Advanced Palladium-Catalyzed Carbonylation for Scalable 4-Acyl-2(5H)-Furanone Production
Introduction to Next-Generation Furanone Synthesis
The landscape of organic synthesis for bioactive heterocycles is constantly evolving, driven by the need for more efficient and sustainable manufacturing processes. A significant breakthrough in this domain is detailed in patent CN112239436A, which discloses a novel preparation method for 4-acyl-2(5H)-furanone compounds. This specific structural backbone is of immense value to the pharmaceutical industry, serving as a core motif in various natural products and bioactive molecules, including antimicrobial agents and analgesics. The traditional routes to access these complex scaffolds often suffer from multi-step sequences, harsh reaction conditions, and poor atom economy. However, the methodology outlined in this patent introduces a streamlined, palladium-catalyzed bis-carbonylation strategy that fundamentally shifts the paradigm. By utilizing readily available propiolic alcohols and aryl trifluoromethanesulfonates as starting materials, this process achieves the construction of the furanone ring and the installation of the acyl group in a single operational step. This represents a critical advancement for a reliable pharmaceutical intermediate supplier aiming to optimize their portfolio with high-value, bioactive precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-acyl-2(5H)-furanone derivatives has been a challenging endeavor for process chemists. Conventional methodologies often rely on the use of gaseous carbon monoxide under high pressure, which necessitates specialized autoclaves and rigorous safety protocols to prevent leakage and exposure. Furthermore, many existing routes require pre-functionalized substrates that are expensive and difficult to source, leading to inflated raw material costs. The multi-step nature of these traditional pathways not only extends the overall production timeline but also results in cumulative yield losses at each stage, significantly impacting the final throughput. Additionally, the purification of intermediates in these older methods can be cumbersome, often generating substantial amounts of chemical waste that complicate environmental compliance. These factors collectively create a bottleneck in the commercial scale-up of complex pharmaceutical intermediates, limiting the ability of manufacturers to respond quickly to market demands for new drug candidates containing this privileged scaffold.
The Novel Approach
In stark contrast to these legacy issues, the invention described in CN112239436A offers a robust and elegant solution through a palladium-catalyzed domino reaction. The core innovation lies in the use of chromium hexacarbonyl as a safe and effective solid carbon monoxide surrogate, which releases CO in situ under the reaction conditions. This eliminates the need for high-pressure gas equipment, drastically simplifying the reactor setup and enhancing operational safety. The reaction proceeds by coupling a propiolic alcohol with an aryl triflate in the presence of a palladium catalyst and a bidentate phosphine ligand. As illustrated in the general reaction scheme below, this one-pot transformation efficiently constructs the target molecule with high precision.
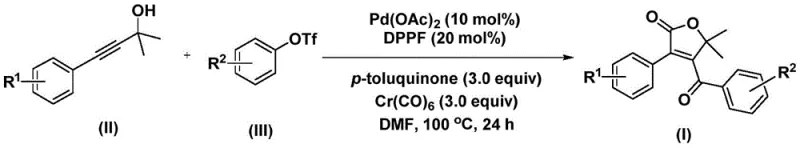
The versatility of this approach is further highlighted by its broad substrate scope, accommodating a wide range of substituents on both the alkyne and the aryl triflate components. This flexibility allows for the rapid generation of diverse libraries of furanone derivatives, facilitating structure-activity relationship (SAR) studies during drug discovery. Moreover, the reaction conditions are relatively mild, typically conducted at 100 °C in polar aprotic solvents like DMF, which ensures compatibility with sensitive functional groups. This novel pathway not only accelerates the synthesis timeline but also aligns with green chemistry principles by improving atom economy and reducing the overall environmental footprint of the manufacturing process.
Mechanistic Insights into Palladium-Catalyzed Bis-Carbonylation
Understanding the mechanistic underpinnings of this transformation is crucial for R&D directors focused on process optimization and impurity control. The catalytic cycle initiates with the oxidative addition of the aryl triflate to the active palladium(0) species, generated in situ from palladium acetate and the DPPF ligand. This forms an aryl-palladium(II) intermediate, which is the entry point for the carbonylation sequence. Subsequently, carbon monoxide, liberated from the chromium hexacarbonyl additive, inserts into the palladium-aryl bond to generate an acyl-palladium species. This acyl intermediate then undergoes migratory insertion with the triple bond of the propiolic alcohol, forming a five-membered vinyl-palladium ring. A second molecule of carbon monoxide inserts into this new palladium-carbon bond, expanding the coordination sphere and setting the stage for ring closure. Finally, reductive elimination occurs, releasing the 4-acyl-2(5H)-furanone product and regenerating the palladium(0) catalyst to continue the cycle. This intricate dance of insertion and elimination steps is meticulously balanced by the choice of ligands and additives.
Impurity control is a paramount concern in the synthesis of pharmaceutical intermediates, and this mechanism offers inherent advantages in that regard. The use of the bulky DPPF ligand helps to stabilize the palladium intermediates and prevents undesirable side reactions such as homocoupling of the aryl triflate or polymerization of the alkyne. Furthermore, the inclusion of p-toluquinone as an additive plays a critical role in re-oxidizing any palladium black that may form, thereby maintaining the catalytic activity throughout the prolonged reaction time of 24 hours. The high regioselectivity observed in the formation of the furanone ring minimizes the generation of isomeric byproducts, simplifying the downstream purification process. As demonstrated by the successful synthesis of various derivatives shown below, the system tolerates electron-rich and electron-deficient groups alike, indicating a robust catalytic cycle that is resistant to electronic perturbations.
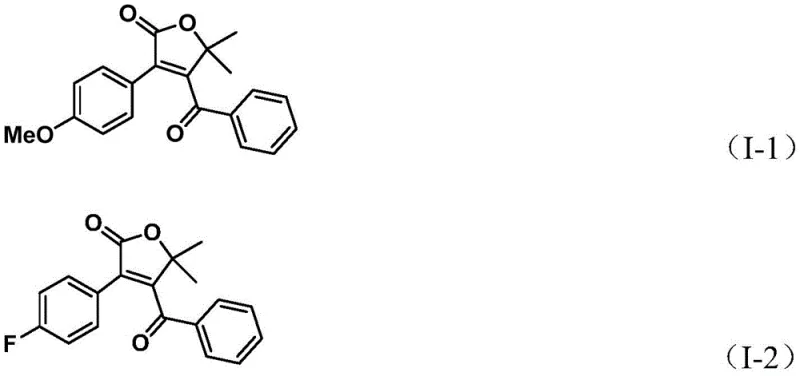
How to Synthesize 4-Acyl-2(5H)-Furanone Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and reaction parameters to ensure maximum yield and purity. The protocol is designed to be user-friendly, utilizing standard Schlenk techniques that are common in organic synthesis laboratories. The key to success lies in the precise ratio of the palladium catalyst to the ligand and the sufficient loading of the carbon monoxide source to drive the bis-carbonylation to completion. Detailed standardized operating procedures for this transformation are essential for reproducibility and quality control.
- Combine palladium acetate, DPPF ligand, chromium hexacarbonyl, p-toluquinone, propiolic alcohol, and aryl triflate in DMF solvent.
- Heat the reaction mixture to 100 °C and stir for 24 hours under inert atmosphere to facilitate bis-carbonylation.
- Filter the mixture, adsorb onto silica gel, and purify via column chromatography to isolate the target 4-acyl-2(5H)-furanone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible strategic benefits that go beyond mere chemical efficiency. The shift towards this novel synthetic route addresses several critical pain points associated with the sourcing and manufacturing of complex heterocyclic intermediates. By streamlining the production process, companies can achieve significant operational improvements that enhance overall competitiveness in the global market.
- Cost Reduction in Manufacturing: The economic implications of this method are profound, primarily driven by the simplification of the synthetic route. By consolidating multiple steps into a single pot, the process eliminates the need for intermediate isolation and purification, which are often the most cost-intensive phases of chemical manufacturing. Furthermore, the use of chromium hexacarbonyl as a CO source avoids the capital expenditure associated with high-pressure gas infrastructure and the ongoing costs of safety monitoring. The starting materials, propiolic alcohols and aryl triflates, are commercially available and can be sourced from a wide network of suppliers, fostering price competition and ensuring cost reduction in pharmaceutical intermediate manufacturing. The high yields reported, often exceeding 80% and reaching up to 95% for certain substrates, mean that less raw material is wasted, directly improving the cost of goods sold (COGS).
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness and flexibility of this chemical platform. The tolerance for diverse functional groups means that a single manufacturing line can be adapted to produce a wide variety of furanone derivatives simply by switching the starting aryl triflate or alkyne. This agility allows manufacturers to respond rapidly to changing customer specifications without the need for extensive process re-validation. Additionally, the reliance on stable, solid reagents rather than hazardous gases reduces the risk of supply disruptions caused by transportation regulations or storage limitations. This reliability ensures a consistent flow of high-quality intermediates, reducing lead time for high-purity pharmaceutical intermediates and enabling faster time-to-market for downstream drug products.
- Scalability and Environmental Compliance: From a sustainability perspective, this process offers a cleaner alternative to traditional carbonylation methods. The absence of high-pressure CO gas significantly lowers the safety risk profile, making it easier to obtain regulatory approvals for larger-scale production facilities. The simplified workup procedure, involving filtration and standard chromatography, generates less solvent waste compared to multi-step extractions and crystallizations. This alignment with green chemistry principles not only reduces waste disposal costs but also enhances the company's environmental, social, and governance (ESG) rating. The proven scalability of the reaction conditions, demonstrated effectively at the gram scale in the patent examples, provides a clear pathway for commercial scale-up to kilogram and ton quantities, ensuring long-term supply continuity for major pharmaceutical partners.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial aspects of this synthesis method, we have compiled answers to common questions based on the patent data. These insights are intended to assist technical teams in evaluating the feasibility of integrating this process into their existing workflows. Understanding the nuances of the reaction conditions and substrate scope is vital for successful implementation.
Q: What is the primary advantage of using Chromium Hexacarbonyl in this synthesis?
A: Chromium hexacarbonyl serves as a solid, easy-to-handle carbon monoxide substitute, eliminating the safety hazards and specialized equipment required for handling high-pressure CO gas in traditional carbonylation reactions.
Q: Can this method tolerate diverse functional groups on the aromatic ring?
A: Yes, the protocol demonstrates excellent substrate compatibility, successfully accommodating electron-donating groups like methoxy and alkyl chains, as well as electron-withdrawing groups such as halogens and trifluoromethyl groups.
Q: Is this process suitable for large-scale manufacturing?
A: The method utilizes commercially available catalysts and simple post-treatment procedures like filtration and chromatography, making it highly amenable to scale-up for industrial production of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Acyl-2(5H)-Furanone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methods like the one described in CN112239436A for the production of high-value pharmaceutical intermediates. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis of novel furanone derivatives or large-scale supply of established intermediates, our infrastructure is designed to support your most demanding projects with precision and reliability.
We invite you to explore how our expertise in palladium-catalyzed carbonylation can optimize your supply chain and reduce your overall manufacturing costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how we can become your trusted partner in bringing next-generation therapeutics to market efficiently and sustainably.
