Advanced Telmisartan Intermediate Synthesis: Scalable Process Optimization for Global Pharma Supply Chains
The pharmaceutical industry continuously seeks robust manufacturing pathways for antihypertensive agents, particularly for blockbuster drugs like Telmisartan. A pivotal advancement in this domain is documented in patent CN103319414A, which outlines a significantly improved preparation process for Telmisartan and its key intermediate, 2-n-propyl-4-methyl-benzimidazole-6-carboxylic acid. This technology addresses longstanding inefficiencies in traditional synthesis routes by fundamentally restructuring the purification strategy. Instead of isolating and purifying every intermediate, the novel method employs a telescoped approach where crude products from nitration, reduction, and cyclization reactions are carried forward without separation. The target molecule is ultimately isolated by precisely adjusting the pH of the hydrolysis reaction mixture to between 6 and 7. This strategic modification not only simplifies the operational workflow but also ensures that the recrystallized crude Telmisartan achieves a purity exceeding 99%, meeting the stringent requirements of global regulatory bodies for active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for 2-n-propyl-4-methyl-benzimidazole-6-carboxylic acid have historically been plagued by cumbersome purification protocols that hinder industrial scalability. As illustrated in the reaction scheme below, the nitration of methyl 3-methyl-4-n-butyramide benzoate typically yields a mixture containing the desired meta-nitro product alongside significant amounts of ortho- and para-nitro isomers. 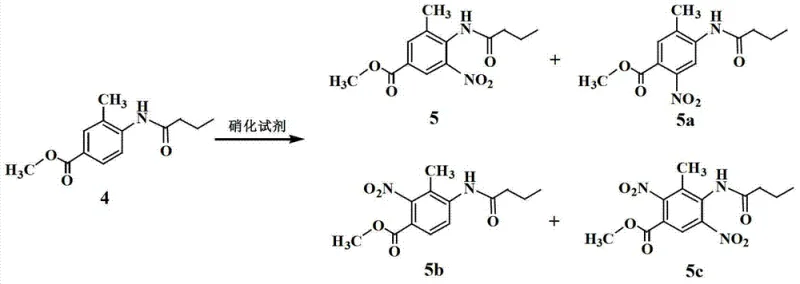 Separating these positional isomers is notoriously difficult due to their similar physicochemical properties, often necessitating multiple recrystallization steps or expensive column chromatography. Literature indicates that each recrystallization step can result in a material loss of approximately 10%, which accumulates significantly over a multi-step synthesis. Furthermore, the reliance on column chromatography for purifying cyclization products introduces prohibitive costs and solvent waste, making such methods economically unviable for commercial-scale production of high-volume antihypertensive intermediates.
Separating these positional isomers is notoriously difficult due to their similar physicochemical properties, often necessitating multiple recrystallization steps or expensive column chromatography. Literature indicates that each recrystallization step can result in a material loss of approximately 10%, which accumulates significantly over a multi-step synthesis. Furthermore, the reliance on column chromatography for purifying cyclization products introduces prohibitive costs and solvent waste, making such methods economically unviable for commercial-scale production of high-volume antihypertensive intermediates.
The Novel Approach
The innovative process described in the patent data circumvents these bottlenecks by eliminating intermediate purification entirely. By accepting the crude reaction mixtures from the nitration and reduction stages, the workflow bypasses the need for isolating unstable or difficult-to-separate intermediates. The critical breakthrough lies in the final hydrolysis stage, where the reaction conditions are tuned to selectively precipitate the target carboxylic acid while leaving impurities in the solution. Adjusting the pH to a narrow range of 6 to 7 triggers the crystallization of the high-purity intermediate directly from the reaction mass. This approach drastically reduces the number of unit operations, minimizes solvent consumption, and prevents the yield erosion associated with repeated solid-liquid separations. Consequently, the overall yield is preserved, and the operational complexity is reduced to a level that is highly favorable for continuous or batch manufacturing in a GMP environment.
Mechanistic Insights into FeCl3-Catalyzed Cyclization and Hydrolysis
The chemical transformation relies on a precise sequence of functional group manipulations that convert the starting benzoate derivative into the benzimidazole core. The initial nitration utilizes fuming nitric acid at low temperatures (-15 to -12°C) to kinetically favor the formation of the 5-nitro isomer over thermodynamic byproducts. Following this, catalytic hydrogenation using Raney Nickel efficiently reduces the nitro group to an amine without affecting the ester or amide functionalities. The subsequent cyclization in glacial acetic acid facilitates the condensation of the amine with the adjacent amide carbonyl, forming the benzimidazole ring system. 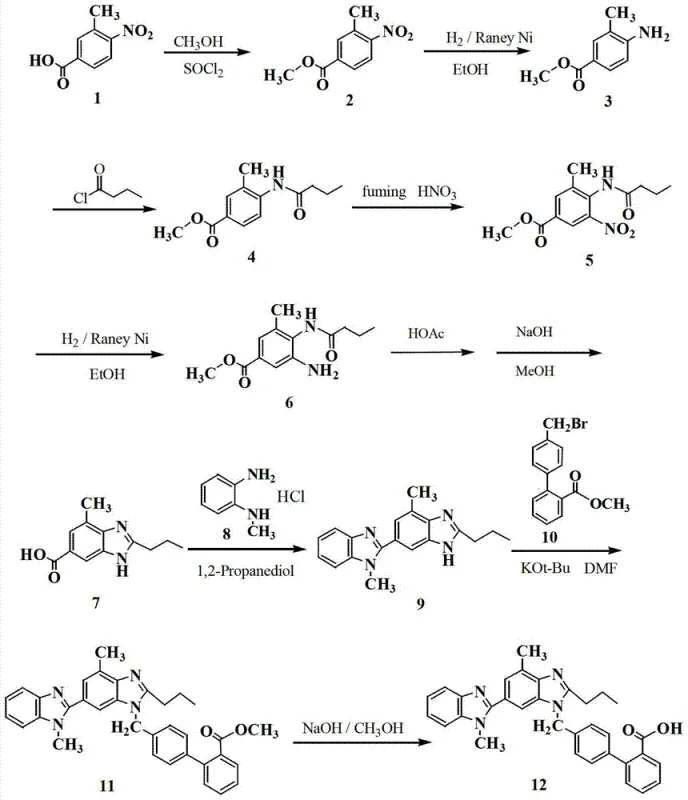 This cascade is carefully managed to ensure that side reactions, such as ester hydrolysis prior to cyclization, are minimized. The final saponification step uses sodium hydroxide in a methanol-water system, where the carboxylate salt remains soluble until the pH is carefully lowered. This pH-controlled precipitation acts as a powerful purification mechanism, leveraging the solubility differences between the zwitterionic target molecule and neutral organic impurities to achieve high purity without chromatographic intervention.
This cascade is carefully managed to ensure that side reactions, such as ester hydrolysis prior to cyclization, are minimized. The final saponification step uses sodium hydroxide in a methanol-water system, where the carboxylate salt remains soluble until the pH is carefully lowered. This pH-controlled precipitation acts as a powerful purification mechanism, leveraging the solubility differences between the zwitterionic target molecule and neutral organic impurities to achieve high purity without chromatographic intervention.
Impurity control is inherently built into the process design through the selective precipitation mechanism. In conventional methods, isomeric impurities like 5a and 5b persist through the synthesis and require aggressive purification at the end. In this improved route, the structural differences between the target benzimidazole carboxylic acid and the byproducts are exploited during the acidification phase. The target compound possesses a specific pKa profile that allows it to precipitate quantitatively at pH 6 to 7, whereas many non-acidic or differently charged impurities remain dissolved in the aqueous-organic mother liquor. Additionally, the use of fuming nitric acid instead of mixed sulfuric-nitric acid reduces the formation of sulfonated byproducts, further simplifying the impurity profile. This results in a crude product that is already of high quality, requiring only a single final recrystallization to meet the >99% purity specification required for downstream coupling reactions to form Telmisartan.
How to Synthesize 2-n-propyl-4-methyl-benzimidazole-6-carboxylic acid Efficiently
Implementing this synthesis route requires careful attention to temperature control and reagent stoichiometry to maximize the benefits of the telescoped process. The protocol begins with the nitration of the butyramide precursor, where maintaining the temperature below -10°C is critical to suppress isomer formation. The subsequent reduction and cyclization steps are performed on the crude materials, eliminating drying and weighing steps that expose intermediates to potential degradation. The final hydrolysis and precipitation step serves as the primary purification gate, where precise pH monitoring is essential to ensure optimal recovery and purity. Detailed standardized operating procedures for each stage, including safety handling of fuming nitric acid and Raney Nickel, are crucial for successful technology transfer.
- Perform nitration of methyl 3-methyl-4-n-butyramide benzoate using fuming nitric acid at -15 to -12°C to obtain crude nitro compound.
- Execute catalytic hydrogenation using Raney Ni to reduce the nitro group to an amino group without purifying the intermediate.
- Conduct cyclization in glacial acetic acid followed by hydrolysis, adjusting pH to 6-7 to precipitate the final high-purity intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this optimized synthesis route translates into tangible strategic advantages regarding cost stability and supply reliability. By removing multiple purification steps such as recrystallization and column chromatography, the manufacturing process becomes significantly leaner. This reduction in unit operations directly correlates to lower labor costs, reduced equipment occupancy time, and decreased consumption of high-purity solvents. The elimination of chromatographic separation, in particular, removes a major bottleneck that often limits batch size and throughput in traditional fine chemical manufacturing. Consequently, the production capacity can be scaled up more easily to meet fluctuating market demands for antihypertensive medications without requiring proportional increases in capital expenditure or facility footprint.
- Cost Reduction in Manufacturing: The streamlined process eliminates the need for expensive stationary phases and large volumes of elution solvents associated with column chromatography. Furthermore, by avoiding intermediate isolation, the yield losses typically incurred during filtration and drying of unstable intermediates are completely prevented. This cumulative saving in material and utility costs allows for a more competitive pricing structure for the final API intermediate. The simplified waste stream, resulting from the substitution of mixed acids with fuming nitric acid and the reduction of solvent usage, also lowers the environmental compliance costs associated with wastewater treatment and hazardous waste disposal.
- Enhanced Supply Chain Reliability: A shorter and less complex synthesis route inherently reduces the risk of batch failures and production delays. With fewer steps where quality deviations can occur, the consistency of the supply is significantly improved. The robustness of the pH-controlled precipitation method ensures that even with minor variations in upstream crude quality, the final product specifications can still be met. This reliability is critical for pharmaceutical buyers who require uninterrupted supply to maintain their own finished drug product inventories. The ability to produce high-purity intermediates consistently reduces the need for safety stock, allowing for leaner inventory management across the supply chain.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing common reagents and standard reactor configurations. The avoidance of complex separations makes the technology easier to transfer from pilot scale to multi-ton commercial production. From an environmental perspective, the reduction in solvent waste and the elimination of silica gel disposal contribute to a greener manufacturing profile. This aligns with the increasing regulatory pressure on pharmaceutical suppliers to adopt sustainable chemistry practices. The simplified effluent profile facilitates easier treatment, ensuring long-term compliance with evolving environmental regulations in major manufacturing hubs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Telmisartan intermediate synthesis technology. These insights are derived directly from the patent specifications and practical considerations for scaling the process in a commercial setting. Understanding these nuances helps stakeholders evaluate the feasibility and benefits of adopting this improved manufacturing route for their supply chains.
Q: How does the new process eliminate purification losses?
A: The process avoids recrystallization and column chromatography for intermediates 5 and 6, instead relying on pH adjustment during the final hydrolysis step to precipitate the target product, significantly reducing material loss.
Q: What are the critical reaction conditions for nitration?
A: The nitration step requires strict temperature control between -15°C and -12°C using 95% fuming nitric acid to minimize the formation of ortho- and para-nitro isomers.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, by removing complex separation steps like column chromatography and reducing solvent usage, the process is designed for industrial scalability with lower environmental impact and operational complexity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Telmisartan Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for high-demand pharmaceutical intermediates like Telmisartan. Our technical team has extensively analyzed advanced processes such as the one described in CN103319414A to ensure our manufacturing capabilities align with the latest industry standards. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of process optimization are realized at volume. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of intermediate meets the exacting requirements necessary for downstream API synthesis.
We invite global partners to collaborate with us to leverage these technological advancements for their product portfolios. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential clients to contact us to obtain specific COA data and route feasibility assessments, ensuring that our solutions perfectly match your quality and timeline expectations for reliable pharmaceutical intermediate supply.
