Advanced Chiral Aluminum Catalysis For Commercial Scale-Up Of Complex Flavor Intermediates
The chemical industry is constantly seeking more efficient pathways to produce high-value chiral intermediates, and patent CN102046572A presents a groundbreaking advancement in this domain. This intellectual property details a novel process for cyclizing compounds containing a formyl group and a double bond capable of undergoing carbonyl-ene cyclization, specifically targeting the production of optically active isopulegol from geranial. The core innovation lies in the utilization of specific chiral aluminum complexes that not only increase the proportion of a specific optical isomer in the cyclized compound but also enhance the optical purity of the unreacted starting material. This dual enhancement mechanism represents a significant leap forward for manufacturers of flavor and fragrance intermediates, as well as pharmaceutical producers requiring high-purity chiral building blocks. By leveraging these advanced catalytic systems, producers can achieve superior enantiomeric excess ratios without relying on expensive and wasteful resolution steps that have traditionally plagued the industry. The technology described offers a robust solution for scaling up the production of l-menthol precursors, addressing critical pain points related to yield, purity, and cost-efficiency in fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the selective cyclization of geranial to produce isopulegol has relied on reagents such as zinc bromide or other non-chiral Lewis acids, which often suffer from significant limitations in terms of stereocontrol. These conventional methods frequently result in mixtures of optical isomers, necessitating complex and costly downstream separation processes to isolate the desired enantiomer, such as l-isopulegol, which is a crucial precursor for l-menthol. The lack of inherent selectivity in traditional catalysts means that a substantial portion of the starting material is converted into unwanted isomers, leading to lower overall yields and increased waste generation. Furthermore, the use of stoichiometric amounts of certain reagents in older processes can drive up raw material costs and create environmental burdens due to the disposal of heavy metal waste. The inability to effectively upgrade the optical purity of the unreacted substrate also means that recycling streams are often contaminated, forcing manufacturers to discard valuable materials or invest in additional purification infrastructure. These inefficiencies create a bottleneck for companies aiming to produce high-purity flavor and fragrance intermediates at a competitive price point.
The Novel Approach
In stark contrast to these legacy methods, the novel approach outlined in the patent utilizes specially designed chiral aluminum complexes that act as highly selective catalysts for the carbonyl-ene cyclization reaction. By employing optically active biaryl diol compounds as ligands, the aluminum center is tuned to preferentially interact with one specific enantiomer of the geranial substrate, driving the formation of the desired isopulegol isomer with remarkable precision. This catalytic system is unique in its ability to perform kinetic resolution, meaning it not only produces the target product with high optical purity but also leaves the unreacted geranial enriched in the opposite enantiomer, which can be recycled or utilized elsewhere. The versatility of this method allows for the use of geranial with varying initial optical purities, including racemic mixtures, while still achieving high enantiomeric excess in the final product. This flexibility reduces the dependency on expensive, high-purity starting materials and simplifies the supply chain logistics for raw material procurement. Additionally, the catalyst loading can be kept relatively low, and the ligands can be recovered, further enhancing the economic viability of this process for large-scale industrial applications.
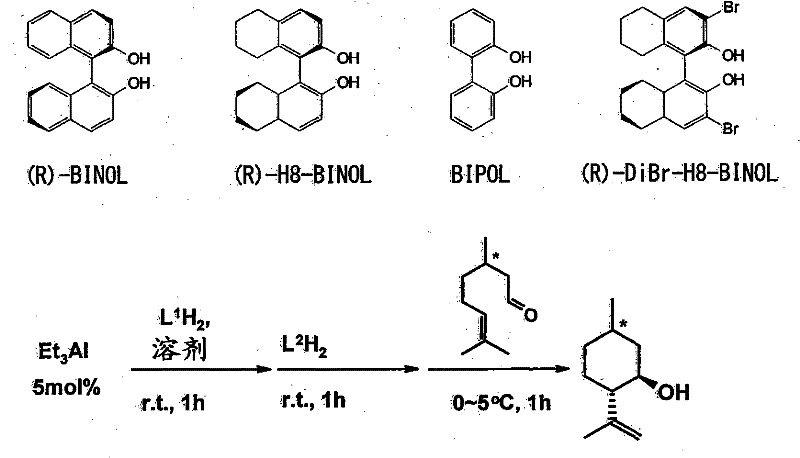
Mechanistic Insights into Chiral Aluminum-Catalyzed Cyclization
The mechanistic foundation of this technology rests on the precise coordination chemistry between the aluminum center and the chiral biaryl diol ligands, which creates a sterically constrained environment around the active site. When the geranial substrate approaches this chiral pocket, the specific spatial arrangement of the ligands dictates the orientation of the formyl group and the double bond, facilitating a highly stereoselective carbonyl-ene reaction. This interaction lowers the activation energy for the formation of the desired transition state while raising it for competing pathways that would lead to unwanted isomers. The aluminum complex effectively differentiates between the enantiomers of the substrate, binding more strongly or reacting faster with one form over the other, which is the essence of the kinetic resolution process observed in the experimental data. The use of biaryl diol ligands, such as BINOL derivatives, provides a rigid backbone that maintains the chiral information throughout the catalytic cycle, ensuring consistent performance across multiple batches. Understanding this mechanism is crucial for R&D teams looking to optimize reaction conditions, as factors like solvent polarity and temperature can influence the conformation of the catalyst-substrate complex and thus the final selectivity.
Impurity control is another critical aspect where this mechanistic understanding translates into practical benefits for manufacturing. The high selectivity of the aluminum complex minimizes the formation of by-products such as neoisopulegol or other structural isomers that are difficult to separate from the target l-isopulegol. By suppressing these side reactions at the molecular level, the process reduces the burden on downstream purification units like distillation columns or crystallization tanks. The patent data indicates that the ratio of the target isomer to other isomers can be exceptionally high, often exceeding 96:4, which simplifies the specification compliance for high-purity applications. Furthermore, the ability to recover the chiral ligand from the reaction mixture prevents the accumulation of aluminum residues or degraded ligand fragments in the product stream. This clean reaction profile is particularly important for pharmaceutical intermediates where strict limits on heavy metals and organic impurities must be met. The robustness of the catalyst system against minor variations in feed quality also contributes to a more stable impurity profile over time, enhancing the overall reliability of the production process.
How to Synthesize Optically Active Isopulegol Efficiently
The synthesis of optically active isopulegol using this patented technology involves a straightforward yet precise sequence of steps that can be adapted for both laboratory and commercial scale operations. The process begins with the in-situ generation of the active aluminum catalyst by mixing a standard aluminum compound, such as triethyl aluminum, with the chosen chiral biaryl diol ligand in an inert organic solvent. This preparation step must be conducted under anhydrous conditions to prevent deactivation of the catalyst, typically under a nitrogen or argon atmosphere. Once the catalyst is formed, the geranial substrate is introduced to the reaction mixture, and the temperature is carefully controlled to optimize the rate of cyclization while maintaining high stereoselectivity. The detailed standardized synthesis steps, including specific molar ratios, solvent choices, and workup procedures, are outlined in the guide below to ensure reproducibility and safety.
- Prepare the chiral aluminum catalyst by reacting an aluminum compound (e.g., triethyl aluminum) with an optically active biaryl diol compound (e.g., BINOL derivatives) in an inert solvent.
- Mix the prepared catalyst solution with geranial substrate under a nitrogen atmosphere, maintaining the temperature between -30°C and 50°C.
- Allow the carbonyl-ene cyclization to proceed, then purify the resulting isopulegol via distillation or crystallization to achieve high optical purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this aluminum complex technology offers substantial strategic advantages that go beyond mere technical performance. The primary benefit lies in the potential for significant cost reduction in manufacturing, driven by the elimination of expensive resolution steps and the ability to use lower-grade starting materials. By achieving high optical purity directly from the reaction, companies can bypass costly chiral separation processes that often consume a large portion of the production budget. Additionally, the recoverability of the chiral ligands means that the effective cost of the catalyst system is amortized over multiple batches, further driving down the unit cost of the final isopulegol product. This economic efficiency allows suppliers to offer more competitive pricing to downstream customers in the flavor, fragrance, and pharmaceutical sectors, strengthening their market position. The process also reduces the consumption of raw materials by minimizing waste, aligning with corporate sustainability goals and reducing the environmental footprint of the supply chain.
- Cost Reduction in Manufacturing: The implementation of this catalytic system drastically simplifies the production workflow by removing the need for stoichiometric chiral auxiliaries or extensive purification trains. Since the catalyst promotes high selectivity, the yield of the desired isomer is maximized, reducing the amount of raw material required per unit of product. The ability to recover and reuse the expensive chiral ligands further contributes to long-term savings, as the initial investment in the ligand is spread across a larger volume of production. Moreover, the milder reaction conditions compared to some traditional methods can lead to lower energy consumption for heating and cooling, adding another layer of operational cost efficiency. These factors combine to create a leaner manufacturing process that is less sensitive to fluctuations in raw material prices.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the robustness of this chemistry enhances the reliability of supply by reducing the risk of batch failures due to purity issues. The tolerance of the catalyst to varying qualities of geranial feedstock means that procurement teams have more flexibility in sourcing raw materials, potentially accessing a broader supplier base without compromising product quality. This flexibility mitigates the risk of supply disruptions caused by shortages of high-purity starting materials. Furthermore, the simplified process flow shortens the overall production cycle time, allowing for faster turnaround on customer orders and improved responsiveness to market demand. The consistency of the product quality also reduces the likelihood of customer rejections or returns, fostering stronger long-term relationships with key accounts.
- Scalability and Environmental Compliance: The scalability of this process is supported by the use of common industrial solvents and standard reaction equipment, making it easy to transition from pilot scale to full commercial production. The reduction in waste generation, particularly the avoidance of heavy metal waste associated with older catalysts, simplifies compliance with increasingly stringent environmental regulations. The ability to recycle the ligand and minimize solvent usage through efficient workup procedures contributes to a greener manufacturing profile, which is increasingly valued by global customers. This environmental advantage can also translate into regulatory benefits, such as faster approval times for new drug filings where the manufacturing process is scrutinized for impurity control. Overall, the process offers a sustainable path for scaling up the production of complex chiral intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral aluminum catalysis technology. These answers are derived directly from the experimental data and claims within the patent documentation to provide accurate guidance for potential adopters. Understanding these details is essential for evaluating the feasibility of integrating this process into existing manufacturing lines or for developing new product pipelines based on high-purity isopulegol. The insights provided here aim to clarify the operational parameters and benefits that distinguish this method from conventional alternatives.
Q: What is the primary advantage of using chiral aluminum complexes over traditional zinc bromide catalysts?
A: The primary advantage is the significantly enhanced enantioselectivity. While traditional methods often struggle with low optical purity, the novel aluminum complexes described in CN102046572A can achieve high selectivity for specific optical isomers (e.g., l-isopulegol) even from racemic or low-purity geranial starting materials, reducing the need for extensive downstream purification.
Q: Can the chiral ligands used in this aluminum complex be recovered and reused?
A: Yes, the patent explicitly details that the chiral biaryl diol ligands can be recovered from the reaction system after cyclization is finished. This recovery process allows the ligands to be reused as part of the aluminum catalyst, which substantially lowers the overall material cost and improves the environmental profile of the manufacturing process.
Q: What are the typical reaction conditions for this cyclization process?
A: The cyclization is typically carried out at temperatures ranging from -30°C to 50°C, with a preferred range of -10°C to 30°C. The reaction can be performed in various inert organic solvents such as toluene, hexane, or dichloromethane, and usually completes within 0.5 to 20 hours depending on the specific catalyst loading and substrate concentration.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isopulegol Supplier
The technological potential of this chiral aluminum catalysis route represents a significant opportunity for the fine chemical industry, and NINGBO INNO PHARMCHEM is well-positioned to help you capitalize on it. As a seasoned CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab to market is seamless and efficient. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of isopulegol or related intermediates meets the highest global standards. We understand the critical importance of consistency in chiral synthesis and have the technical expertise to optimize these aluminum complex systems for maximum yield and selectivity. Partnering with us means gaining access to a team that is dedicated to solving complex synthesis challenges while maintaining the highest levels of quality and safety.
We invite you to initiate a conversation about optimizing your supply chain for high-purity flavor and fragrance intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. By collaborating with us, you can request specific COA data and route feasibility assessments to verify the compatibility of this technology with your current operations. Let us help you reduce lead time for high-purity intermediates and secure a stable, cost-effective supply of critical building blocks for your business growth.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
