Advanced Palladium-Catalyzed Carbonylation for Scalable 1,5-Dihydro-2H-Pyrrole-2-One Production
Introduction to Next-Generation Pyrrol-2-One Synthesis
The structural motif of 1,5-dihydro-2H-pyrrol-2-one represents a critical pharmacophore found in numerous bioactive natural products and therapeutic agents, necessitating robust and scalable synthetic methodologies for modern drug discovery. As detailed in the recent patent CN112694430B, a novel preparation method has been developed that leverages palladium-catalyzed bis-carbonylation to construct this valuable heterocyclic core with exceptional efficiency. This technological breakthrough addresses the longstanding challenges associated with synthesizing complex lactam scaffolds, offering a streamlined pathway that integrates readily available starting materials such as propargylamines and benzyl chlorides. The significance of this scaffold is underscored by its presence in potent antibiotics like Althiomycin and hypoglycemic agents such as Glimepiride, highlighting the urgent industrial demand for reliable production capabilities.
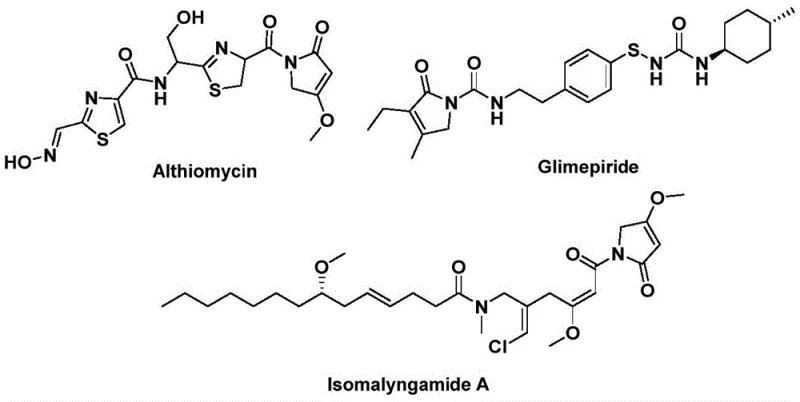
Furthermore, the versatility of this synthetic approach allows for the rapid generation of diverse analog libraries, which is essential for structure-activity relationship (SAR) studies in early-stage pharmaceutical development. By utilizing a solid carbon monoxide substitute rather than hazardous gaseous CO, the process inherently improves safety profiles while maintaining high atom economy. For global procurement teams and R&D directors seeking a reliable pharmaceutical intermediate supplier, understanding the mechanistic nuances and scalability of this patented route provides a strategic advantage in securing supply chains for next-generation therapeutics. The following analysis dissects the technical merits and commercial implications of adopting this advanced carbonylation technology.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes to 1,5-dihydro-2H-pyrrol-2-one derivatives often rely on multi-step sequences involving the condensation of amines with activated carboxylic acid derivatives followed by separate cyclization events. These classical approaches frequently suffer from poor atom economy, requiring stoichiometric amounts of coupling reagents and generating significant chemical waste that complicates downstream purification. Moreover, many conventional methods necessitate the use of harsh reaction conditions, such as strong acids or high temperatures, which can lead to the decomposition of sensitive functional groups and limit the scope of compatible substrates. The reliance on gaseous carbon monoxide in some carbonylation protocols introduces severe safety hazards and engineering complexities, particularly when attempting to scale these reactions from milligram to kilogram quantities in a standard laboratory or pilot plant setting.
The Novel Approach
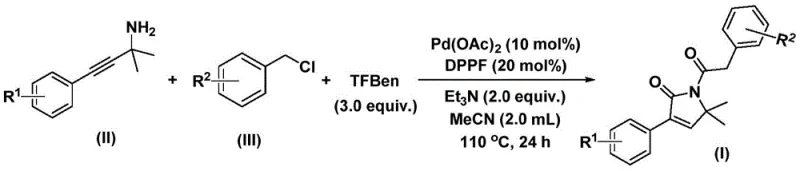
In stark contrast, the methodology disclosed in patent CN112694430B introduces a transformative one-pot strategy that merges oxidative addition, double carbonyl insertion, and cyclization into a single operational sequence. This novel approach utilizes a palladium catalyst system in conjunction with a phenol ester derivative (TFBen) as a safe and convenient solid source of carbon monoxide, effectively bypassing the logistical nightmares associated with gas handling. The reaction proceeds under relatively mild thermal conditions, typically between 100°C and 120°C, allowing for the preservation of delicate functional groups that would otherwise be compromised in more aggressive synthetic environments. By directly coupling propargylamines with benzyl chlorides, this method drastically reduces the number of unit operations required, thereby minimizing solvent consumption and labor costs while maximizing overall throughput for industrial applications.
Mechanistic Insights into Palladium-Catalyzed Bis-Carbonylation
The catalytic cycle underpinning this transformation is a sophisticated orchestration of organometallic steps that ensures high regioselectivity and yield. The reaction initiates with the oxidative addition of the palladium(0) species into the carbon-chlorine bond of the benzyl chloride substrate, generating a reactive benzyl-palladium(II) intermediate. Subsequently, carbon monoxide, which is liberated in situ from the thermal decomposition of the 1,3,5-tricarboxylic acid phenol ester, inserts into the palladium-carbon bond to form an acyl-palladium species. This acyl intermediate then undergoes nucleophilic attack by the propargylamine, followed by a second crucial carbon monoxide insertion event that extends the carbon chain and sets the stage for ring closure.
Following the second CO insertion, the resulting six-membered palladium intermediate undergoes an intramolecular cyclization to form the five-membered lactam ring characteristic of the 1,5-dihydro-2H-pyrrol-2-one core. The final step involves reductive elimination, which releases the desired product and regenerates the active palladium(0) catalyst to continue the cycle. This mechanistic pathway is highly robust, demonstrating excellent tolerance for a wide array of substituents on both the amine and the benzyl chloride components, including electron-donating methoxy groups and electron-withdrawing halogens. From an impurity control perspective, the specificity of the palladium coordination sphere minimizes side reactions such as homocoupling or over-carbonylation, ensuring that the crude reaction mixture is clean and amenable to straightforward purification techniques like column chromatography.
How to Synthesize 1,5-Dihydro-2H-Pyrrole-2-One Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize the efficiency of the palladium catalytic cycle. The protocol dictates the use of acetonitrile as the preferred organic solvent due to its ability to dissolve both polar and non-polar reactants while stabilizing the charged intermediates formed during the catalytic turnover. Operators must maintain a strict molar ratio of catalyst to ligand to base, typically employing palladium acetate and DPPF in conjunction with triethylamine to ensure optimal turnover numbers. While the general procedure is robust, attention to detail regarding the quality of the CO surrogate and the exclusion of oxygen is paramount for achieving the high yields reported in the patent literature, which can reach up to 92% for optimized substrates.
- Combine palladium acetate, DPPF ligand, triethylamine, TFBen (CO source), propargylamine, and benzyl chloride in acetonitrile.
- Heat the reaction mixture to 110°C and stir for 24 hours under inert atmosphere to facilitate oxidative addition and carbonyl insertion.
- Filter the mixture, adsorb onto silica gel, and purify via column chromatography to isolate the target pyrrol-2-one compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers tangible strategic benefits that extend beyond mere chemical elegance. The shift towards a one-pot methodology fundamentally alters the cost structure of manufacturing these valuable intermediates by collapsing multiple production steps into a single reactor charge. This consolidation not only reduces the capital expenditure required for additional processing equipment but also significantly lowers the operational overhead associated with material handling, quality control testing between steps, and solvent recovery. By streamlining the production workflow, manufacturers can achieve a drastic simplification of the supply chain, reducing the risk of bottlenecks that often plague multi-step synthetic campaigns.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the utilization of commodity chemicals such as benzyl chlorides and propargylamines, which are abundantly available from global chemical suppliers at competitive price points. Furthermore, the elimination of gaseous carbon monoxide removes the need for specialized high-pressure reactors and extensive safety infrastructure, leading to substantial capital savings and lower insurance premiums for production facilities. The high atom economy of the bis-carbonylation reaction ensures that raw material costs are minimized, as a greater proportion of the input mass is converted into the final valuable product rather than being lost as waste byproducts.
- Enhanced Supply Chain Reliability: Sourcing stability is greatly improved because the key reagents, including the palladium catalyst and the solid CO surrogate, are commercially available off-the-shelf items with established global supply networks. Unlike processes that rely on custom-synthesized exotic reagents or unstable intermediates that require immediate use, this method allows for the stockpiling of raw materials, thereby buffering the production schedule against market volatility or logistical disruptions. The robustness of the reaction conditions also means that the process is less susceptible to minor variations in raw material quality, ensuring consistent output even when sourcing from secondary vendors.
- Scalability and Environmental Compliance: The transition from laboratory scale to commercial production is facilitated by the use of standard heating and stirring equipment, avoiding the engineering challenges associated with handling toxic gases or cryogenic conditions. From an environmental standpoint, the use of a solid CO source significantly reduces the carbon footprint and safety risks associated with traditional carbonylation, aligning with increasingly stringent global regulations on industrial emissions and workplace safety. The simplified workup procedure, which often involves basic filtration and chromatography, reduces the volume of aqueous waste streams, lowering the costs and complexities associated with wastewater treatment and disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed carbonylation technology. These insights are derived directly from the experimental data and scope limitations defined within the patent documentation, providing a realistic overview of what partners can expect during technology transfer. Understanding these nuances is critical for R&D teams planning pilot runs and for procurement officers evaluating the long-term feasibility of this supply route.
Q: What are the key advantages of this carbonylation method over traditional cyclization?
A: This method utilizes a palladium-catalyzed bis-carbonylation strategy that operates in a single pot, eliminating the need for pre-functionalized acid chlorides or harsh cyclization conditions often required in conventional routes.
Q: Is the carbon monoxide source safe for large-scale manufacturing?
A: Yes, the process employs 1,3,5-trimethoxybenzene-derived phenol esters (TFBen) as a solid carbon monoxide surrogate, significantly enhancing operational safety compared to handling gaseous CO cylinders.
Q: What is the substrate scope for the benzyl chloride component?
A: The reaction demonstrates excellent functional group tolerance, successfully accommodating electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups such as fluoro, chloro, bromo, and trifluoromethyl.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-Dihydro-2H-Pyrrole-2-One Supplier
At NINGBO INNO PHARMCHEM, we recognize the pivotal role that high-quality heterocyclic intermediates play in the development of life-saving medications and advanced agrochemicals. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to full-scale manufacturing is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 1,5-dihydro-2H-pyrrole-2-one meets the exacting standards required by top-tier pharmaceutical companies worldwide.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this streamlined process. We encourage you to contact us today to obtain specific COA data for our catalog compounds or to initiate a dialogue regarding route feasibility assessments for your proprietary targets, ensuring a secure and cost-effective supply chain for your future projects.
