Advanced Photocatalytic Strategy for Scalable Production of 1-Sulfonylmethyl-3,4-Dihydronaphthalene Derivatives
The pharmaceutical and fine chemical industries are constantly seeking more efficient and sustainable pathways to construct complex heterocyclic scaffolds, particularly those containing sulfur motifs which are prevalent in bioactive molecules. A significant breakthrough in this domain is detailed in Chinese Patent CN110540516B, which discloses a novel preparation method for 1-sulfonylmethyl-substituted 3,4-dihydronaphthalene compounds. This technology leverages visible-light mediation to drive a challenging C-C sigma-bond sulfonylation and arylation cascade, transforming readily available vinylcyclopropane compounds and sulfonyl chlorides into valuable dihydronaphthalene derivatives. Unlike traditional thermal methods that often require aggressive reagents or extreme temperatures, this photocatalytic approach operates under remarkably mild conditions, utilizing inexpensive light sources to generate reactive radical intermediates. For R&D directors and process chemists, this represents a paradigm shift towards greener synthesis, offering a robust platform for accessing structurally diverse sulfur-containing building blocks that are critical for drug discovery and agrochemical development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of dihydronaphthalene frameworks bearing sulfonyl groups has relied heavily on classical electrophilic aromatic substitution or transition-metal catalyzed cross-coupling reactions, both of which present significant operational challenges. Traditional Friedel-Crafts sulfonylation often necessitates the use of strong Lewis acids or corrosive protic acids, leading to poor functional group compatibility and difficult waste disposal issues due to the generation of stoichiometric amounts of acidic byproducts. Furthermore, achieving regioselectivity in these thermal processes can be problematic, often resulting in complex mixtures of isomers that require tedious purification steps, thereby lowering the overall process efficiency. Additionally, many existing methods for introducing sulfonyl groups rely on pre-functionalized substrates or expensive sulfur transfer reagents, which drastically increases the raw material costs and limits the economic viability of scaling these reactions to multi-kilogram production levels. The reliance on high temperatures and harsh conditions also poses safety risks in a manufacturing environment, particularly when dealing with sensitive intermediates that may decompose under thermal stress.
The Novel Approach
In stark contrast, the methodology described in patent CN110540516B introduces a transformative visible-light mediated strategy that circumvents these traditional bottlenecks by utilizing the inherent strain energy of vinylcyclopropanes. This innovative route enables the direct coupling of vinylcyclopropanes with sulfonyl chlorides, which are cheap, abundant, and commercially available sulfur sources, to forge the target 1-sulfonylmethyl-3,4-dihydronaphthalene skeleton in a single pot. 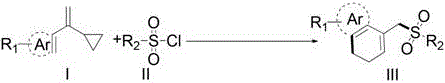 The reaction proceeds through a radical mechanism initiated by the photocatalytic reduction of the sulfonyl chloride, generating a sulfonyl radical that adds to the alkene moiety of the vinylcyclopropane. This triggers a rapid ring-opening event followed by an intramolecular radical cyclization onto the pendant aromatic ring, effectively constructing the fused bicyclic system with high precision. The use of visible light, typically from blue LEDs, allows the reaction to proceed at moderate temperatures (around 100°C in an oil bath for heating, but driven by photon energy), significantly reducing the thermal load on the system and enhancing safety profiles. This approach not only simplifies the synthetic sequence by merging multiple bond-forming events into one operation but also expands the chemical space accessible to chemists by tolerating a broad spectrum of functional groups that would otherwise be incompatible with harsh acidic conditions.
The reaction proceeds through a radical mechanism initiated by the photocatalytic reduction of the sulfonyl chloride, generating a sulfonyl radical that adds to the alkene moiety of the vinylcyclopropane. This triggers a rapid ring-opening event followed by an intramolecular radical cyclization onto the pendant aromatic ring, effectively constructing the fused bicyclic system with high precision. The use of visible light, typically from blue LEDs, allows the reaction to proceed at moderate temperatures (around 100°C in an oil bath for heating, but driven by photon energy), significantly reducing the thermal load on the system and enhancing safety profiles. This approach not only simplifies the synthetic sequence by merging multiple bond-forming events into one operation but also expands the chemical space accessible to chemists by tolerating a broad spectrum of functional groups that would otherwise be incompatible with harsh acidic conditions.
Mechanistic Insights into Visible-Light Mediated C-C Sigma-Bond Activation
The core of this technological advancement lies in the sophisticated interplay between the photocatalyst and the substrates to achieve C-C sigma-bond cleavage and functionalization. Upon irradiation with visible light, the photocatalyst, such as Ru(bpy)3Cl2, enters an excited state capable of engaging in single-electron transfer (SET) processes. In this specific transformation, the excited catalyst likely facilitates the reduction of the sulfonyl chloride (R-SO2-Cl), cleaving the S-Cl bond to release a chloride anion and generate a highly reactive sulfonyl radical (R-SO2•). This radical species then adds regioselectively to the terminal carbon of the vinyl group in the cyclopropane substrate, forming a new C-S bond and a transient carbon-centered radical adjacent to the cyclopropane ring. The relief of ring strain in the cyclopropane moiety drives the subsequent homolytic cleavage of the distal C-C bond, a process known as radical-induced ring opening, which generates a stabilized benzylic or homoallylic radical intermediate. This newly formed radical is positioned perfectly for an intramolecular attack on the ortho-position of the tethered aromatic ring, closing the second ring to form the dihydronaphthalene core. Finally, rearomatization or hydrogen atom transfer restores the neutral product, completing the catalytic cycle.  Understanding this mechanistic pathway is crucial for impurity control, as it highlights the importance of maintaining an inert atmosphere to prevent radical quenching by oxygen, which could lead to oxidation byproducts. Furthermore, the choice of base, such as 2,6-lutidine, plays a vital role in scavenging the HCl generated during the reaction, preventing acid-catalyzed side reactions or catalyst decomposition, thus ensuring high purity of the final API intermediate.
Understanding this mechanistic pathway is crucial for impurity control, as it highlights the importance of maintaining an inert atmosphere to prevent radical quenching by oxygen, which could lead to oxidation byproducts. Furthermore, the choice of base, such as 2,6-lutidine, plays a vital role in scavenging the HCl generated during the reaction, preventing acid-catalyzed side reactions or catalyst decomposition, thus ensuring high purity of the final API intermediate.
How to Synthesize 1-Sulfonylmethyl-3,4-Dihydronaphthalene Efficiently
To implement this synthesis effectively in a laboratory or pilot plant setting, operators must adhere to strict protocols regarding light intensity and atmospheric control to maximize yield and reproducibility. The patent outlines a standardized procedure where the vinylcyclopropane and sulfonyl chloride are combined with the photocatalyst and base in a polar aprotic solvent like 1,4-dioxane, which solubilizes the reagents effectively while remaining stable under the reaction conditions. The detailed standardized synthesis steps are provided below to ensure consistent quality and performance across different batches.
- Charge a Schlenk tube with vinylcyclopropane substrate, sulfonyl chloride, Ru(bpy)3Cl2 photocatalyst, and 2,6-lutidine base in 1,4-dioxane solvent.
- Purge the reactor with argon to establish an inert atmosphere and seal the system to prevent oxygen quenching of the excited state catalyst.
- Illuminate the reaction mixture with a 5W blue LED lamp while heating to 100°C for 24 hours, followed by standard workup and silica gel purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this visible-light mediated synthesis offers compelling economic and logistical benefits that directly impact the bottom line and operational resilience. The shift from expensive, specialized sulfur reagents to commodity sulfonyl chlorides represents a fundamental change in the cost structure of producing these intermediates, as sulfonyl chlorides are produced on a massive industrial scale for various applications, ensuring a stable and competitive supply market. Moreover, the elimination of harsh acidic reagents and the reduction in reaction severity simplify the downstream processing requirements, potentially reducing the consumption of neutralizing agents and the volume of hazardous waste generated, which translates to lower disposal costs and a smaller environmental footprint. The robustness of the reaction across a wide range of substrates means that a single manufacturing line can be utilized to produce a diverse library of analogues without extensive retooling or process re-validation, enhancing asset utilization and flexibility in responding to changing market demands.
- Cost Reduction in Manufacturing: The economic viability of this process is significantly enhanced by the use of earth-abundant or recyclable photocatalysts at low loadings, eliminating the need for costly precious metal catalysts like palladium or rhodium that are common in traditional cross-coupling strategies. By replacing expensive transition metals with organic dyes or ruthenium complexes that can be recovered or used in minute quantities, the direct material cost per kilogram of product is drastically lowered. Additionally, the mild reaction conditions reduce energy consumption associated with heating and cooling cycles, contributing to further operational savings over the lifecycle of the product. The high atom economy of the reaction, where most atoms from the starting materials are incorporated into the final product, minimizes raw material waste and maximizes the value derived from every kilogram of input.
- Enhanced Supply Chain Reliability: Relying on widely available starting materials such as vinylcyclopropanes and sulfonyl chlorides mitigates the risk of supply chain disruptions often associated with niche or custom-synthesized reagents. These precursors are manufactured by numerous global suppliers, creating a competitive marketplace that prevents vendor lock-in and ensures continuity of supply even during periods of high demand or geopolitical instability. The simplicity of the reaction setup, requiring only standard glassware or stainless steel reactors equipped with LED lighting arrays, means that production can be easily scaled or transferred between different manufacturing sites without the need for specialized high-pressure or cryogenic equipment. This flexibility allows for a more agile supply chain capable of rapidly ramping up production volumes to meet urgent client requirements or clinical trial timelines.
- Scalability and Environmental Compliance: From a regulatory and sustainability perspective, this photochemical method aligns perfectly with modern green chemistry principles, facilitating easier compliance with increasingly stringent environmental regulations. The absence of heavy metal residues in the final product simplifies the purification process and reduces the burden on quality control laboratories to perform extensive metal scavenging and testing, accelerating the release of batches for clinical or commercial use. The ability to run the reaction in safer solvents like 1,4-dioxane or potentially greener alternatives, combined with the lack of toxic gas evolution, creates a safer working environment for plant personnel and reduces the complexity of废气 treatment systems. As the industry moves towards more sustainable manufacturing practices, adopting such photochemical technologies positions companies as leaders in eco-friendly production, enhancing their brand reputation and appeal to environmentally conscious partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic technology, drawing directly from the experimental data and scope defined in the patent literature. These insights are intended to clarify the practical aspects of the synthesis for potential partners evaluating this route for their own supply chains.
Q: What are the primary advantages of this visible-light method over traditional thermal cyclization?
A: This method utilizes mild visible-light irradiation to activate stable C-C sigma bonds in vinylcyclopropanes, avoiding the harsh acidic or high-temperature conditions often required for traditional Friedel-Crafts type cyclizations, thereby improving functional group tolerance.
Q: Is the photocatalyst system cost-effective for large-scale manufacturing?
A: Yes, the process employs robust photocatalysts like Ru(bpy)3Cl2 at low loadings (typically 5 mol%), and the use of inexpensive sulfonyl chlorides as sulfur sources significantly reduces raw material costs compared to specialized sulfone reagents.
Q: What is the substrate scope for the sulfonyl chloride component?
A: The reaction demonstrates excellent adaptability, tolerating a wide range of substituents on the sulfonyl chloride including electron-withdrawing groups like nitro and cyano, as well as electron-donating alkyl and alkoxy groups, allowing for diverse library synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Sulfonylmethyl-3,4-Dihydronaphthalene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of visible-light mediated synthesis in accelerating the development of next-generation pharmaceutical intermediates. Our team of expert process chemists has extensively evaluated the protocol described in CN110540516B and possesses the technical expertise to optimize these photochemical reactions for industrial application. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with advanced photoreactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 1-sulfonylmethyl-3,4-dihydronaphthalene delivered meets the highest standards of quality required for GMP environments.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating exactly how this route can improve your margins. We encourage you to contact us today to discuss your requirements, obtain specific COA data for our catalog compounds, and receive comprehensive route feasibility assessments that will empower your R&D and supply chain strategies.
