Advanced Catalytic Routes for High-Purity 2-Imidazolidinone and Dialkoxy Derivatives Manufacturing
Introduction to Next-Generation Imidazolidinone Synthesis
The chemical landscape for producing heterocyclic intermediates is undergoing a significant transformation driven by the need for greener, more efficient, and scalable processes. A pivotal development in this sector is detailed in patent CN101395140B, which outlines a robust methodology for preparing 2-imidazolidinone compounds and their synthetic precursors, the 4,5-dialkoxy-2-imidazolidinone compounds. This technology addresses long-standing challenges in the industry, specifically the reliance on hazardous reagents like phosgene and dimethyl sulfate, which have traditionally plagued the synthesis of these valuable scaffolds. By shifting towards a catalytic hydrogenation approach coupled with solid acid catalysis, this innovation offers a pathway to high-purity products with substantially improved safety profiles and operational simplicity. For R&D directors and process chemists, understanding the nuances of this dual-catalyst system is critical for optimizing impurity profiles and ensuring regulatory compliance in pharmaceutical and agrochemical applications.
The core of this invention lies in a two-stage sequence that maximizes atom economy while minimizing waste generation. The first stage involves the condensation of a dicarbonyl compound, a urea derivative, and an alcohol in the presence of a solid acid catalyst to generate the dialkoxy intermediate. This is followed by a second stage where the intermediate undergoes hydrogenation over a metal catalyst to yield the final 2-imidazolidinone structure. This strategic decoupling of the cyclization and reduction steps allows for precise control over reaction parameters, ensuring consistent quality across batches. As global supply chains demand more reliable pharmaceutical intermediate suppliers, adopting such well-defined, patent-protected methodologies becomes a strategic imperative for maintaining competitive advantage and supply continuity.
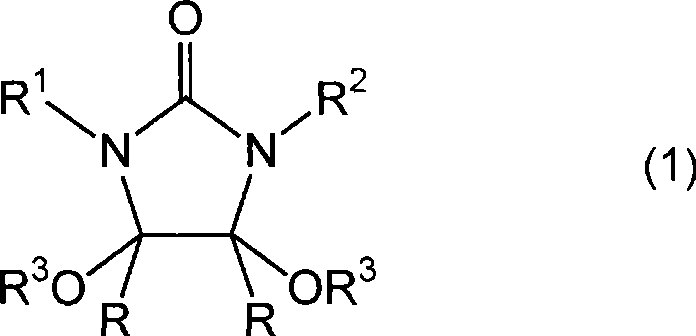
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of 2-imidazolidinone compounds has been fraught with significant technical and safety hurdles. Traditional routes often necessitate the use of phosgene, a highly toxic gas that requires specialized containment infrastructure and rigorous safety protocols, thereby inflating capital expenditure and operational risk. Alternative methods utilizing dimethyl sulfate introduce severe toxicity concerns related to alkylation agents, posing health risks to personnel and complicating waste disposal. Furthermore, earlier attempts to synthesize the requisite 4,5-dialkoxy-2-imidazolidinone precursors relied heavily on liquid mineral acids like concentrated sulfuric acid. While effective in driving the reaction, liquid acids create a massive downstream burden; they require energy-intensive neutralization steps, generate large volumes of saline wastewater, and often lead to corrosion issues in reactor vessels. These factors collectively render conventional methods economically inefficient and environmentally unsustainable for modern large-scale manufacturing.
The Novel Approach
In stark contrast, the novel approach described in the patent data leverages the power of heterogeneous catalysis to circumvent these legacy issues. By employing solid acid catalysts, such as sulfonic acid type cation exchange resins (e.g., Amberlite, Dowex, Nafion), the process eliminates the need for liquid acid neutralization entirely. The solid catalyst can be easily separated from the reaction mixture via simple filtration, allowing for potential regeneration and reuse, which drastically reduces raw material consumption. Following this, the conversion to the final product is achieved through catalytic hydrogenation using readily available metals like palladium, platinum, or nickel. This hydrogenation step is clean, producing only alcohol byproducts that can often be recycled, and operates under manageable temperature and pressure conditions. This shift from stoichiometric hazardous reagents to catalytic systems represents a paradigm shift in cost reduction in pharmaceutical intermediate manufacturing, aligning perfectly with green chemistry principles.
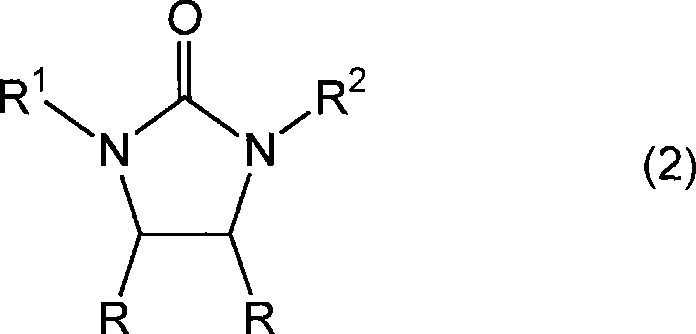
Mechanistic Insights into Solid Acid Catalysis and Hydrogenation
The mechanistic elegance of this process begins with the formation of the 4,5-dialkoxy-2-imidazolidinone ring system. In the presence of a solid acid catalyst possessing Brønsted acid sites, the carbonyl groups of the dicarbonyl compound (such as glyoxal) are activated towards nucleophilic attack by the urea nitrogen atoms. Simultaneously, the alcohol acts as both a solvent and a reactant, facilitating the formation of the acetal functionality at the 4 and 5 positions of the imidazolidinone ring. The solid surface of the catalyst provides a confined environment that enhances local concentration effects, promoting cyclization while suppressing polymerization side reactions that are common in homogeneous acid media. This controlled environment is crucial for maintaining high selectivity, ensuring that the resulting intermediate has a clean impurity profile before it even enters the reduction stage.
The subsequent hydrogenation mechanism involves the activation of molecular hydrogen on the surface of the metal catalyst particles. The 4,5-dialkoxy-2-imidazolidinone adsorbs onto the metal surface, where the carbon-oxygen bonds of the alkoxy groups are cleaved and replaced by hydrogen atoms. This reductive deprotection effectively removes the alkoxy groups, restoring the methylene bridges characteristic of the 2-imidazolidinone core. The choice of metal catalyst—whether palladium on carbon, platinum oxide, or Raney nickel—allows for fine-tuning of the reaction kinetics. For instance, palladium-based catalysts often exhibit superior activity at lower temperatures, while nickel-based systems offer a cost-effective alternative for less sensitive substrates. Understanding these mechanistic details enables process engineers to optimize parameters such as hydrogen pressure (typically 0.1 to 10 MPa) and temperature (0 to 200°C) to maximize yield and minimize the formation of over-reduced byproducts.
How to Synthesize 2-Imidazolidinone Efficiently
Implementing this synthesis route requires careful attention to catalyst loading and reaction conditions to ensure reproducibility on a commercial scale. The process begins by mixing the dicarbonyl compound, urea derivative, and alcohol with the solid acid catalyst, typically at temperatures ranging from 20°C to 200°C. Once the 4,5-dialkoxy intermediate is formed, it can be isolated or transferred directly to the hydrogenation vessel. For the reduction step, the intermediate is combined with the metal catalyst in a suitable solvent, such as an alcohol or water, and pressurized with hydrogen. Detailed standardized operating procedures are essential to manage the exothermic nature of hydrogenation and to ensure safe handling of pyrophoric catalysts.
- React a dicarbonyl compound, a urea compound, and an alcohol in the presence of a solid acid catalyst (e.g., sulfonic acid resin) to form 4,5-dialkoxy-2-imidazolidinone.
- Isolate the 4,5-dialkoxy-2-imidazolidinone intermediate or proceed directly to the next step without purification.
- Subject the intermediate to hydrogenation using a metal catalyst (Pd, Pt, or Ni) under pressure to yield the final 2-imidazolidinone compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this catalytic process offers tangible benefits that extend beyond mere chemical efficiency. The elimination of toxic reagents like phosgene removes a major bottleneck in raw material sourcing, as suppliers of such hazardous chemicals are becoming increasingly scarce due to regulatory pressures. Instead, the process relies on commodity chemicals like glyoxal, ureas, and alcohols, which are widely available from multiple global sources, thereby enhancing supply chain resilience and reducing the risk of disruption. Furthermore, the use of solid acid catalysts simplifies the purification workflow, removing the need for extensive washing and neutralization steps that consume time and utilities. This streamlined workflow translates directly into shorter production cycles and improved throughput capabilities.
- Cost Reduction in Manufacturing: The economic impact of switching to this method is profound, primarily driven by the reduction in waste treatment costs and the elimination of expensive stoichiometric reagents. By replacing liquid sulfuric acid with reusable solid acid resins, the facility avoids the recurring costs associated with acid neutralization and saline waste disposal. Additionally, the metal catalysts used in the hydrogenation step, particularly when supported on carbon, can often be recovered and refined, further lowering the cost per kilogram of the final product. The ability to run the reaction without isolating the intermediate in certain configurations also saves on solvent usage and drying energy, contributing to substantial cost savings in API manufacturing without compromising on quality.
- Enhanced Supply Chain Reliability: Dependence on a single source for hazardous reagents is a significant vulnerability in any supply chain. This new methodology diversifies the raw material base, allowing procurement teams to source dicarbonyl compounds and ureas from a broader network of certified vendors. The robustness of the solid acid catalysts also means that production is less susceptible to fluctuations in the quality of acidic reagents, which can vary significantly between batches in traditional processes. This consistency ensures that delivery schedules are met reliably, reducing the need for safety stock and freeing up working capital for other strategic investments.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies environmental risks, but this catalytic route is inherently designed for scalability. The heterogeneous nature of the catalysts facilitates easy separation in large reactors, avoiding the emulsions and phase separation issues common with liquid acids. From a regulatory standpoint, the absence of genotoxic alkylating agents and toxic gases simplifies the environmental impact assessment and permitting process for new production lines. This alignment with strict environmental standards future-proofs the manufacturing asset against tightening global regulations on volatile organic compounds and hazardous waste, ensuring long-term operational viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these specifics helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of using solid acid catalysts over liquid acids in this synthesis?
A: Solid acid catalysts, such as sulfonic acid cation exchange resins, eliminate the need for difficult neutralization and wastewater treatment associated with liquid sulfuric acid, significantly simplifying downstream processing and reducing environmental impact.
Q: Can the 4,5-dialkoxy-2-imidazolidinone intermediate be used without isolation?
A: Yes, the patent explicitly states that the 4,5-dialkoxy-2-imidazolidinone compound can be continuously used in the subsequent hydrogenation reaction without isolation and purification, which streamlines the production workflow.
Q: Which metal catalysts are suitable for the hydrogenation step?
A: The process supports a variety of metal catalysts including palladium on carbon (Pd/C), platinum on carbon (Pt/C), platinum-palladium alloys, and Raney nickel, offering flexibility based on availability and cost considerations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Imidazolidinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving demands of the global pharmaceutical and agrochemical industries. Our technical team has extensively analyzed the pathways described in CN101395140B and possesses the expertise to implement these catalytic hydrogenation processes with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale innovation to industrial reality is seamless. Our facilities are equipped with state-of-the-art high-pressure hydrogenation reactors and rigorous QC labs capable of meeting stringent purity specifications required for GMP-grade intermediates.
We invite you to collaborate with us to leverage these technological advancements for your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a sustainable, cost-effective, and high-quality supply of 2-imidazolidinone derivatives that will strengthen your product pipeline and enhance your market competitiveness.
