Advanced Manufacturing of Anastrozole via Mild Temperature Phase Transfer Catalysis
Advanced Manufacturing of Anastrozole via Mild Temperature Phase Transfer Catalysis
The pharmaceutical industry continuously seeks robust synthetic pathways for critical oncology medications, and the production of 1-[3,5-bis(2,2-dimethyl)ethylcyano]benzyl triazole, commonly known as Anastrozole, represents a significant area of process optimization. Patent CN101074215A introduces a refined methodology that addresses longstanding challenges in yield, purity, and operational complexity associated with traditional manufacturing routes. This technical insight explores how the disclosed two-step synthesis leverages mild reaction conditions and efficient phase transfer catalysis to deliver a superior quality active pharmaceutical ingredient suitable for large-scale commercialization. By shifting away from harsh thermal conditions and complex purification techniques, this approach offers a compelling value proposition for stakeholders focused on supply chain resilience and cost efficiency in the competitive breast cancer treatment market.
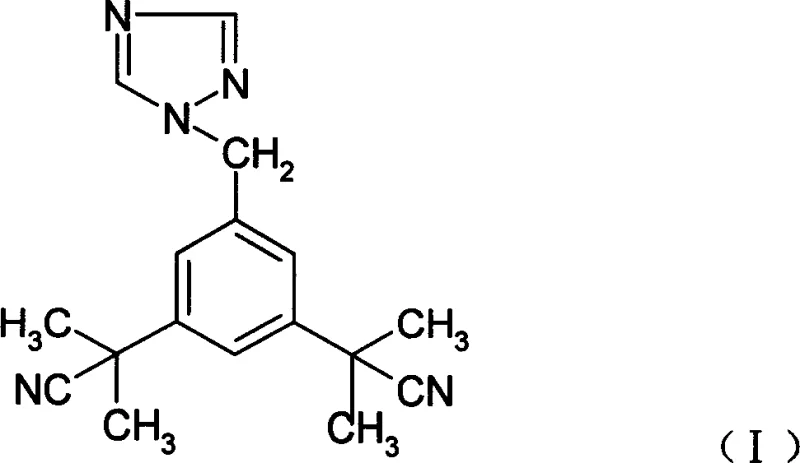
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Anastrozole has been plagued by inefficient multi-step sequences and demanding purification requirements that hinder industrial scalability. Early methodologies, such as those described in EP0296749, relied on starting materials like 3,5-dibromomethyltoluene which necessitated complex cyanation and methylation steps before the final coupling, often resulting in low overall yields and difficult post-treatment protocols requiring column chromatography. Furthermore, alternative routes utilizing methyl 3,5-dimethylbenzoate involved up to six distinct reaction steps, incorporating expensive reducing agents like sodium borohydride and suffering from yields typically below 50 percent. Even more recent attempts using phase transfer catalysis often required high-temperature reflux in xylenes, which inadvertently promoted the formation of structural isomers and increased the total impurity profile to unacceptable levels for direct pharmaceutical use without extensive recrystallization.
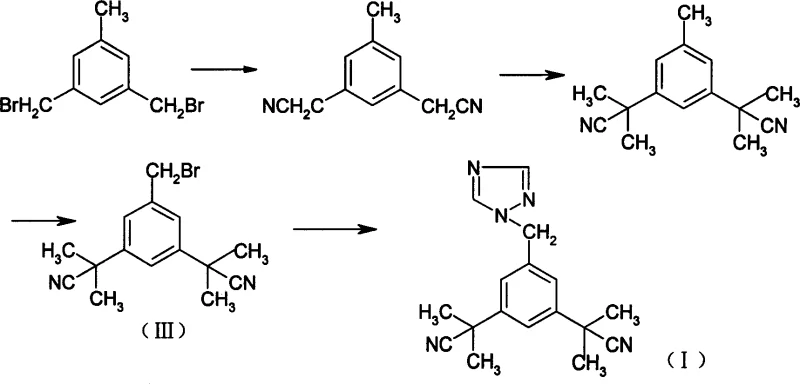
The Novel Approach
In stark contrast to these legacy processes, the method disclosed in CN101074215A streamlines production by utilizing 3,5-bis[(2,2-dimethyl)cyanomethyl]toluene as a readily accessible starting material, effectively bypassing the need for early-stage functional group manipulations. The core innovation lies in the final coupling step, where the brominated intermediate reacts with 1,2,4-triazole under mild conditions ranging from 20 to 40 degrees Celsius in solvents such as dichloromethane or ethyl acetate. This strategic shift not only simplifies the operational workflow to merely two primary steps but also fundamentally alters the impurity landscape by suppressing thermal degradation and isomerization side reactions. The result is a highly efficient pathway that eliminates the need for column chromatography entirely, relying instead on a straightforward acid-base workup to achieve high-purity crystalline product suitable for immediate downstream formulation.
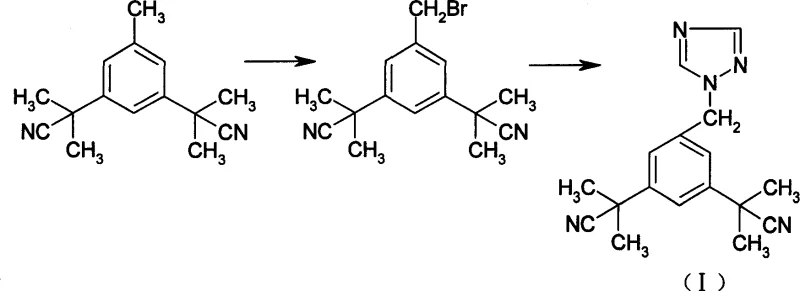
Mechanistic Insights into Phase Transfer Catalyzed N-Alkylation
The success of this optimized route hinges on the precise application of phase transfer catalysis to facilitate the nucleophilic substitution between the benzyl bromide intermediate and the triazole nucleophile. By employing catalysts such as tetrabutylammonium bromide or tetrabutylammonium iodide, the reaction system effectively transports the triazole anion into the organic phase where the alkylation occurs, significantly accelerating the reaction rate without the need for elevated temperatures. This mechanistic advantage is critical because it allows the reaction to proceed to completion within 6 to 10 hours at ambient or slightly warmed conditions, thereby preserving the integrity of the sensitive nitrile groups and preventing the thermal rearrangement of the triazole ring that plagues high-temperature alternatives. The choice of solvent further enhances this selectivity, as dichloromethane and ethyl acetate provide an ideal medium for the phase transfer agent to operate while maintaining the solubility of the organic intermediates.
Controlling the impurity profile is another paramount aspect of this chemical mechanism, specifically regarding the suppression of the 1,3,4-triazole isomer which shares similar mass spectral properties with the target molecule. The patent data indicates that maintaining strict temperature control between 20 and 40 degrees Celsius is essential to kinetically favor the formation of the desired 1,2,4-triazole linkage over the thermodynamic isomer. Additionally, the purity of the starting 1,2,4-triazole reagent must be maintained above 98 percent to prevent the introduction of pre-existing isomeric contaminants that could propagate through the synthesis. Through this rigorous control of reaction parameters and reagent quality, the process consistently achieves total impurity levels below 0.3 percent and specific isomer content below 0.1 percent, demonstrating a level of chemical precision that far exceeds previous industrial standards.
How to Synthesize Anastrozole Efficiently
The implementation of this synthesis strategy requires careful attention to the bromination step followed by the critical phase transfer alkylation to ensure maximum yield and purity. The process begins with the radical bromination of the toluene derivative using N-bromosuccinimide, followed by the coupling reaction which demands precise pH control during the workup phase to isolate the free base effectively. Operators must adhere to the specified temperature ranges and catalyst loading to replicate the high-efficiency results documented in the patent examples, ensuring that the final crystallization yields a product with the correct polymorphic form and particle size distribution. For a detailed breakdown of the specific reagent quantities, reaction times, and isolation procedures validated in the patent examples, please refer to the standardized synthesis guide below.
- Brominate 3,5-bis[(2,2-dimethyl)cyanomethyl]toluene using N-bromosuccinimide (NBS) and benzoyl peroxide in chloroform or carbon tetrachloride under reflux to obtain the bromo-intermediate.
- React the bromo-intermediate with 1,2,4-triazole in dichloromethane or ethyl acetate using a phase transfer catalyst like tetrabutylammonium bromide at 20-40°C.
- Perform acid-base workup by dissolving the crude oil in hydrochloric acid, filtering insolubles, adjusting pH to 8-9 with sodium hydroxide to precipitate pure Anastrozole.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this manufacturing route offers substantial benefits by fundamentally restructuring the cost drivers associated with Anastrozole production. The elimination of column chromatography represents a massive reduction in both solvent consumption and processing time, removing a significant bottleneck that typically limits batch throughput and increases operational expenditures in fine chemical manufacturing. Furthermore, the ability to utilize readily available starting materials reduces dependency on complex, multi-step precursor supply chains, thereby enhancing the overall reliability and continuity of supply for procurement managers who must mitigate risks associated with raw material shortages. The simplified post-treatment protocol also translates to reduced labor costs and faster turnaround times between batches, allowing manufacturing facilities to respond more agilely to market demand fluctuations without compromising on product quality standards.
- Cost Reduction in Manufacturing: The removal of expensive purification steps like column chromatography and the replacement of high-boiling solvents with recoverable options like dichloromethane drastically lowers the variable cost per kilogram of produced API. By avoiding the use of costly reducing agents and minimizing the number of unit operations from six down to two, the process inherently reduces energy consumption and waste disposal costs, leading to significant margin improvements for the final product. This lean manufacturing approach ensures that the economic viability of the project remains robust even when facing fluctuations in raw material pricing or regulatory compliance costs.
- Enhanced Supply Chain Reliability: Sourcing 3,5-bis[(2,2-dimethyl)cyanomethyl]toluene is significantly more straightforward than procuring specialized esters or managing the logistics of multiple interim intermediates required by older synthetic routes. This simplification of the bill of materials reduces the number of potential failure points in the supply chain, ensuring that production schedules can be maintained with greater predictability and less risk of disruption due to vendor delays. Additionally, the robustness of the reaction conditions means that the process is less sensitive to minor variations in utility supply or environmental conditions, further stabilizing the manufacturing output.
- Scalability and Environmental Compliance: The mild reaction temperatures and the use of standard organic solvents make this process highly amenable to scale-up in existing multipurpose reactors without requiring specialized high-pressure or high-temperature equipment. The simplified workup generates less hazardous waste compared to routes involving heavy metal catalysts or large volumes of silica gel, aligning better with modern environmental, health, and safety regulations. This ease of scalability ensures that production capacity can be expanded rapidly to meet global demand while maintaining a smaller environmental footprint and lower regulatory burden.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this specific Anastrozole synthesis pathway. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation to ensure accuracy and relevance for technical decision-makers. Understanding these nuances is critical for evaluating the feasibility of technology transfer and the potential impact on your current manufacturing portfolio.
Q: How does this new process improve impurity profiles compared to prior art?
A: By maintaining reaction temperatures between 20-40°C and using specific phase transfer catalysts, the formation of the 1,3,4-triazole isomer impurity is suppressed to below 0.1%, significantly higher purity than previous methods which often exceeded 1% impurities.
Q: Why is column chromatography eliminated in this manufacturing route?
A: The novel workup procedure utilizes a simple acid-base extraction and crystallization method. The crude product is dissolved in acid, filtered to remove insolubles, and then precipitated by pH adjustment, rendering expensive and time-consuming column chromatography unnecessary for industrial scale.
Q: What are the cost advantages of the starting materials used?
A: The process utilizes 3,5-bis[(2,2-dimethyl)cyanomethyl]toluene, which is readily available on the market, avoiding the multi-step synthesis from expensive precursors like methyl 3,5-dimethylbenzoate required in older six-step routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Anastrozole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of deploying advanced synthetic methodologies to ensure the consistent availability of high-quality oncology therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our state-of-the-art facilities to implement processes like the one described in CN101074215A with precision and efficiency. We are committed to meeting stringent purity specifications through our rigorous QC labs, ensuring that every batch of Anastrozole we produce meets the exacting standards required for global pharmaceutical registration and patient safety.
We invite you to engage with our technical procurement team to discuss how this optimized manufacturing route can be tailored to your specific volume requirements and cost targets. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this streamlined process. We encourage you to contact us today to索取 specific COA data and route feasibility assessments that will demonstrate our capability to be your long-term strategic partner in the supply of high-value pharmaceutical ingredients.
