Scalable Cu(I)-Catalyzed Synthesis of 2,3-Disubstituted Indoles for Commercial Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust, cost-effective methodologies for constructing complex heterocyclic scaffolds, particularly the ubiquitous indole nucleus found in countless bioactive molecules. A significant breakthrough in this domain is detailed in patent CN109851548B, which discloses a novel preparation method for 2,3-disubstituted indole compounds. This technology leverages a monovalent copper [Cu(I)] catalytic system to drive a tandem cyclization reaction between ortho-propargyl alcohol substituted anilines and dicarbonyl compounds. Unlike traditional methods that rely on scarce and expensive noble metals, this approach utilizes abundant copper salts to achieve high conversion rates under remarkably mild conditions. For R&D directors and process chemists, this represents a pivotal shift towards more sustainable and economically viable synthetic routes, offering a pathway to high-purity intermediates with reduced environmental impact and simplified downstream processing requirements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,3-disubstituted indoles has heavily relied on transition metal catalysis involving gold, silver, or platinum complexes. While these noble metal catalysts are effective, they present severe drawbacks for commercial manufacturing, primarily due to their exorbitant cost and the stringent regulatory limits on residual metal content in pharmaceutical ingredients. The removal of trace gold or platinum often necessitates additional purification steps, such as specialized scavenging resins or repeated recrystallizations, which drastically increase production time and waste generation. Furthermore, many conventional protocols require harsh reaction conditions, including high temperatures or strong acids, which can compromise the integrity of sensitive functional groups on the substrate, leading to lower overall yields and complex impurity profiles that are difficult to characterize and control during scale-up.
The Novel Approach
The methodology described in CN109851548B fundamentally addresses these bottlenecks by substituting precious metals with inexpensive monovalent copper catalysts. This switch not only lowers the direct material cost but also simplifies the workup procedure, as copper residues are generally easier to manage and remove to acceptable levels compared to noble metals. The reaction proceeds efficiently in common organic solvents like 1,2-dichloroethane at moderate temperatures ranging from 40°C to 80°C, eliminating the need for energy-intensive heating or cryogenic cooling. By employing a tandem cyclization strategy, the process constructs the indole core and installs the 2,3-substituents in a single operational step, thereby reducing the number of unit operations, minimizing solvent consumption, and significantly enhancing the overall atom economy of the synthesis.
Mechanistic Insights into Cu(I)-Catalyzed Tandem Cyclization
The core of this innovation lies in the ability of the Cu(I) species to activate the alkyne moiety of the ortho-propargyl alcohol substituted aniline, facilitating a nucleophilic attack by the adjacent amine nitrogen. This initial cyclization event generates an indole intermediate, which subsequently undergoes a Friedel-Crafts-type alkylation or addition with the dicarbonyl compound. The copper catalyst likely coordinates with the carbonyl oxygen or the enol form of the dicarbonyl substrate, increasing its electrophilicity and directing the regioselective attachment at the C3 position of the newly formed indole ring. This dual activation mode ensures that the reaction cascade proceeds smoothly without the accumulation of unstable intermediates, resulting in a clean transformation with minimal side reactions. The tolerance of the catalyst system to various electronic environments on both the aniline and the dicarbonyl components underscores its versatility for generating diverse chemical libraries.
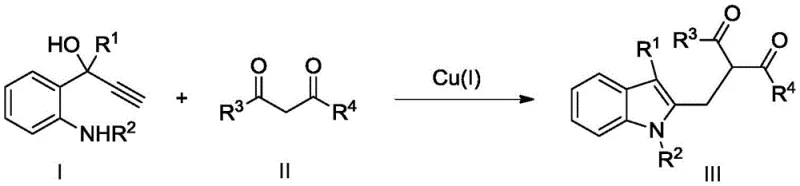
From an impurity control perspective, the mild nature of the copper catalysis is advantageous because it suppresses decomposition pathways often triggered by harsher Lewis acids or oxidants. The reaction generates very few by-products, as evidenced by the high isolated yields reported in the patent examples, which simplifies the chromatographic purification process. For quality control teams, this means a cleaner crude profile and a more consistent final product specification. The mechanism avoids the formation of polymeric tars or over-alkylated species that frequently plague indole functionalization reactions. Consequently, the process offers a predictable and reproducible route to 2,3-disubstituted indoles, ensuring that the critical quality attributes (CQAs) of the intermediate remain within tight specifications batch after batch, which is essential for regulatory compliance in API manufacturing.
How to Synthesize 2,3-Disubstituted Indoles Efficiently
To implement this synthesis effectively, one must carefully control the stoichiometry and reaction environment as outlined in the patent disclosure. The process typically involves dissolving the ortho-propargyl alcohol substituted aniline and the dicarbonyl compound in a solvent such as 1,2-dichloroethane, followed by the addition of a catalytic amount of a Cu(I) salt like Cu(CH3CN)4PF6. The mixture is then heated to a moderate temperature, typically around 60°C, and stirred for a period of 4 to 24 hours depending on the specific substrate reactivity. Detailed standardized synthetic steps, including precise molar ratios, solvent volumes, and workup procedures, are provided in the technical guide below to ensure reproducibility and safety during laboratory or pilot plant execution.
- Dissolve the ortho-propargyl alcohol substituted aniline compound and the dicarbonyl compound in an organic solvent such as 1,2-dichloroethane.
- Add a monovalent copper catalyst (e.g., CuI, CuCl, or Cu(CH3CN)4PF6) to the reaction mixture under air atmosphere.
- Stir the reaction mixture at a temperature between 40-80°C for 4-24 hours, then purify the resulting 2,3-disubstituted indole via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this copper-catalyzed route offers tangible strategic benefits beyond mere technical feasibility. The primary advantage is the drastic reduction in raw material costs associated with replacing noble metal catalysts with commodity copper salts, which are available in bulk quantities at a fraction of the price. This substitution directly impacts the cost of goods sold (COGS), allowing for more competitive pricing of the final pharmaceutical intermediates. Additionally, the use of common solvents and the avoidance of specialized reagents reduce supply chain risks, as these materials are widely sourced from multiple vendors, ensuring continuity of supply even during market fluctuations or geopolitical disruptions.
- Cost Reduction in Manufacturing: The elimination of expensive gold or platinum catalysts removes a significant cost driver from the bill of materials. Furthermore, the simplified purification process, necessitated by the cleaner reaction profile and easier removal of copper residues, reduces the consumption of silica gel, solvents, and labor hours required for chromatography. This holistic reduction in processing complexity translates to substantial cost savings per kilogram of product, making the manufacturing process leaner and more profitable without compromising on quality standards.
- Enhanced Supply Chain Reliability: Relying on copper catalysts and standard dicarbonyl building blocks mitigates the risk of supply shortages often associated with rare earth or noble metals. The robustness of the reaction conditions, which tolerate air and moisture better than many sensitive organometallic processes, allows for more flexible manufacturing scheduling and reduces the likelihood of batch failures due to minor environmental deviations. This reliability ensures consistent delivery timelines to downstream API manufacturers, strengthening the overall resilience of the pharmaceutical supply network.
- Scalability and Environmental Compliance: The mild reaction temperatures (40-80°C) and the use of established solvents make this process inherently scalable from gram to multi-ton quantities without requiring exotic reactor materials or extreme pressure ratings. From an environmental standpoint, the high atom economy and low by-product generation align with green chemistry principles, reducing the volume of hazardous waste requiring disposal. This facilitates easier compliance with increasingly stringent environmental regulations and supports corporate sustainability goals, enhancing the marketability of the produced intermediates to eco-conscious global partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this copper-catalyzed indole synthesis. These answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the method's capabilities and limitations for potential adopters and stakeholders evaluating the technology for their own production pipelines.
Q: What are the advantages of using Cu(I) catalysts over noble metals for indole synthesis?
A: Using Cu(I) catalysts significantly reduces raw material costs compared to gold, silver, or platinum. Furthermore, it eliminates the need for expensive and complex heavy metal removal processes required for pharmaceutical grade products, streamlining the purification workflow.
Q: What is the typical yield range for this Cu-catalyzed cyclization method?
A: According to patent CN109851548B, the reaction consistently achieves yields not lower than 62%, with specific optimized examples reaching up to 78%, demonstrating high efficiency and robustness across various substrates.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method operates under mild conditions (40-80°C) and uses readily available solvents and catalysts. The absence of sensitive noble metals and the generation of minimal by-products make it highly suitable for commercial scale-up and green manufacturing standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Disubstituted Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the modern pharmaceutical landscape. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory methodologies like the Cu(I)-catalyzed indole synthesis are successfully translated into robust industrial processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2,3-disubstituted indole meets the highest international standards, providing our partners with the confidence needed to advance their drug development programs without delay.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how switching to this copper-catalyzed method can optimize your budget. Please contact us today to request specific COA data for our available indole derivatives and to discuss route feasibility assessments for your custom synthesis projects, ensuring a seamless path from concept to commercial reality.
