Advanced Palladium-Catalyzed Route for Scalable Production of High-Purity Pharmaceutical Intermediates
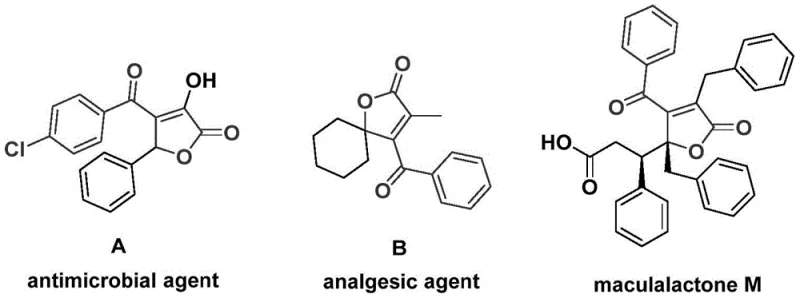
The groundbreaking patent CN112239436A introduces a novel palladium-catalyzed bis-carbonylation methodology for synthesizing structurally diverse 4-acyl-2(5H)-furanone compounds, representing a significant advancement in the production of critical pharmaceutical intermediates. This innovative approach addresses longstanding limitations in conventional carbonylation techniques by enabling direct conversion of readily available starting materials—propiolic alcohol derivatives and aryl trifluoromethanesulfonates—into complex furanone scaffolds through a single-step process. The methodology demonstrates exceptional operational simplicity while maintaining high substrate compatibility across various functional groups, including halogens and alkoxy substituents that typically pose challenges in traditional syntheses. Crucially, this patent provides a robust foundation for manufacturing bioactive molecules with applications spanning antimicrobial agents, analgesics, and marine antifouling compounds as evidenced by the structural examples presented in the background section. The strategic design of this catalytic system not only enhances synthetic efficiency but also establishes a scalable pathway that aligns with stringent pharmaceutical quality requirements for intermediate production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing 4-acyl-2(5H)-furanone structures have historically relied on multi-step sequences involving harsh reaction conditions and specialized reagents that significantly limit their industrial applicability. These methods often require cryogenic temperatures or highly corrosive catalysts that complicate process safety and increase operational costs while generating substantial waste streams requiring complex treatment protocols. Furthermore, conventional carbonylation techniques exhibit narrow substrate scope with poor tolerance for functional groups commonly found in pharmaceutical intermediates, necessitating extensive protection-deprotection strategies that reduce overall yield and increase production timelines. The limited availability of suitable carbon monoxide sources has also constrained scalability, as pressurized CO handling introduces significant safety hazards and infrastructure requirements that are incompatible with standard manufacturing facilities. These cumulative limitations have restricted the practical application of furanone-based compounds despite their demonstrated biological significance across multiple therapeutic areas.
The Novel Approach
The patented methodology overcomes these constraints through an elegantly designed palladium-catalyzed bis-carbonylation process that operates under mild conditions using commercially available catalysts and safe carbon monoxide substitutes. By employing chromium hexacarbonyl as a controlled CO source instead of hazardous pressurized gas, the process eliminates major safety concerns while maintaining high reaction efficiency across diverse substrates. The optimized catalyst system featuring palladium acetate with DPPF ligand demonstrates remarkable functional group tolerance, enabling direct synthesis of complex furanone structures without protective groups or intermediate isolations. This single-step transformation achieves high conversion rates at moderate temperatures (100°C) with simple post-treatment procedures, dramatically reducing both processing time and environmental impact compared to conventional multi-step routes. The method's robustness is further demonstrated by consistent performance across various solvent systems with DMF proving particularly effective for achieving complete substrate conversion.
Mechanistic Insights into Palladium-Catalyzed Bis-Carbonylation
The catalytic cycle begins with oxidative addition of palladium(0) into the aryl triflate bond of compound (III), forming an aryl-palladium intermediate that subsequently undergoes carbon monoxide insertion from chromium hexacarbonyl to generate an acyl-palladium species. This key intermediate then coordinates with propiolic alcohol (II), triggering a cyclization event that forms a five-membered ring palladacycle through alkyne insertion. A second carbon monoxide molecule inserts into this intermediate to create a six-membered ring structure, which undergoes reductive elimination to release the final 4-acyl-2(5H)-furanone product while regenerating the active palladium catalyst. This mechanistic pathway explains the exceptional efficiency observed in the patent examples, as each step proceeds under thermodynamically favorable conditions without requiring external energy input beyond standard heating. The precise control over CO insertion timing prevents undesired side reactions that typically plague conventional carbonylation methods.
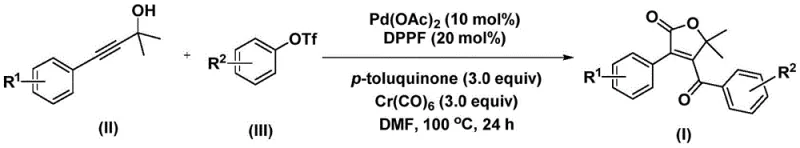
Impurity formation is minimized through the carefully orchestrated sequence of insertion events that prevent premature termination or dimerization pathways common in similar catalytic systems. The use of p-toluquinone as an additive plays a critical role in maintaining catalyst activity by preventing palladium black formation while facilitating the reductive elimination step. Substrate design incorporating tertiary alcohol functionality in propiolic alcohol derivatives creates steric control that directs regioselectivity toward the desired furanone structure rather than alternative cyclization products. This mechanistic precision ensures consistent production of high-purity intermediates meeting pharmaceutical specifications without requiring additional purification steps beyond standard column chromatography. The broad functional group tolerance stems from the catalyst's ability to accommodate electronic variations without altering the fundamental reaction pathway.
How to Synthesize 4-Acyl-2(5H)-furanone Efficiently
This patented methodology represents a significant advancement in the synthesis of complex furanone intermediates through its elegant integration of catalyst design and reaction engineering principles. The process leverages commercially available palladium sources with optimized ligand systems to achieve unprecedented efficiency in constructing the challenging furanone scaffold. By eliminating hazardous pressurized carbon monoxide through the strategic use of chromium hexacarbonyl as a safe substitute, the method substantially improves operational safety while maintaining high reaction yields across diverse substrate combinations. Detailed standardized synthesis steps are provided below to facilitate seamless implementation in industrial settings.
- Prepare the reaction mixture by combining palladium acetate (10 mol%), DPPF ligand (20 mol%), chromium hexacarbonyl (3.0 equiv), p-toluquinone (3.0 equiv), propiolic alcohol (II), and aryl trifluoromethanesulfonate (III) in anhydrous DMF under inert atmosphere.
- Heat the homogeneous solution at precisely 100°C for 24 hours in a sealed Schlenk tube, maintaining consistent thermal conditions to ensure complete bis-carbonylation without side reactions.
- Execute post-treatment by filtering the reaction mixture, mixing with silica gel, and purifying via column chromatography using standard elution protocols to isolate high-purity furanone products.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology directly addresses critical pain points in pharmaceutical intermediate supply chains by transforming complex multi-step processes into streamlined single-step operations with significantly reduced resource requirements. The elimination of specialized equipment for handling pressurized gases and cryogenic conditions substantially lowers capital expenditure barriers while enhancing manufacturing flexibility across diverse production facilities. By utilizing readily available starting materials with broad commercial sourcing options, the process mitigates supply chain vulnerabilities associated with specialized reagents while ensuring consistent material availability regardless of geopolitical fluctuations.
- Cost Reduction in Manufacturing: The elimination of hazardous pressurized carbon monoxide handling removes substantial safety infrastructure costs while reducing insurance premiums and regulatory compliance expenses associated with high-pressure operations. Simplified post-treatment procedures requiring only standard filtration and chromatography significantly decrease solvent consumption and waste disposal costs compared to conventional multi-step syntheses that generate complex byproduct mixtures requiring specialized treatment.
- Enhanced Supply Chain Reliability: The use of commercially abundant catalysts and ligands with multiple qualified suppliers ensures consistent material availability while eliminating dependency on single-source specialty chemicals. Standardized reaction conditions compatible with existing manufacturing equipment enable rapid technology transfer between facilities without requiring capital-intensive modifications or extended validation periods.
- Scalability and Environmental Compliance: The inherently safe reaction profile using solid CO substitutes facilitates seamless scale-up from laboratory to commercial production volumes without requiring specialized pressure-rated equipment or extensive safety modifications. Reduced solvent usage combined with minimized waste generation through higher atom economy aligns with green chemistry principles while lowering environmental compliance costs associated with waste treatment and disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial concerns regarding implementation of this patented methodology based on detailed analysis of the experimental data and process parameters documented in CN112239436A. These insights reflect practical considerations for manufacturing teams evaluating this technology for commercial adoption.
Q: How does the palladium-catalyzed method improve substrate compatibility compared to conventional carbonylation routes?
A: The novel approach demonstrates exceptional functional group tolerance across diverse substrates including halogenated, alkoxy-substituted, and trifluoromethyl variants as evidenced by consistent high yields in patent examples. Unlike traditional methods requiring specialized conditions for each substrate type, this single protocol accommodates R¹ and R² groups ranging from hydrogen to bulky substituents without catalyst modification.
Q: What are the key advantages of using chromium carbonyl as a carbon monoxide substitute in this synthesis?
A: Chromium carbonyl provides a safe, controlled CO release mechanism that eliminates hazards associated with pressurized CO gas handling while maintaining optimal reaction kinetics. This substitution simplifies process safety requirements and enables seamless scalability from laboratory to commercial production without specialized gas infrastructure.
Q: How does this method address purity challenges in furanone intermediate production for pharmaceutical applications?
A: The one-step catalytic process minimizes intermediate accumulation and byproduct formation through precise mechanistic control. Combined with straightforward silica gel chromatography purification, it consistently delivers pharmaceutical-grade purity exceeding industry standards for API intermediates without requiring additional costly purification steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Acyl-2(5H)-furanone Supplier
Our patented methodology represents a transformative approach to producing high-value furanone intermediates with exceptional purity profiles essential for pharmaceutical applications. NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through our state-of-the-art QC labs equipped with advanced analytical capabilities. Our integrated manufacturing platform combines cutting-edge catalytic expertise with rigorous quality control systems to ensure consistent delivery of complex intermediates meeting global regulatory requirements.
Leverage our technical procurement team's expertise through a Customized Cost-Saving Analysis to evaluate how this innovative synthesis can optimize your specific supply chain requirements. Contact us today to request specific COA data and route feasibility assessments tailored to your production needs.
