Advanced Palladium-Catalyzed Carbonylation for Scalable 1,5-Dihydro-2H-Pyrrole-2-One Production
Advanced Palladium-Catalyzed Carbonylation for Scalable 1,5-Dihydro-2H-Pyrrole-2-One Production
The landscape of pharmaceutical intermediate synthesis is constantly evolving, driven by the need for more efficient, safer, and cost-effective methodologies. A significant breakthrough in this domain is detailed in patent CN112694430B, which discloses a novel preparation method for 1,5-dihydro-2H-pyrrole-2-one compounds. This structural backbone is of paramount importance in medicinal chemistry, serving as the core scaffold for potent bioactive molecules such as the antibiotic althiomycin, the hypoglycemic agent glimepiride, and the anticancer candidate isomalyngamide A. As shown in the structural diversity of these natural products and drugs, the ability to construct this five-membered lactam ring efficiently is a critical capability for any advanced chemical manufacturer.
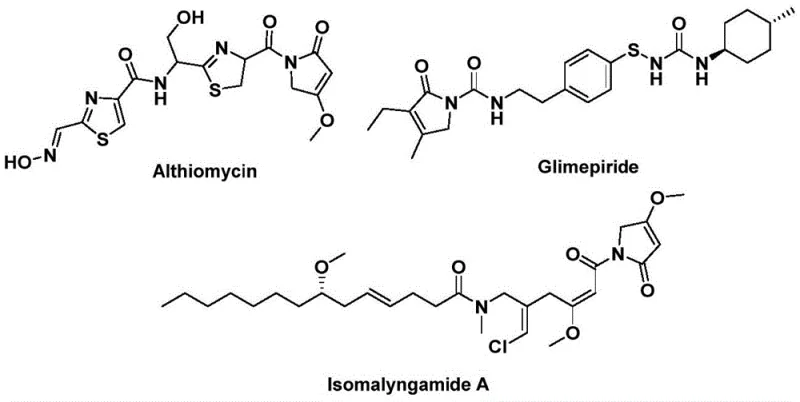
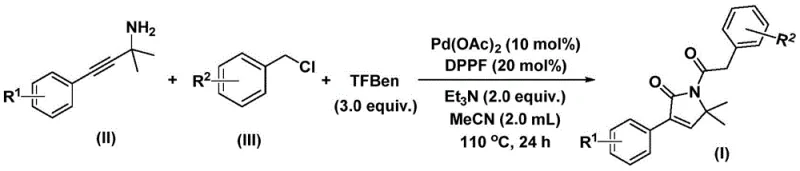
For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this patent represents a shift towards more streamlined manufacturing. The disclosed method leverages palladium-catalyzed bis-carbonylation, utilizing readily available starting materials like propargylamines and benzyl chlorides. By replacing hazardous gaseous carbon monoxide with a solid substitute, the process not only enhances operational safety but also simplifies the engineering controls required for scale-up. This innovation directly addresses the industry's demand for cost reduction in API manufacturing by minimizing complex multi-step sequences and reducing the reliance on expensive or dangerous reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of 1,5-dihydro-2H-pyrrol-2-one skeletons has been fraught with challenges that hinder large-scale production. Conventional routes often rely on multi-step sequences involving harsh cyclization conditions, which can lead to poor atom economy and significant waste generation. Furthermore, many established methods require the use of gaseous carbon monoxide, a highly toxic and flammable reagent that necessitates specialized high-pressure equipment and rigorous safety protocols, thereby inflating capital expenditure and operational costs. The limited substrate scope of older methodologies often results in low yields when electron-withdrawing or sterically hindered groups are present, forcing process chemists to engage in time-consuming optimization campaigns. These factors collectively create bottlenecks in the supply chain, extending lead times and increasing the overall cost of goods for downstream drug developers.
The Novel Approach
In stark contrast, the methodology described in patent CN112694430B offers a transformative one-pot solution that bypasses these historical hurdles. By employing a palladium catalyst system in conjunction with a solid carbon monoxide substitute (TFBen), the reaction achieves efficient bis-carbonylation under relatively mild thermal conditions (100–120°C). This approach eliminates the need for handling toxic CO gas, significantly lowering the barrier to entry for safe manufacturing. The reaction exhibits remarkable functional group tolerance, accommodating a wide array of substituents on both the propargylamine and benzyl chloride components without compromising yield. This robustness ensures that the process is not merely a laboratory curiosity but a viable industrial pathway capable of delivering high-purity pharmaceutical intermediates with consistent quality, thereby facilitating the commercial scale-up of complex heterocyclic compounds.
Mechanistic Insights into Palladium-Catalyzed Bis-Carbonylation
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to optimize the process for specific analogs. The reaction initiates with the oxidative addition of the palladium catalyst into the carbon-chlorine bond of the benzyl chloride, generating a reactive benzylpalladium intermediate. Subsequently, carbon monoxide, which is liberated in situ from the phenol 1,3,5-tricarboxylate surrogate, inserts into this intermediate to form an acylpalladium species. This acyl species then undergoes nucleophilic attack by the propargylamine, setting the stage for the formation of the five-membered ring palladium intermediate. The elegance of this mechanism lies in the second insertion of a carbon monoxide molecule, which expands the coordination sphere before a final reductive elimination step releases the desired 1,5-dihydro-2H-pyrrol-2-one product and regenerates the active catalyst.
From an impurity control perspective, this mechanism offers distinct advantages. The use of a solid CO surrogate allows for a controlled, steady release of carbon monoxide, preventing the accumulation of free CO that could lead to side reactions or catalyst poisoning. Furthermore, the specific ligand environment provided by DPPP (1,1'-bis(diphenylphosphino)ferrocene) stabilizes the palladium center throughout the catalytic cycle, minimizing the formation of palladium black and other metal-derived impurities that are notoriously difficult to remove from final API batches. The reaction conditions, specifically the use of acetonitrile as a solvent and triethylamine as a base, are optimized to solubilize the intermediates while maintaining a pH environment that favors the desired cyclization over polymerization of the alkyne. This precise control over the reaction trajectory ensures a clean impurity profile, which is a critical metric for regulatory compliance in pharmaceutical manufacturing.
How to Synthesize 1,5-Dihydro-2H-Pyrrole-2-One Efficiently
The practical execution of this synthesis is designed for simplicity and reproducibility, making it highly attractive for process development teams. The protocol involves charging a reaction vessel with the palladium catalyst, ligand, base, CO surrogate, and the two primary organic substrates in acetonitrile. The mixture is then heated to promote the cascade of carbonylation and cyclization events. Following the reaction period, the workup is straightforward, involving filtration to remove insoluble salts and catalyst residues, followed by standard purification techniques. For a detailed breakdown of the specific stoichiometric ratios, temperature profiles, and workup procedures validated in the patent examples, please refer to the standardized guide below.
- Combine palladium acetate, DPPP ligand, solid CO surrogate (TFBen), triethylamine, propargylamine, and benzyl chloride in acetonitrile.
- Heat the reaction mixture to 110°C and stir for 24 hours under inert atmosphere to facilitate bis-carbonylation and cyclization.
- Filter the mixture, adsorb onto silica gel, and purify via column chromatography to isolate the target pyrrol-2-one compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible strategic benefits beyond mere chemical yield. The shift towards this palladium-catalyzed route fundamentally alters the cost structure of producing these valuable heterocycles by leveraging commodity chemicals. The starting materials, specifically benzyl chlorides and propargylamines, are widely available from global chemical suppliers, ensuring a resilient supply chain that is not dependent on niche or single-source vendors. This abundance of raw materials mitigates the risk of supply disruptions and price volatility, which are common concerns in the fine chemical sector. Moreover, the operational simplicity of the one-pot process reduces the requirement for specialized reactor configurations, allowing for flexible manufacturing across existing multipurpose facilities.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the elimination of hazardous gas handling infrastructure and the reduction of unit operations. By utilizing a solid CO surrogate, manufacturers avoid the capital and maintenance costs associated with high-pressure CO reactors and scrubbing systems. Additionally, the high conversion rates and selectivity observed in this method minimize the loss of valuable starting materials, leading to substantial improvements in overall material efficiency. The simplified downstream processing, which avoids complex extraction or distillation steps often required to remove volatile byproducts, further contributes to a lower cost of goods sold, enabling more competitive pricing for downstream clients.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent output quality, which is vital for maintaining just-in-time delivery schedules. Since the reaction tolerates a broad range of functional groups, the same manufacturing line can be easily adapted to produce various analogs within the 1,5-dihydro-2H-pyrrole-2-one family without extensive requalification. This flexibility allows suppliers to respond rapidly to changing market demands or custom synthesis requests. The use of stable, shelf-stable reagents like the solid CO surrogate also simplifies inventory management, reducing the need for specialized storage conditions and extending the shelf life of critical production inputs.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram or ton scale is facilitated by the homogeneous nature of the reaction and the absence of exothermic gas feeds. The mild temperature range (100–120°C) is compatible with standard heating media like steam or thermal oil, avoiding the need for cryogenic cooling or extreme heating. From an environmental standpoint, the atom economy of the bis-carbonylation is superior to traditional condensation methods, resulting in less chemical waste. The ability to run the reaction in acetonitrile, a solvent with well-established recovery and recycling protocols, aligns with modern green chemistry principles and helps facilities meet increasingly stringent environmental regulations regarding VOC emissions and waste disposal.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology for their specific applications, we have compiled answers to common inquiries regarding the process parameters and scope. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a realistic view of the method's capabilities and limitations. Understanding these nuances is essential for integrating this synthetic route into existing development pipelines.
Q: What is the primary advantage of this carbonylation method over traditional cyclization?
A: This method utilizes a solid carbon monoxide substitute instead of toxic CO gas, enabling safer operation while achieving high yields through a direct one-pot bis-carbonylation process.
Q: Does this protocol tolerate diverse functional groups on the benzyl chloride substrate?
A: Yes, the reaction demonstrates excellent substrate compatibility, successfully accommodating electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups such as halogens and trifluoromethyl.
Q: What are the typical reaction conditions required for optimal conversion?
A: Optimal conversion is achieved using palladium acetate (10 mol%) and DPPP ligand in acetonitrile at 110°C for 24 hours, ensuring complete consumption of the propargylamine starting material.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-Dihydro-2H-Pyrrole-2-One Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and development. Our team of expert process chemists has thoroughly analyzed the potential of the palladium-catalyzed bis-carbonylation route described in CN112694430B and is prepared to implement it for your projects. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop to plant floor is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of intermediate meets the exacting standards required for pharmaceutical applications.
We invite you to collaborate with us to leverage this cutting-edge technology for your next campaign. By partnering with our technical team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and target molecules. We encourage you to contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary candidates. Let us help you secure a competitive advantage through superior chemistry and reliable supply.
