Advanced Manufacturing of LE14 Linker Drug Conjugates for Scalable ADC Development
The rapid evolution of Antibody-Drug Conjugates (ADCs) has placed immense pressure on the supply chain to deliver high-purity linker drug conjugates with consistent quality. Patent CN116178386A introduces a transformative preparation method for the linker drug conjugate LE14 and its critical intermediates, addressing long-standing stability and cost issues in ADC manufacturing. This technology specifically provides a robust preparation method for compound LE14 by condensing a stable intermediate of formula II with compound of formula III or its mesylate. The innovation lies in the ability to prepare high-purity intermediate II, which can be directly condensed with exatecan mesylate to obtain compound I, and subsequently processed into the final product LE14. By utilizing intermediate II as the starting material for process production, the method significantly reduces process steps, facilitates easier quality control, and drastically lowers impurity levels in the final product. Furthermore, the use of more economical exatecan for condensation greatly reduces production costs, making this route highly suitable for industrial-scale production and reliable supply chain integration for global pharmaceutical partners.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
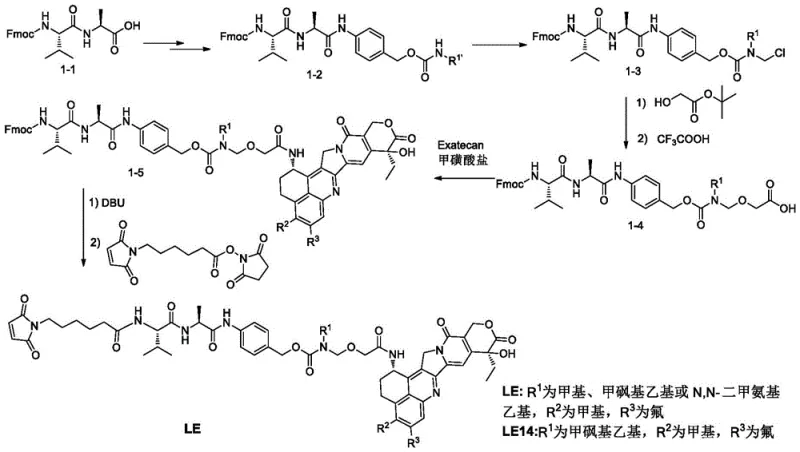
Conventional synthetic routes for ADC linkers often suffer from critical instability issues during the intermediate stages, particularly when dealing with carboxylic acid intermediates. In prior art routes, such as Route 1 and Route 2, the preparation of carboxylic acid intermediates requires the removal of tert-butyl protecting groups using trifluoroacetic acid. This deprotection step is notoriously severe and creates significant decomposition risks for both the raw materials and the resulting products. The generated intermediates, such as 1-4 and 2-2a, are highly unstable under acid and alkali conditions, leading to almost complete loss during purification processes. Consequently, manufacturers are forced to use crude products directly in subsequent reactions without purification. This compromise introduces obvious impurities with polarities similar to the final product, making purification extremely difficult and negatively impacting the overall quality and purity of the final LE14 compound. These technical bottlenecks limit the commercial viability and scalability of traditional ADC linker synthesis.
The Novel Approach
The novel approach disclosed in the patent overcomes these stability hurdles by introducing a silyl-based protecting group strategy that ensures the intermediate remains stable throughout the synthesis. Instead of relying on unstable tert-butyl protection, the new method generates a purified carboxylic acid intermediate II product that can be stably obtained and used as a starting material for process production. This stability allows for standard purification techniques, such as silica gel column chromatography, to be effectively employed, ensuring that impurities are removed before the final coupling steps. The process steps are obviously reduced compared to conventional methods, and the impurity content of the final product LE14 is significantly reduced. By avoiding the decomposition risks associated with harsh deprotection conditions, this method ensures a more robust and reliable manufacturing process that is capable of meeting the stringent quality requirements of modern ADC drug development and commercial supply.
Mechanistic Insights into Amide Condensation and Stabilization
The core of this technological breakthrough lies in the efficient amide condensation reaction between the stable intermediate of formula II and the compound of formula III or its mesylate. This reaction is carried out in the presence of a condensing agent, a base, and a solvent under controlled conditions to obtain the compound of formula I. The selection of condensing agents such as DMTMM or HATU, combined with organic bases like N,N-diisopropylethylamine, facilitates a high-yield coupling reaction at mild temperatures ranging from 20°C to 50°C. The molar ratios are carefully optimized, with the condensing agent typically used in a 1 to 1.5 molar ratio relative to the intermediate II. This precise control over reaction conditions minimizes side reactions and ensures that the amide bond is formed with high fidelity. The use of light-protected conditions and inert gas atmospheres further preserves the integrity of the sensitive functional groups within the molecule, preventing degradation and ensuring consistent batch quality.
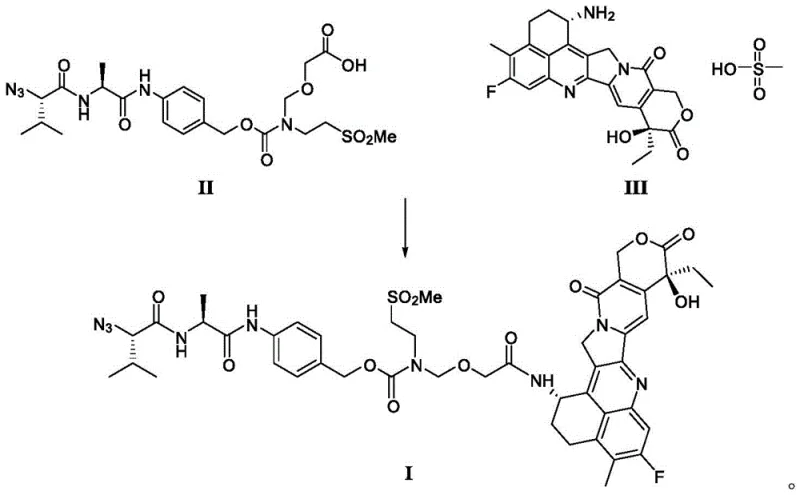
Impurity control is another critical aspect of this mechanistic design, achieved through the stability of the silyl-protected intermediate. Unlike prior art intermediates that decompose during purification, intermediate II can withstand the conditions of removing the protecting group using fluoride reagents such as tetrabutylammonium fluoride or potassium fluoride. This deprotection reaction is conducted at moderate temperatures between 40°C and 70°C, allowing for the stable existence of the carboxylic acid intermediate. The purified intermediate II is then utilized for the condensation reaction, which greatly improves the purity of the intermediate I and reduces the generation of impurities. By preventing the introduction of impurities into subsequent reaction steps, the purity of the final product LE14 is obviously improved, with the maximum single impurity reduced to below 0.1%. This level of impurity control is essential for meeting regulatory standards and ensuring the safety and efficacy of the final ADC therapeutic.
How to Synthesize LE14 Efficiently
The synthesis of LE14 efficiently requires a strategic approach that prioritizes intermediate stability and purification at every stage. The process begins with the preparation of the stable carboxylic acid intermediate II, which serves as the foundation for the entire synthetic route. Detailed standardized synthesis steps involve the deprotection of silyl-protected precursors followed by the critical amide condensation with exatecan mesylate. The operational background of this patent breakthrough allows for the use of commercially available raw materials and standard laboratory equipment, making it accessible for scale-up. The following guide outlines the critical phases of this synthesis, ensuring that technical teams can replicate the high-purity results demonstrated in the patent examples. For the complete standardized synthesis steps and specific reaction parameters, please refer to the detailed guide below.
- Prepare stable Intermediate II via deprotection of silyl-protected precursor using fluoride reagents.
- Perform amide condensation between Intermediate II and Exatecan mesylate using DMTMM and base.
- Complete the synthesis by reducing the azide group and coupling with maleimide linker to form LE14.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis route offers substantial commercial advantages for procurement and supply chain teams by addressing key pain points related to cost, reliability, and scalability. The traditional reliance on expensive DXd derivatives as payload sources has long been a bottleneck in ADC manufacturing, driving up costs and complicating supply chains. By utilizing more economical exatecan or its mesylate as a key raw material for condensation, this method greatly reduces the production cost associated with payload acquisition. Furthermore, the ability to obtain a purified stable carboxylic acid intermediate means that the process steps are simplified, reducing the overall manufacturing time and resource consumption. This simplification translates directly into enhanced supply chain reliability, as the risk of batch failure due to intermediate instability is significantly mitigated. Companies can expect a more predictable production schedule and reduced waste, contributing to a more sustainable and cost-effective manufacturing operation.
- Cost Reduction in Manufacturing: The elimination of expensive DXd derivatives in favor of economical exatecan mesylate results in substantial cost savings in raw material procurement. Additionally, the ability to purify intermediates before final coupling reduces the loss of high-value materials due to impurity-related failures. The streamlined process steps further lower operational costs by reducing solvent usage and processing time. These factors combine to create a significantly more cost-efficient manufacturing model compared to conventional routes that suffer from low yields and high material loss.
- Enhanced Supply Chain Reliability: The stability of intermediate II ensures that the supply chain is not disrupted by the decomposition of critical materials during storage or transport. Unlike unstable intermediates that require immediate use or special handling, this intermediate can be produced, purified, and stored with confidence. This reliability allows for better inventory management and reduces the risk of production delays caused by material quality issues. Procurement teams can secure raw materials with greater flexibility, knowing that the synthesis route is robust enough to handle variations in supply without compromising final product quality.
- Scalability and Environmental Compliance: The use of mild reaction conditions and standard purification techniques makes this route highly scalable for commercial production. The reduction in hazardous reagents and the ability to recycle solvents contribute to better environmental compliance and reduced waste disposal costs. The process is designed to be easily transferred from laboratory scale to industrial scale, ensuring that supply can meet growing market demand without the need for complex re-engineering. This scalability is crucial for supporting the long-term commercial success of ADC drugs utilizing the LE14 linker.
Frequently Asked Questions (FAQ)
The following frequently asked questions are based on the technical details and advantages outlined in the patent data. They address common concerns regarding the stability, cost, and purity of the LE14 synthesis route. These answers provide clarity on how this method compares to prior art and what benefits it offers for commercial manufacturing. Understanding these technical nuances is essential for making informed decisions about adopting this technology for your ADC development pipeline. The responses are derived directly from the experimental data and comparative analysis provided in the patent documentation.
Q: Why is Intermediate II considered superior to prior art intermediates?
A: Intermediate II utilizes a silyl-based protecting group strategy that allows for stable purification via column chromatography, unlike the unstable tert-butyl protected intermediates in conventional routes which decompose during purification.
Q: How does this method reduce production costs for ADC drugs?
A: The process utilizes economically available Exatecan mesylate directly for condensation, avoiding the need for expensive pre-derivatized DXd payloads, thereby significantly lowering raw material costs.
Q: What purity levels can be achieved with this synthetic route?
A: The optimized route enables the production of LE14 with purity exceeding 99.5% and maximum single impurity content below 0.1%, meeting stringent pharmaceutical standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable LE14 Supplier
The technical potential of this LE14 synthesis route is immense, offering a pathway to high-purity ADC linkers that meet the rigorous demands of the pharmaceutical industry. NINGBO INNO PHARMCHEM, as a CDMO expert, possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with stringent purity specifications and rigorous QC labs to ensure that every batch of LE14 or related intermediates meets the highest quality standards. We understand the complexities of ADC linker synthesis and have the capability to implement this novel route effectively, ensuring that your supply chain remains robust and compliant with global regulatory requirements.
We invite you to initiate a supply chain optimization inquiry to explore how this technology can benefit your specific projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your production volumes and quality needs. Please contact us to request specific COA data and route feasibility assessments that demonstrate the viability of this method for your commercial manufacturing goals. Partnering with us ensures access to cutting-edge synthesis technology and a reliable supply of critical ADC intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
