gas that complicates venting systems, the hydrolysis of simple alkyl esters generates volatile alcohols that are easily removed during solvent recovery. This mechanistic simplicity ensures that the final product precipitates cleanly upon acidification, minimizing the entrapment of solvent or salt impurities. The result is a crystalline product with excellent physical properties, facilitating filtration and drying operations which are often bottlenecks in large-scale API production.
How to Synthesize Telmisartan Efficiently
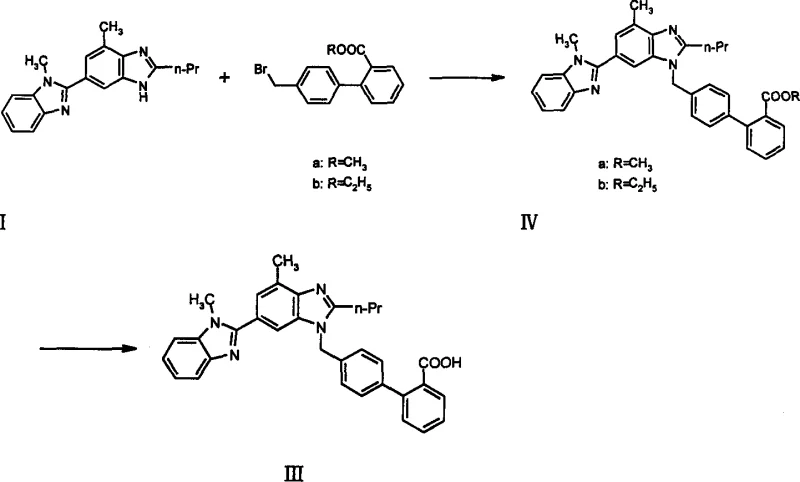
The implementation of this patented route involves a precise sequence of unit operations designed to maximize yield while maintaining safety and environmental compliance. The process begins with the careful selection of the base and solvent system to ensure complete conversion of the starting materials, followed by a controlled quench to isolate the intermediate ester. Detailed standard operating procedures regarding temperature ramps, addition rates, and crystallization parameters are essential to replicate the high purity described in the patent examples. For a comprehensive understanding of the specific stoichiometric ratios and thermal profiles required to achieve the reported 80%+ yields, operators must refer to the detailed experimental embodiments provided in the full technical documentation.
- Perform nucleophilic substitution between intermediate I and 4'-bromomethylbiphenyl-2-carboxylate ester using a base like K2CO3 in DMF at 20-100°C.
- Isolate the intermediate ester (IV) through extraction and washing protocols.
- Hydrolyze the ester intermediate under acidic or alkaline conditions to yield the final Telmisartan acid (III).
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the shift to methyl and ethyl esters represents a significant strategic advantage, primarily due to the ubiquitous availability and low cost of these raw materials compared to specialized tert-butyl derivatives. The supply chain for methanol and ethanol-based esters is mature and robust, insulating manufacturers from the volatility often seen in niche chemical markets. This accessibility ensures that production schedules are not disrupted by raw material shortages, a critical factor for maintaining supply chain reliability in the competitive generics market. Furthermore, the enhanced stability of these intermediates reduces the need for specialized storage conditions, such as strict temperature control or inert atmospheres, thereby lowering warehousing costs and simplifying logistics management for bulk chemical transport.
- Cost Reduction in Manufacturing: The elimination of unstable protecting groups fundamentally alters the cost structure of Telmisartan production by removing the need for expensive remediation steps. In conventional processes, the formation of impurities due to tert-butyl cleavage often necessitates additional chromatography or recrystallization cycles, which consume significant amounts of solvent and labor. By preventing these side reactions at the source, the novel process drastically reduces solvent consumption and waste disposal costs. Additionally, the higher yields obtained through better reaction control mean that less starting material is required per kilogram of final product, directly improving the material cost of goods sold (COGS) and enhancing overall project economics without compromising quality standards.
- Enhanced Supply Chain Reliability: The robustness of the methyl and ethyl ester intermediates translates directly into more predictable manufacturing timelines. Because the reaction is less sensitive to minor fluctuations in pH or moisture content, the risk of batch rejection is significantly minimized. This reliability allows supply chain managers to operate with leaner safety stocks, knowing that the production process is stable and reproducible. Moreover, the ease of preparing the starting esters means that vertical integration is a viable option; manufacturers can produce their own coupling partners in-house, further securing the supply chain against external market disruptions and ensuring a consistent flow of materials for continuous API synthesis.
- Scalability and Environmental Compliance: Scaling this process to multi-ton quantities is facilitated by the benign nature of the byproducts generated during hydrolysis. The release of simple alcohols like methanol or ethanol during deprotection is far easier to manage and recover than the gaseous isobutylene produced from tert-butyl groups, which requires specialized scrubbing systems. This simplifies the engineering requirements for large-scale reactors and aligns the process with modern green chemistry principles by reducing hazardous emissions. The ability to run the reaction at moderate temperatures (20-100°C) also reduces energy consumption for heating and cooling, contributing to a lower carbon footprint for the manufacturing facility and supporting corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this synthesis route, focusing on the practical aspects of reaction control and impurity management. Understanding these nuances is vital for process engineers tasked with transferring this technology from the laboratory to pilot and commercial scales. The answers provided are derived directly from the experimental data and technical assertions found within the patent specification, ensuring accuracy and relevance for technical decision-makers.
Q: Why is the methyl/ethyl ester route superior to the tert-butyl ester route for Telmisartan?
A: The tert-butyl ester used in conventional methods is unstable under acidic conditions, leading to premature deprotection and impurity formation. The methyl and ethyl esters proposed in CN1344712A are significantly more stable, ensuring better reaction control and higher purity.
Q: What are the typical reaction conditions for the key substitution step?
A: The nucleophilic substitution typically employs organic or mineral bases such as potassium carbonate or sodium ethoxide in polar aprotic solvents like DMF or DMSO, operating within a temperature range of 20°C to 100°C.
Q: How does this process impact commercial scalability?
A: By utilizing stable, easily prepared starting materials and robust reaction conditions that minimize side reactions, this process reduces batch-to-batch variability and simplifies downstream purification, making it highly suitable for large-scale industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Telmisartan Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to a more efficient synthesis route requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN1344712A are fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to monitor every critical quality attribute of the Telmisartan intermediate and API. Our commitment to quality assurance guarantees that every batch meets the exacting standards required by global regulatory agencies, providing our partners with the confidence needed to navigate complex approval processes.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this advanced synthesis technology for their Telmisartan projects. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this methyl/ethyl ester route for your specific volume requirements. We encourage you to contact us today to discuss your specific needs,索取 specific COA data, and review our comprehensive route feasibility assessments to ensure your supply chain is optimized for the future.
