Advanced Platinum Carborane Complexes for Scalable Pharmaceutical Intermediate Production
Advanced Platinum Carborane Complexes for Scalable Pharmaceutical Intermediate Production
The chemical industry is constantly seeking more efficient pathways to synthesize high-value intermediates, and patent CN112661794B introduces a groundbreaking advancement in this domain. This patent discloses a novel platinum complex containing a diphosphine meta-carborane ligand, which serves as a highly effective catalyst for the direct coupling of phenolic compounds with ethyl acrylate. The significance of this technology lies in its ability to produce ortho-alkenyl phenols, which are critical building blocks for a wide array of natural products and active pharmaceutical ingredients. Unlike traditional methods that often struggle with selectivity and harsh conditions, this new catalytic system operates under remarkably mild parameters while maintaining high activity. For R&D directors and procurement managers alike, this represents a shift towards more sustainable and cost-effective manufacturing processes that do not compromise on purity or yield. The stability of the complex against air and water further simplifies the logistical challenges associated with handling sensitive organometallic reagents in a production environment.
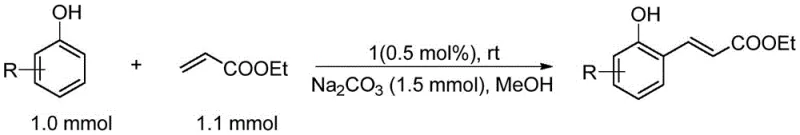
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ortho-alkenyl phenols has relied heavily on the Claisen rearrangement of alpha-allyl phenols, a process fraught with inherent inefficiencies and limitations for large-scale operations. The primary drawback of this traditional approach is the difficulty in controlling reaction selectivity, which frequently leads to the formation of undesirable multisite substitution products that complicate downstream purification. Furthermore, many modern alternatives require the installation of directing groups that are not only expensive and difficult to synthesize but also necessitate additional synthetic steps for their subsequent removal. These extra steps inevitably increase the overall material cost and extend the production lead time, creating bottlenecks in the supply chain for high-purity pharmaceutical intermediates. Additionally, conventional catalytic systems often demand high temperatures or inert atmospheres, which escalate energy consumption and require specialized equipment that increases capital expenditure. The instability of many existing catalysts also poses significant safety risks and storage challenges, making them less attractive for continuous manufacturing processes.
The Novel Approach
In stark contrast to these legacy methods, the platinum complex described in patent CN112661794B offers a streamlined and robust solution that directly addresses the pain points of modern chemical manufacturing. This novel approach utilizes a unique bisphosphine meta-carborane ligand that stabilizes the platinum center, enabling the direct ortho-alkenylation of phenols without the need for cumbersome directing groups. The reaction proceeds under mild conditions, often at room temperature, which drastically reduces energy requirements and minimizes the thermal degradation of sensitive substrates. The catalyst demonstrates a broad substrate scope, accommodating various electronic and steric substituents on the phenol ring, which provides flexibility for synthesizing diverse chemical libraries. Moreover, the high catalytic activity allows for significantly lower catalyst loading, which directly translates to reduced metal contamination in the final product and lower raw material costs. This method effectively eliminates the need for high-temperature rearrangements, thereby simplifying the process flow and enhancing the overall safety profile of the manufacturing facility.
Mechanistic Insights into Pt-Carborane Pincer Catalysis
The exceptional performance of this catalytic system can be attributed to the unique electronic and steric properties imparted by the meta-carborane cage within the pincer ligand structure. The carborane unit acts as a robust scaffold that enforces a rigid geometry around the platinum center, preventing the catalyst from decomposing into inactive species during the reaction cycle. This structural integrity is crucial for maintaining high turnover numbers over extended periods, ensuring consistent reaction rates even in complex reaction mixtures. The electron-withdrawing nature of the carborane cage also modulates the electron density at the metal center, facilitating the oxidative addition and reductive elimination steps that are critical for the coupling mechanism. For technical teams, understanding this mechanistic advantage is key to optimizing reaction parameters for specific substrates, as the catalyst's stability allows for fine-tuning without the risk of rapid deactivation. The ability of the ligand to withstand harsh chemical environments means that the catalyst can be recycled or used in continuous flow systems, further enhancing process efficiency.
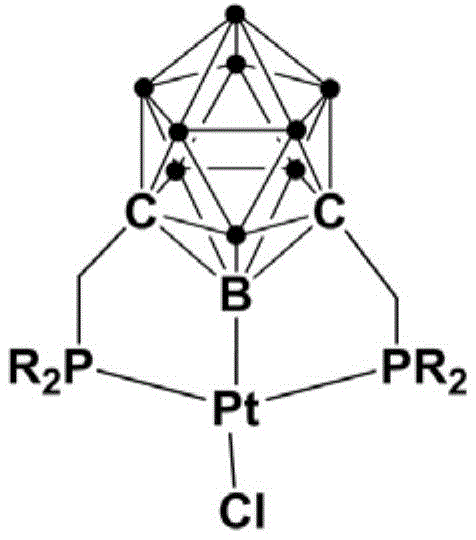
Impurity control is another critical aspect where this mechanistic design excels, particularly for the production of high-purity pharmaceutical intermediates where regulatory standards are stringent. The rigid pincer structure restricts the conformational freedom of the substrate during the coordination phase, thereby enforcing high regioselectivity for the ortho-position. This geometric constraint significantly suppresses the formation of para-substituted byproducts or polysubstituted impurities that are common in less selective catalytic systems. By minimizing the generation of side products at the molecular level, the need for extensive chromatographic purification is reduced, which lowers solvent consumption and waste generation. The stability of the catalyst also prevents the leaching of platinum into the product stream, ensuring that the final API intermediate meets strict heavy metal residue specifications. This level of control over the impurity profile is essential for reducing the burden on quality control laboratories and accelerating the release of batches for clinical or commercial use.
How to Synthesize Platinum Complex Efficiently
The preparation of this high-performance catalyst is designed to be straightforward and scalable, utilizing readily available starting materials to ensure a reliable supply chain for the catalyst itself. The synthesis involves a sequential one-pot reaction strategy that minimizes the need for intermediate isolation, thereby reducing processing time and solvent usage. Initially, meta-carborane is lithiated at low temperatures to activate the cage structure, followed by the introduction of a halogenated phosphine to construct the ligand framework. This ligand is then immediately coordinated with a platinum precursor in the same reaction vessel, streamlining the workflow and reducing the potential for material loss. The resulting complex is air-stable, which simplifies the workup procedure as it does not require rigorous exclusion of oxygen or moisture during filtration and drying. This ease of synthesis makes it feasible for chemical manufacturers to produce the catalyst in-house or source it reliably from specialized suppliers without facing significant logistical hurdles.
- React n-BuLi with meta-carborane at low temperature to generate the lithiated intermediate.
- Add halogenated phosphine to the reaction mixture and allow it to react at room temperature to form the ligand.
- Introduce (COD)PtCl2 to the system and heat to complete the coordination, followed by purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this catalytic technology offers substantial strategic advantages for procurement managers and supply chain heads looking to optimize their manufacturing costs and reliability. The ability to operate under mild conditions means that existing reactor infrastructure can often be utilized without the need for expensive upgrades to high-pressure or high-temperature systems. This compatibility with standard equipment significantly lowers the barrier to entry for implementing this new process, allowing for a faster return on investment. Furthermore, the use of cheap and readily available raw materials, such as ethyl acrylate and simple phenol derivatives, ensures that the cost of goods sold remains competitive even at large production volumes. The robustness of the catalyst also reduces the frequency of reactor downtime associated with catalyst replacement or system cleaning, thereby maximizing asset utilization rates. These factors combine to create a more resilient supply chain that is less susceptible to fluctuations in raw material prices or equipment availability.
- Cost Reduction in Manufacturing: The implementation of this platinum complex leads to significant cost savings primarily through the drastic reduction in catalyst loading required to achieve high yields. Unlike traditional systems that may require stoichiometric or near-stoichiometric amounts of reagents, this catalyst operates effectively at very low concentrations, minimizing the expenditure on precious metals. Additionally, the elimination of directing group synthesis and removal steps removes entire stages from the production workflow, which reduces labor costs and solvent consumption. The high selectivity of the reaction also means that less raw material is wasted on forming byproducts, improving the overall atom economy of the process. These cumulative efficiencies result in a lower cost per kilogram of the final intermediate, providing a competitive edge in the global market for fine chemical intermediates.
- Enhanced Supply Chain Reliability: The air and moisture stability of the catalyst fundamentally changes the logistics of storing and transporting this critical reagent. There is no longer a need for specialized cold chain shipping or inert atmosphere storage facilities, which simplifies inventory management and reduces the risk of supply disruptions due to handling errors. The broad substrate scope of the catalyst also means that a single catalytic system can be used to produce a wide variety of intermediates, reducing the need to stock multiple specialized catalysts. This versatility allows procurement teams to consolidate their supplier base and negotiate better terms for bulk purchases of the catalyst precursor. The reliability of the supply is further bolstered by the simplicity of the catalyst synthesis, which ensures that production can be ramped up quickly to meet surges in demand without long lead times.
- Scalability and Environmental Compliance: Scaling this process from the laboratory to commercial production is facilitated by the mild reaction conditions and the use of common solvents like methanol and ether. The absence of extreme temperatures or pressures reduces the safety risks associated with large-scale exothermic reactions, making it easier to obtain regulatory approval for new manufacturing lines. The high yield and selectivity also contribute to a greener process by minimizing the generation of chemical waste that requires treatment or disposal. This alignment with environmental compliance standards is increasingly important for multinational corporations that are under pressure to reduce their carbon footprint. The ability to run the reaction at room temperature also lowers the energy consumption of the facility, contributing to overall sustainability goals and potentially qualifying the process for green manufacturing incentives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this platinum carborane catalyst in industrial settings. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation to ensure accuracy. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this technology into their existing production pipelines. The information provided covers aspects of catalyst stability, reaction scope, and purification requirements to give a comprehensive overview of the operational expectations. This transparency helps in making informed decisions regarding process adoption and resource allocation for new product development.
Q: What are the stability advantages of this platinum carborane catalyst?
A: The catalyst exhibits exceptional stability against air and moisture, allowing for easier handling and storage compared to traditional sensitive organometallic catalysts.
Q: Can this catalyst be used for large-scale pharmaceutical intermediate production?
A: Yes, the mild reaction conditions and high turnover number make it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Q: How does this method improve selectivity in ortho-alkenylation?
A: The unique pincer ligand structure enforces strict geometric control, significantly reducing multisite substitution byproducts common in conventional methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Platinum Complex Containing Diphosphine Meta-Carborane Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this advanced catalytic technology for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab to market is seamless. Our facilities are equipped with stringent purity specifications and rigorous QC labs to guarantee that every batch of catalyst or intermediate meets the highest international standards. We understand the critical nature of supply chain continuity and are committed to providing a stable and reliable source of these specialized chemical solutions. Our technical team is ready to assist you in optimizing this process for your specific manufacturing needs, ensuring maximum efficiency and yield.
We invite you to collaborate with us to leverage this innovative chemistry for your next project and achieve significant operational improvements. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your current production volumes. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how this technology can enhance your competitive position. By partnering with us, you gain access to not just a product, but a comprehensive support system dedicated to your long-term success in the fine chemical industry.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
