Advanced Telmisartan Manufacturing: Overcoming Crystallization Barriers for Commercial Scale-up
Advanced Telmisartan Manufacturing: Overcoming Crystallization Barriers for Commercial Scale-up
The global demand for angiotensin-II-receptor antagonists continues to drive innovation in the synthesis of key antihypertensive agents like telmisartan. Patent CN100418953C introduces a transformative methodology that addresses critical bottlenecks in the industrial production of this active pharmaceutical ingredient. By shifting from traditional tert-butyl ester precursors to a robust nitrile-based coupling strategy, this technology offers a pathway to significantly enhanced process efficiency and product quality. The core innovation lies in the strategic selection of starting materials and reaction conditions that prevent the formation of problematic crystal morphologies often encountered in legacy processes. This report analyzes the technical merits of this approach, providing R&D and procurement leaders with actionable insights into optimizing their supply chains for high-purity cardiovascular therapeutics.
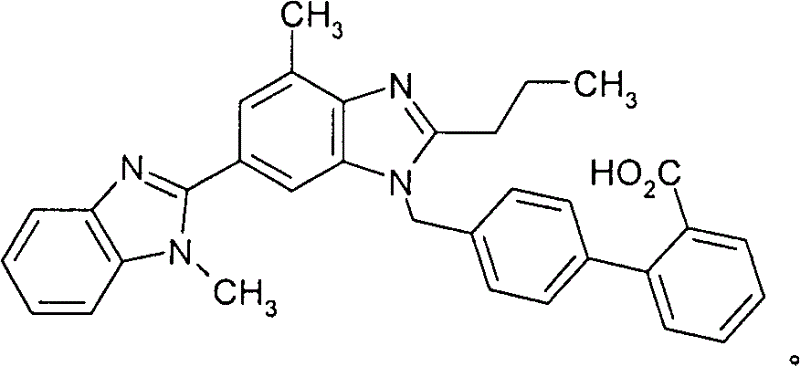
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of telmisartan has relied on the coupling of 2-n-propyl-4-methyl-6-(1′-methylbenzimidazol-2′-yl)-benzimidazole with 4′-bromomethyl-biphenyl-2-carboxylic acid tert-butyl ester. While chemically feasible, scaling this route presents severe engineering challenges that impact throughput and operational expenditure. A primary defect of this conventional pathway is the physical nature of the final product; the active substance tends to precipitate in the form of long needles. These needle-like crystals create a dense filter cake that resists efficient washing and isolation, leading to prolonged centrifugation cycles and extended drying times. Furthermore, the drying process often results in the formation of hard, large lumps that are difficult to mill, and the resulting dry powder exhibits significant static electricity, complicating downstream handling and packaging operations.
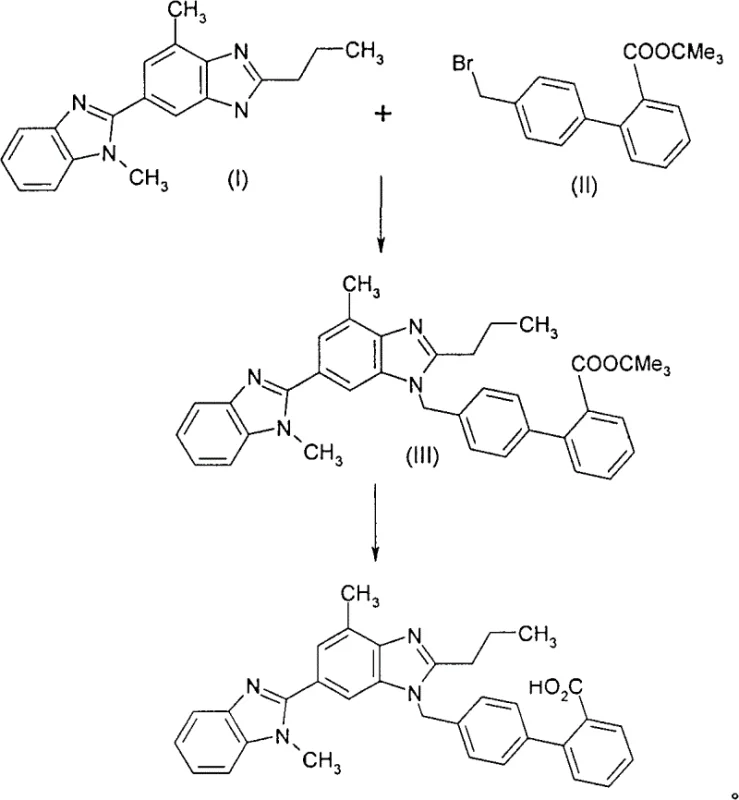
The Novel Approach
In stark contrast, the methodology disclosed in CN100418953C circumvents these physical limitations by employing a nitrile-functionalized coupling partner, specifically compounds of general formula (IV) where Z represents a leaving group such as bromine. This strategic modification allows for the formation of a stable nitrile intermediate, 2-cyano-4′-[2″-n-propyl-4″-methyl-6″-(1″′-methylbenzimidazol-2″′-yl)benzimidazol-1″-ylmethyl]biphenyl, which exhibits superior crystallization properties. Unlike the free acid or tert-butyl ester derivatives, this nitrile intermediate can be induced to precipitate rapidly and uniformly by treatment with solvents like tert-butyl methyl ether. This results in a free-flowing solid that is exceptionally easy to filter and wash, eliminating the need for the cumbersome recrystallization steps required by older methods to achieve acceptable purity levels. The subsequent hydrolysis of the nitrile function to the carboxylic acid is performed under controlled alkaline conditions, further ensuring a clean conversion to the final API.
Mechanistic Insights into Nucleophilic Substitution and Nitrile Hydrolysis
The success of this novel synthetic route hinges on the precise control of the nucleophilic substitution reaction in the first step. The reaction between the benzimidazole derivative and the bromomethyl-biphenyl nitrile is conducted in polar aprotic solvents such as dimethylacetamide (DMA) or dimethylformamide (DMF), often in the presence of strong bases like potassium hydroxide or potassium tert-butoxide. Maintaining the reaction temperature between 0°C and 30°C is critical to minimizing side reactions while ensuring complete conversion. The choice of base and solvent system facilitates the deprotonation of the benzimidazole nitrogen, generating a highly reactive nucleophile that attacks the benzylic carbon of the coupling partner. This step is remarkably efficient, typically achieving yields between 80-90% of theory with HPLC purity exceeding 99.5%, a testament to the chemoselectivity of the conditions employed.
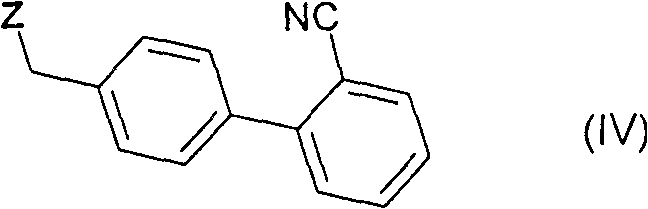
Following the coupling, the transformation of the nitrile group into the requisite carboxylic acid is achieved through alkaline hydrolysis. This step utilizes high-boiling solvent systems, preferably a mixture of ethylene glycol and water, heated to temperatures ranging from 140°C to 200°C. The use of ethylene glycol allows the reaction to proceed at elevated temperatures without excessive pressure, promoting rapid hydrolysis kinetics. Potassium hydroxide serves as the hydrolytic agent, converting the cyano group into a carboxylate salt. A distinct advantage of this mechanism is the ability to isolate the product directly as the hydrochloride salt by acidifying the reaction mixture with hydrochloric acid. This salt formation not only drives the precipitation of the product but also acts as a powerful purification step, as impurities remain soluble in the acidic mother liquor, thereby delivering the final API in a highly pure state suitable for pharmaceutical formulation.
How to Synthesize Telmisartan Efficiently
The implementation of this synthesis route requires careful attention to solvent removal and precipitation dynamics to maximize yield and purity. The process begins with the coupling reaction in DMA, followed by the distillation of the solvent under vacuum. The critical isolation step involves suspending the residue in tert-butyl methyl ether, which induces the crystallization of the nitrile intermediate. This solid is then subjected to hydrolysis in ethylene glycol/water with KOH, followed by acidification to precipitate the telmisartan hydrochloride. For detailed operational parameters, stoichiometry, and specific workup procedures validated by experimental data, please refer to the standardized synthesis guide below.
- Coupling Reaction: React 2-n-propyl-4-methyl-6-(1'-methylbenzimidazol-2'-yl)-benzimidazole with 4'-bromomethyl-2-cyanobiphenyl in DMA using KOH or t-BuOK at 0-30°C.
- Isolation of Nitrile Intermediate: Remove solvent and precipitate the crude nitrile product using tert-butyl methyl ether, yielding >99.5% purity material.
- Hydrolysis and Salt Formation: Hydrolyze the nitrile group in ethylene glycol/water with KOH at 155-185°C, then precipitate Telmisartan Hydrochloride using HCl.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process translates into tangible operational efficiencies and risk mitigation. The shift away from complex tert-butyl ester precursors to readily available nitrile building blocks simplifies the raw material sourcing landscape. Furthermore, the elimination of difficult filtration steps and the reduction in processing time directly correlate to increased reactor turnover rates and lower utility consumption per kilogram of produced API. The robustness of the crystallization profile ensures consistent batch-to-batch quality, reducing the likelihood of production delays caused by out-of-specification material. These factors collectively contribute to a more resilient and cost-effective manufacturing ecosystem for high-volume antihypertensive drugs.
- Cost Reduction in Manufacturing: The novel process eliminates the need for expensive and hazardous reagents like concentrated hydrobromic acid or trifluoroacetic acid used in traditional saponification steps. By utilizing common inorganic bases such as potassium hydroxide and standard organic solvents, the direct material costs are significantly lowered. Additionally, the high purity of the intermediate (>99.5%) reduces the burden on downstream purification, effectively cutting the number of unit operations required. This streamlining of the workflow leads to substantial savings in labor, energy, and waste disposal costs, enhancing the overall economic viability of large-scale telmisartan production.
- Enhanced Supply Chain Reliability: The starting material, 4′-bromomethyl-2-cyanobiphenyl, is a commodity chemical that can be produced in large quantities and is commercially available at competitive prices. This contrasts with specialized ester precursors that may have limited supplier bases. The improved physical properties of the intermediate, specifically its free-flowing crystal habit, ensure that manufacturing campaigns can proceed without the mechanical bottlenecks associated with needle-like solids. This reliability minimizes the risk of unplanned downtime and ensures a steady flow of finished goods to meet market demand, securing the supply chain against volatility.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies issues related to heat transfer and mass transfer, particularly during filtration and drying. The new method's use of easily filterable precipitates mitigates these scale-up risks, allowing for seamless transition from pilot plant to multi-ton commercial production. Moreover, the avoidance of halogenated acids and the use of recyclable high-boiling solvents like ethylene glycol align with modern green chemistry principles. This reduces the environmental footprint of the manufacturing process, facilitating compliance with increasingly stringent regulatory standards regarding solvent emissions and waste generation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced telmisartan synthesis technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on the practical benefits of adopting this route over conventional methods.
Q: Why does the conventional telmisartan synthesis face filtration challenges?
A: Conventional methods using tert-butyl ester precursors often result in products precipitating as long needles. This morphology causes slow filtration, difficult washing, and the formation of hard, static-prone lumps during drying, significantly hindering large-scale processing.
Q: How does the nitrile route improve product purity?
A: The novel process utilizes a nitrile intermediate that can be precipitated with high selectivity using ether solvents like tert-butyl methyl ether. This yields an intermediate with >99.5% purity (HPLC) before the final hydrolysis step, reducing the need for multiple recrystallizations.
Q: What are the advantages of isolating Telmisartan as a hydrochloride salt?
A: Isolating the product as the hydrochloride salt facilitates easier filtration and purification compared to the free acid form. The salt form avoids the polymorphic instability issues associated with the free acid and simplifies the final isolation workflow.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Telmisartan Supplier
At NINGBO INNO PHARMCHEM, we recognize that the theoretical advantages of a patent must be translated into practical manufacturing excellence. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this novel nitrile-based route are fully realized in your supply chain. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of telmisartan intermediate or API meets the highest international standards. We understand the critical nature of cardiovascular medications and are committed to delivering consistent quality and reliability.
We invite you to engage with our technical team to explore how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the potential economic impact of switching to this superior process. Contact our technical procurement team today to obtain specific COA data and comprehensive route feasibility assessments tailored to your production goals, ensuring a partnership built on transparency and scientific rigor.
