Advanced Palladium-Catalyzed Synthesis of 1,5-Dihydro-2H-Pyrrole-2-One Scaffolds for Commercial API Production
Introduction to Novel Pyrrol-2-One Synthesis Technology
The structural motif of 1,5-dihydro-2H-pyrrol-2-one represents a critical pharmacophore found in numerous bioactive natural products and therapeutic agents, necessitating robust and efficient synthetic methodologies for pharmaceutical development. As detailed in Chinese Patent CN112694430B, a groundbreaking preparation method has been established that leverages palladium-catalyzed bis-carbonylation to construct this valuable heterocyclic core with exceptional precision. This technology addresses the longstanding challenges associated with traditional carbonylation reactions, which often suffer from harsh conditions or limited substrate scope, by utilizing readily available benzyl chlorides and propargyl amines as starting materials. The significance of this scaffold is underscored by its presence in potent antibiotics like althiomycin and hypoglycemic agents such as glimepiride, highlighting the urgent need for reliable supply chains capable of delivering high-purity intermediates. By integrating a solid carbon monoxide substitute, specifically phenyl 1,3,5-tricarboxylate, the process eliminates the safety hazards associated with handling gaseous CO, thereby streamlining operational protocols for industrial applications.
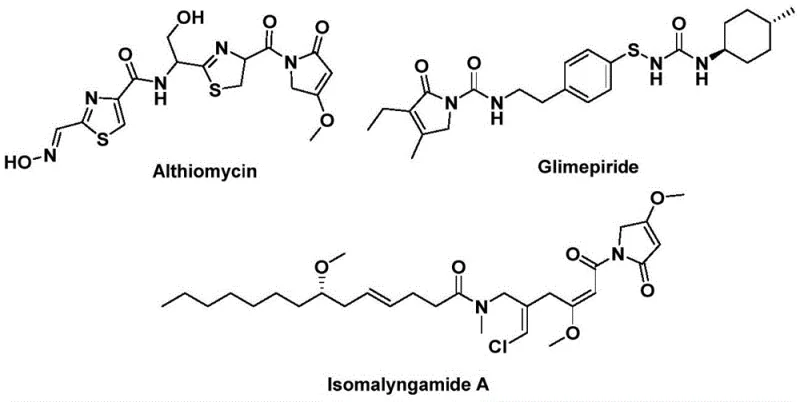
Furthermore, the versatility of this synthetic approach allows for the rapid generation of diverse libraries of pyrrol-2-one derivatives, facilitating structure-activity relationship (SAR) studies essential for drug discovery programs. The patent explicitly outlines a one-step efficient synthesis that not only accelerates the timeline from raw materials to final product but also ensures high atom economy and reduced waste generation. For procurement managers and supply chain directors, this translates to a more resilient sourcing strategy for complex pharmaceutical intermediates, reducing dependency on multi-step, low-yield legacy processes. The ability to tolerate a wide array of functional groups means that a single optimized protocol can service multiple project pipelines, significantly enhancing the overall agility of R&D operations. Consequently, adopting this methodology positions manufacturers as a reliable pharmaceutical intermediate supplier capable of meeting the rigorous demands of modern medicinal chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 1,5-dihydro-2H-pyrrol-2-one skeletons via carbonylation has been fraught with significant technical hurdles that impede large-scale adoption in the fine chemical industry. Traditional methods frequently rely on the direct use of toxic and flammable carbon monoxide gas, which necessitates specialized high-pressure equipment and stringent safety protocols that drastically increase capital expenditure and operational complexity. Moreover, conventional catalytic systems often exhibit poor functional group tolerance, leading to side reactions and difficult purification scenarios that erode overall process yields and inflate production costs. Many existing routes require pre-functionalized substrates that are expensive and difficult to source, creating bottlenecks in the supply chain that delay critical project milestones. The lack of generality in older methodologies means that slight modifications to the substrate structure often require a complete re-optimization of reaction conditions, rendering them inefficient for the rapid iteration required in contemporary drug development. Additionally, the use of stoichiometric amounts of hazardous reagents generates substantial chemical waste, posing environmental compliance challenges that modern manufacturers strive to avoid.
The Novel Approach
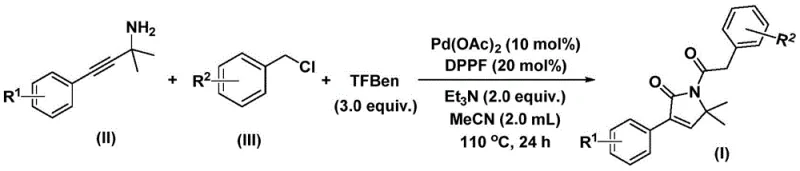
In stark contrast, the novel palladium-catalyzed bis-carbonylation method described in the patent offers a transformative solution by utilizing bench-stable solid reagents to generate carbon monoxide in situ. This approach replaces dangerous gaseous CO with phenyl 1,3,5-tricarboxylate, allowing the reaction to proceed safely in standard glassware at atmospheric pressure or mild sealing, significantly lowering the barrier to entry for implementation. The use of simple benzyl chlorides and propargyl amines as starting materials ensures that the feedstock is cheap, abundant, and commercially accessible, directly addressing the cost reduction in pharmaceutical intermediate manufacturing. The reaction conditions are remarkably mild, operating effectively between 100°C and 120°C in acetonitrile, which simplifies thermal management and energy consumption profiles. Most importantly, this method achieves high reaction efficiency with yields reaching up to 92% for certain substrates, demonstrating superior performance compared to legacy techniques. The broad substrate compatibility allows for the seamless integration of electron-rich and electron-deficient groups, providing a versatile platform for synthesizing a wide spectrum of bioactive analogues without the need for extensive protective group chemistry.
Mechanistic Insights into Palladium-Catalyzed Bis-Carbonylation
The mechanistic pathway of this transformation involves a sophisticated sequence of organometallic steps orchestrated by the palladium catalyst, ensuring high regioselectivity and conversion rates. The cycle initiates with the oxidative addition of the palladium(0) species into the carbon-chlorine bond of the benzyl chloride, generating a reactive benzyl-palladium intermediate that serves as the foundation for subsequent carbonyl insertion. Following this activation, a molecule of carbon monoxide, released thermally from the phenyl 1,3,5-tricarboxylate surrogate, inserts into the palladium-carbon bond to form an acyl-palladium species. This acyl intermediate then undergoes a nucleophilic attack by the nitrogen atom of the propargyl amine, facilitating the formation of a five-membered ring palladium complex through an intramolecular cyclization event. The elegance of this mechanism lies in the second carbonyl insertion, where another CO molecule inserts into the newly formed palladium-carbon bond, expanding the ring system transiently before reductive elimination occurs. This final reductive elimination step releases the desired 1,5-dihydro-2H-pyrrol-2-one product and regenerates the active palladium(0) catalyst, closing the catalytic loop and sustaining the reaction turnover.
Understanding the impurity profile is crucial for R&D directors focused on purity specifications, and this mechanism inherently minimizes common byproducts through its concerted nature. The use of the specific ligand 1,1'-bis(diphenylphosphino)ferrocene (DPPF) stabilizes the palladium center, preventing premature decomposition or the formation of inactive palladium black, which is a common failure mode in carbonylation reactions. The controlled release of CO from the tricarboxylate source prevents local concentration spikes that could lead to oligomerization or polymerization of the alkyne moiety, ensuring a clean reaction profile. Furthermore, the basic conditions provided by triethylamine facilitate the deprotonation steps necessary for cyclization without promoting hydrolysis of the sensitive lactam ring. This precise control over the reaction environment results in a crude product mixture that is significantly cleaner than those obtained from non-catalytic thermal cyclizations, thereby reducing the burden on downstream purification units. The robustness of this catalytic cycle against various substituents confirms its utility for producing high-purity pharmaceutical intermediates with consistent quality attributes.
How to Synthesize 1,5-Dihydro-2H-Pyrrole-2-One Efficiently
The practical execution of this synthesis is designed to be straightforward and adaptable to standard laboratory and pilot plant infrastructure, requiring no exotic equipment or specialized handling beyond typical organic synthesis protocols. To initiate the process, the requisite amounts of palladium acetate, DPPF ligand, triethylamine base, phenyl 1,3,5-tricarboxylate, propargyl amine derivative, and benzyl chloride derivative are charged into a reaction vessel containing acetonitrile as the solvent. The mixture is then heated to a temperature range of 100°C to 120°C and maintained under stirring for a period of 24 to 48 hours, allowing sufficient time for the bis-carbonylation and cyclization events to reach completion. Upon confirmation of reaction completion via analytical monitoring, the mixture is cooled, filtered to remove insoluble palladium residues, and the filtrate is concentrated. The resulting crude material is typically purified using standard silica gel column chromatography to afford the target 1,5-dihydro-2H-pyrrole-2-one compound in high purity. Detailed standardized synthesis steps see the guide below.
- Combine palladium acetate, DPPP ligand, triethylamine, phenyl 1,3,5-tricarboxylate (CO source), propargyl amine, and benzyl chloride in acetonitrile solvent within a reaction vessel.
- Heat the reaction mixture to a temperature range of 100-120°C and maintain stirring for a duration of 24 to 48 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify the crude product via column chromatography to isolate the target 1,5-dihydro-2H-pyrrole-2-one compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers compelling advantages that directly address the pain points of cost, reliability, and scalability inherent in complex intermediate manufacturing. The shift from gaseous carbon monoxide to a solid CO surrogate fundamentally alters the risk profile of the operation, eliminating the need for expensive high-pressure autoclaves and specialized gas handling infrastructure, which translates to significant capital cost avoidance. The reliance on commodity chemicals like benzyl chlorides and simple alkynes ensures a stable and competitive supply base, mitigating the risks associated with sourcing custom-built or scarce reagents that often plague niche synthetic routes. High reaction yields, experimentally demonstrated to exceed 90% in optimal cases, mean that less raw material is wasted per kilogram of product, driving down the variable cost of goods sold and improving overall margin potential. The simplicity of the workup procedure, involving basic filtration and chromatography, reduces the demand for complex separation technologies and minimizes solvent consumption, aligning with green chemistry principles and reducing waste disposal costs. These factors collectively enhance the economic viability of producing this scaffold, making it an attractive option for long-term supply contracts.
- Cost Reduction in Manufacturing: The elimination of high-pressure equipment and hazardous gas handling significantly lowers the barrier to entry and operational overheads for manufacturers. By utilizing inexpensive and widely available starting materials such as benzyl chlorides and propargyl amines, the direct material costs are minimized compared to routes requiring complex precursors. The high catalytic efficiency and excellent yields reduce the amount of feedstock required per unit of output, directly improving the cost-efficiency ratio. Furthermore, the simplified purification process reduces solvent usage and labor hours associated with extensive workup procedures, contributing to substantial overall cost savings in the production lifecycle.
- Enhanced Supply Chain Reliability: The use of commercially off-the-shelf reagents ensures that supply disruptions are unlikely, as these materials are produced at scale by multiple global vendors. The robustness of the reaction conditions allows for flexible scheduling and batch processing without the need for highly specialized operator training or unique facility requirements. This flexibility enables manufacturers to respond quickly to fluctuating demand signals from downstream API producers, ensuring continuity of supply even during market volatility. The stability of the solid CO source also simplifies logistics and storage, removing the complexities associated with transporting and storing compressed gases.
- Scalability and Environmental Compliance: The reaction operates in common organic solvents like acetonitrile, which are well-understood in terms of recovery and recycling, facilitating easy scale-up from gram to tonne quantities. The absence of toxic gaseous reagents simplifies environmental permitting and reduces the regulatory burden related to air emissions and worker safety exposure limits. The high atom economy of the bis-carbonylation process ensures that a maximum proportion of reactant mass is incorporated into the final product, minimizing the generation of chemical waste streams. This alignment with sustainable manufacturing practices enhances the corporate social responsibility profile of the supply chain and meets the increasingly strict environmental standards of global pharmaceutical clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed synthesis route, derived directly from the patent specifications and experimental data. These insights are intended to clarify the operational parameters and potential benefits for stakeholders evaluating this technology for integration into their manufacturing portfolios. Understanding these details is essential for assessing the feasibility of adopting this method for specific project requirements and ensuring alignment with quality and safety standards. The answers reflect the proven capabilities of the method as demonstrated in the cited examples.
Q: What are the key advantages of this palladium-catalyzed method over traditional carbonylation?
A: This method utilizes bench-stable benzyl chlorides and propargyl amines instead of hazardous gaseous carbon monoxide, employing phenyl 1,3,5-tricarboxylate as a safe CO surrogate. It operates under relatively mild conditions (100-120°C) with excellent substrate compatibility and high isolated yields up to 92%.
Q: Is this synthesis route scalable for industrial API intermediate production?
A: Yes, the protocol uses commercially available reagents like palladium acetate and standard organic solvents (acetonitrile). The workup involves simple filtration and standard column chromatography, avoiding complex purification steps that often hinder scale-up, making it highly suitable for commercial manufacturing.
Q: What types of functional groups are tolerated in this reaction system?
A: The reaction demonstrates broad functional group tolerance, successfully accommodating electron-donating groups (methoxy, methyl) and electron-withdrawing groups (fluoro, chloro, bromo, trifluoromethyl, cyano) on both the benzyl chloride and propargyl amine substrates without significant loss in efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-Dihydro-2H-Pyrrole-2-One Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in accelerating drug development and securing the global supply of vital medicines. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries like this palladium-catalyzed route can be seamlessly transitioned to industrial reality. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in process optimization allows us to fine-tune reaction parameters to maximize yield and minimize impurities, guaranteeing a consistent and reliable product stream for our partners. By leveraging advanced catalytic technologies, we help our clients reduce time-to-market and achieve their commercial objectives with confidence.
We invite you to collaborate with us to explore the full potential of this innovative synthesis for your specific pipeline needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and quality targets. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our technical prowess can drive value for your organization. Together, we can build a more resilient and efficient supply chain for the next generation of therapeutic agents.
