Optimizing TPO Photoinitiator Production: A Novel Acetate Recrystallization Strategy for Commercial Scale-Up
The global demand for high-efficiency photoinitiators in UV curing applications has necessitated a re-evaluation of purification protocols for critical intermediates like 2,4,6-trimethylbenzoyl-diphenylphosphine oxide, commonly known as TPO. As detailed in patent CN111057106A, a significant technological breakthrough has been achieved regarding the recrystallization of this compound, addressing long-standing issues related to purity, yield, and operational safety. TPO is a cornerstone molecule in the photochemical industry, serving as a Type I free radical photoinitiator with a broad absorption spectrum ranging from 350nm to 420nm, making it indispensable for LED curing systems, optical fiber coatings, and advanced composite materials. 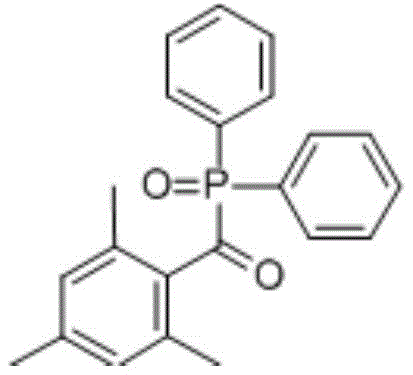 The structural integrity and purity of TPO are paramount, as commercial standards strictly require purity levels exceeding 99%, a threshold that traditional purification methods often struggle to meet consistently without sacrificing yield. This new preparation method introduces a specialized recrystallization step utilizing specific acetate esters, offering a robust solution for producing high-purity TPO from crude reaction mixtures with exceptional efficiency.
The structural integrity and purity of TPO are paramount, as commercial standards strictly require purity levels exceeding 99%, a threshold that traditional purification methods often struggle to meet consistently without sacrificing yield. This new preparation method introduces a specialized recrystallization step utilizing specific acetate esters, offering a robust solution for producing high-purity TPO from crude reaction mixtures with exceptional efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of TPO has relied heavily on solvents such as petroleum ether, n-hexane, or mixed solvent systems involving ethyl acetate and petroleum ether, as disclosed in various prior art patents like US4298738 and CN109293697. These conventional approaches suffer from inherent deficiencies that complicate large-scale manufacturing and compromise economic viability. For instance, hydrocarbon solvents like petroleum ether and n-hexane possess extremely low flash points, often below 0°C, creating severe fire and explosion hazards during industrial operations where large volumes of solvent are handled. Furthermore, while mixed solvent systems can achieve acceptable purity, they introduce complex downstream processing requirements; separating and recovering multiple solvents from the mother liquor is energy-intensive and technically demanding, leading to increased operational costs and higher VOC emissions. Additionally, the use of highly polar solvents like ethanol, while effective for purity, typically results in unacceptably low yields due to the high solubility of the product in the mother liquor, rendering the process economically inefficient for mass production.
The Novel Approach
In stark contrast to these legacy methods, the novel approach outlined in the patent leverages specific acetate esters, such as butyl acetate, isopropyl acetate, and sec-butyl acetate, to achieve a superior balance between solubility and crystallization efficiency. 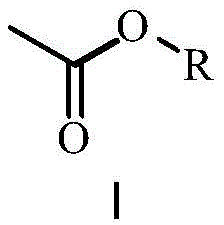 By selecting solvents with moderate polarity, this method ensures that impurities remain dissolved in the mother liquor while the target TPO product crystallizes out with high efficiency. The data indicates that this strategy allows for the production of TPO with purity levels consistently above 99% while maintaining yields exceeding 80%, a performance metric that significantly outperforms traditional hydrocarbon or alcohol-based systems. Moreover, the selected acetates possess higher flash points compared to petroleum ether, drastically improving the intrinsic safety profile of the manufacturing process. This shift not only mitigates safety risks but also simplifies solvent recovery, as single-component acetate systems do not require the complex distillation columns needed for separating mixed solvent azeotropes, thereby streamlining the entire production workflow.
By selecting solvents with moderate polarity, this method ensures that impurities remain dissolved in the mother liquor while the target TPO product crystallizes out with high efficiency. The data indicates that this strategy allows for the production of TPO with purity levels consistently above 99% while maintaining yields exceeding 80%, a performance metric that significantly outperforms traditional hydrocarbon or alcohol-based systems. Moreover, the selected acetates possess higher flash points compared to petroleum ether, drastically improving the intrinsic safety profile of the manufacturing process. This shift not only mitigates safety risks but also simplifies solvent recovery, as single-component acetate systems do not require the complex distillation columns needed for separating mixed solvent azeotropes, thereby streamlining the entire production workflow.
Mechanistic Insights into Acetate-Mediated Recrystallization
The success of this purification strategy lies in the precise thermodynamic interaction between the acetate solvent molecules and the TPO crystal lattice. Acetate esters exhibit a unique solubility profile where they demonstrate poor solubility for TPO at lower temperatures but excellent solubility for common organic impurities generated during the synthesis of TPO, whether via rearrangement or addition-oxidation pathways. This differential solubility is the driving force behind the high purity achieved; as the solution cools, the TPO molecules aggregate into a stable crystal lattice, excluding impurity molecules which remain solvated in the liquid phase. Crucially, the patent highlights that certain acetates, particularly butyl acetate, maintain chemical stability over multiple recycling cycles. Unlike ethyl acetate, which is prone to hydrolysis and an increase in acid value upon repeated use—leading to the decomposition of the acid-sensitive TPO molecule—the selected higher acetates resist degradation. This stability ensures that the solvent quality does not deteriorate over time, preserving the purity specifications of the final product even after the mother liquor has been reused up to four times.
Furthermore, the crystallization kinetics facilitated by these acetates promote the formation of well-defined, larger crystals rather than fine powders. This morphological control is mechanistically significant because it reduces the surface area-to-volume ratio of the product particles, minimizing the adsorption of residual solvent and impurities on the crystal surface. From a processing standpoint, larger crystals improve filtration rates and reduce the retention of mother liquor, directly contributing to higher isolated yields. The avoidance of fine powder formation also has practical implications for downstream handling; it eliminates issues related to static electricity and dust generation, which are common pain points in the packaging and feeding of photoinitiators. This mechanistic advantage translates directly into improved operational safety and product consistency, ensuring that the TPO supplied to end-users meets rigorous quality standards without requiring additional milling or processing steps that could introduce contamination.
How to Synthesize 2,4,6-Trimethylbenzoyl-diphenylphosphine Oxide Efficiently
The synthesis and subsequent purification of TPO using this acetate-based recrystallization method represent a streamlined pathway for industrial chemists aiming to optimize their production lines. The process begins with the dissolution of crude TPO, typically obtained from standard rearrangement or oxidation synthesis routes, into a calculated amount of the selected acetate solvent. The patent suggests a solvent-to-crude mass ratio ranging from 0.5 to 3 times, with an optimal range of 0.8 to 1.2 times, which is significantly lower than the solvent volumes required for mixed solvent systems. This dissolution can be performed under heating to ensure complete solubilization, followed by a controlled cooling phase to induce crystallization. The detailed standardized synthesis steps, including specific temperature ramps, stirring rates, and filtration protocols necessary to replicate these results in a GMP environment, are provided in the section below.
- Dissolve the crude TPO product in a specific acetate solvent (such as butyl acetate or isopropyl acetate) under heating or stirring until completely dissolved.
- Cool the solution naturally or via controlled cooling to a temperature between -10°C and 30°C to induce crystallization.
- Filter the crystals, wash with cold solvent, and dry using vacuum or airflow drying to obtain high-purity TPO.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this acetate-based recrystallization technology offers profound strategic benefits that extend beyond simple chemical yield improvements. The primary advantage lies in the substantial reduction of operational complexity and risk exposure associated with solvent management. By eliminating the need for hazardous, low-flash-point solvents like petroleum ether, facilities can significantly lower their insurance premiums and compliance costs related to fire safety regulations. Furthermore, the ability to reuse the acetate mother liquor multiple times without loss of performance creates a closed-loop system that drastically reduces raw material consumption. This circular approach not only lowers the direct cost of goods sold but also aligns with increasingly stringent environmental regulations regarding VOC emissions and waste disposal, positioning the manufacturer as a sustainable partner in the global supply chain.
- Cost Reduction in Manufacturing: The implementation of this novel purification route drives cost efficiency through multiple mechanisms, primarily by optimizing solvent usage and energy consumption. Since the required volume of acetate solvent is roughly half that of traditional mixed solvent systems, the energy load on recovery distillation units is correspondingly reduced, leading to lower utility costs per kilogram of product. Additionally, the high yield achieved (>80%) means that less crude material is required to produce the same amount of finished goods, effectively maximizing the throughput of existing reactor capacity. The elimination of complex solvent separation steps further reduces maintenance costs and downtime associated with specialized distillation equipment, resulting in a leaner, more cost-effective manufacturing operation that can offer competitive pricing without compromising margins.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of commodity acetate solvents, which are widely available and less subject to the supply volatility often seen with specialized hydrocarbon blends. The robustness of the process, characterized by its tolerance for varying crude qualities and its ability to consistently deliver >99% purity, ensures a steady flow of high-quality material to downstream customers. This reliability minimizes the risk of production stoppages due to off-spec batches, a critical factor for just-in-time manufacturing environments in the coatings and electronics sectors. Moreover, the improved crystal form of the product enhances its flowability and stability during storage and transport, reducing the likelihood of caking or degradation during long-distance shipping, thereby ensuring that the product arrives at the customer's site in optimal condition.
- Scalability and Environmental Compliance: Scaling this process from pilot plant to commercial production is inherently safer and more straightforward due to the favorable physical properties of the acetate solvents. The higher flash points allow for operation at elevated temperatures with reduced risk of ignition, facilitating faster reaction cycles and higher batch frequencies. From an environmental perspective, the reduction in total solvent volume and the ability to recycle the mother liquor translate to a significantly smaller carbon footprint and lower hazardous waste generation. This aligns perfectly with global sustainability goals and helps corporate partners meet their Scope 3 emission targets. The process design inherently supports green chemistry principles by preventing waste at the source rather than treating it post-generation, making it an ideal candidate for facilities aiming to achieve ISO 14001 certification or similar environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this acetate recrystallization technology for TPO production. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for R&D teams evaluating process transfer and for procurement specialists assessing the long-term viability of this supply source.
Q: Why are acetate solvents preferred over petroleum ether for TPO purification?
A: Acetate solvents offer a superior balance of solubility, allowing for high yields (>80%) while achieving purity levels above 99%. Unlike petroleum ether, they have higher flash points, significantly reducing fire hazards during large-scale production, and avoid the complex separation issues associated with mixed solvent systems.
Q: Can the recrystallization mother liquor be reused in this process?
A: Yes, the patent data indicates that acetate mother liquors, particularly butyl acetate, remain stable and effective for up to four recycling cycles without compromising the purity of the final TPO product, unlike ethyl acetate which may degrade due to acid value increases.
Q: What is the impact of this method on the physical form of the final product?
A: This recrystallization method promotes the formation of well-defined crystals rather than fine powders. This improves handling safety by reducing static electricity and dust飞扬 (flying powder), protecting both manufacturers and downstream users during feeding processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4,6-Trimethylbenzoyl-diphenylphosphine oxide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced purification technologies like the acetate-based recrystallization of TPO requires a partner with deep technical expertise and proven scale-up capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a commercial setting. We operate stringent purity specifications and maintain rigorous QC labs equipped to verify the >99% purity and optimal crystal morphology of every batch, guaranteeing that our photoinitiators meet the exacting demands of the UV curing and electronic materials industries. Our commitment to quality assurance means that every shipment is backed by comprehensive analytical data, providing our partners with the confidence needed to integrate our materials into their high-performance formulations.
We invite you to engage with our technical procurement team to discuss how this optimized TPO production method can enhance your supply chain efficiency and product performance. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the potential economic benefits specific to your volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to validate the compatibility of our high-purity TPO with your existing manufacturing processes. Let us collaborate to drive innovation and efficiency in your photoinitiator supply chain, leveraging cutting-edge chemistry to secure your competitive advantage in the global market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
