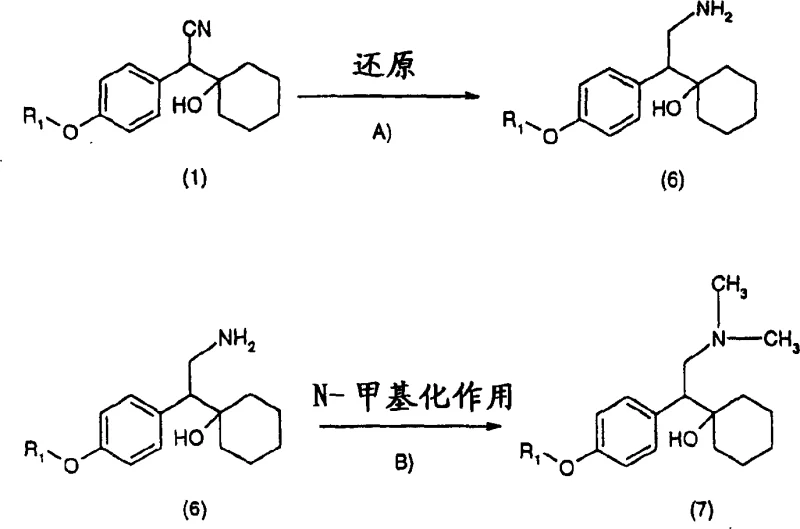
Advanced Manufacturing of Venlafaxine Intermediates Using Aqueous Phase Transfer Catalysis
The pharmaceutical industry continuously seeks robust, scalable, and environmentally benign synthetic routes for critical active pharmaceutical ingredient (API) intermediates. Patent CN1276913C represents a significant technological breakthrough in the synthesis of substituted phenylacetonitrile compounds, which serve as pivotal precursors for central nervous system antidepressants such as venlafaxine. This intellectual property discloses a novel methodology that replaces hazardous organometallic reagents with a greener, aqueous-based phase transfer catalysis (PTC) system. By shifting from traditional cryogenic organolithium chemistry to a mild aqueous alkaline environment, this process addresses long-standing challenges regarding yield optimization, operational safety, and waste management. For global procurement and R&D teams, understanding the nuances of this patent is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials with consistent quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of key intermediates like those described in Formula (1) relied heavily on the methodology outlined in US-A-4,535,186. This conventional approach necessitates the use of n-butyllithium, a pyrophoric reagent that demands stringent handling protocols and specialized equipment to prevent ignition and ensure operator safety. Furthermore, the reaction requires anhydrous organic solvents such as tetrahydrofuran (THF) and cyclohexane, which introduce significant volatility risks and environmental burdens related to solvent recovery and disposal. From a process chemistry perspective, the reliance on cryogenic conditions to control the exothermic nature of the organolithium addition often results in inconsistent mixing and localized hot spots, leading to variable product quality. Most critically, the overall yield of this legacy method rarely exceeds 50%, creating substantial material inefficiencies that drive up the cost of goods sold (COGS) and limit the economic viability of large-scale production campaigns.
The Novel Approach
In stark contrast, the technology protected under CN1276913C introduces a paradigm shift by utilizing an aqueous base in conjunction with a phase transfer catalyst to facilitate the condensation reaction between alkoxyphenylacetonitriles and cyclohexanone. This innovative route operates effectively at temperatures ranging from 0 to 60°C, with optimal performance observed at ambient conditions between 15 to 35°C, thereby eliminating the energy-intensive cooling requirements of the prior art. The elimination of bulk organic solvents during the reaction phase not only simplifies the workup procedure but also drastically reduces the facility's fire load and volatile organic compound (VOC) emissions. Experimental data within the patent demonstrates that yields can be consistently improved to the range of 90% to 95%, nearly doubling the efficiency of the traditional process. This transition to a biphasic aqueous system exemplifies the principles of green chemistry while simultaneously enhancing the economic profile of the manufacturing process.

Mechanistic Insights into Phase Transfer Catalyzed Condensation
The core innovation of this process lies in the sophisticated application of phase transfer catalysis to overcome the immiscibility of the organic reactants and the aqueous base. In this system, the phase transfer catalyst, typically a quaternary ammonium salt such as tetrabutylammonium bromide (TBAB) or tetrabutylammonium chloride (TBACl), acts as a molecular shuttle. It transports hydroxide ions from the aqueous phase into the organic interface or organic phase, where they can deprotonate the alpha-carbon of the phenylacetonitrile derivative to generate a reactive nucleophile. This nucleophile then attacks the carbonyl group of cyclohexanone to form the desired carbon-carbon bond. The choice of catalyst loading is critical; the patent specifies a range of 0.0001 to 0.1 molar equivalents, with preferred embodiments utilizing as little as 0.0005 to 0.05 equivalents. This high catalytic efficiency ensures that the reaction proceeds rapidly without the need for stoichiometric amounts of expensive phase transfer agents, further contributing to cost reduction in pharmaceutical intermediate manufacturing.
Furthermore, the mechanistic pathway offers superior control over impurity profiles compared to organometallic routes. The mild basicity of aqueous sodium hydroxide or potassium hydroxide prevents the aggressive side reactions often associated with strong bases like n-butyllithium, such as over-alkylation or decomposition of sensitive functional groups. The reaction stoichiometry is carefully balanced, with the ketone (Formula 3) used in a slight excess of 0.9 to 1.8 molar equivalents relative to the nitrile (Formula 2) to drive the equilibrium forward without generating excessive byproducts. Following the reaction, the product precipitates or can be easily separated, allowing for a straightforward isolation via filtration. This simplicity in downstream processing minimizes the number of unit operations required, reducing both capital expenditure on equipment and operational time, which is a crucial factor for supply chain heads focused on reducing lead time for high-purity pharmaceutical intermediates.
How to Synthesize Substituted Phenylacetonitrile Efficiently
Implementing this synthesis requires precise adherence to the optimized parameters defined in the patent to ensure maximum yield and purity. The process begins with the preparation of the reaction mixture, where the alkoxyphenylacetonitrile and cyclohexanone are combined, followed by the controlled addition of the aqueous alkaline solution and the catalyst. The absence of organic solvents during the reaction phase simplifies the initial setup, but vigorous stirring is essential to maintain the emulsion and ensure efficient mass transfer between the phases. Temperature control remains important, with the patent highlighting that reactions can proceed effectively at room temperature (e.g., 27°C) or slightly cooler conditions (e.g., 18°C), offering flexibility depending on the specific thermal management capabilities of the production facility. Detailed standardized synthetic steps see the guide below.
- Mix alkoxyphenylacetonitrile (Formula 2) and cyclohexanone (Formula 3) without organic solvents.
- Add aqueous alkali metal hydroxide solution (e.g., 10% NaOH) and a quaternary ammonium salt catalyst like TBAB.
- Stir the mixture at 0-60°C (preferably room temperature) until completion, then filter and dry the solid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the technology described in CN1276913C offers transformative benefits that extend far beyond simple yield improvements. The shift away from hazardous reagents and volatile solvents fundamentally alters the risk profile of the supply chain, ensuring greater continuity of supply by mitigating the risks associated with the transport and storage of dangerous goods. Additionally, the simplified purification process reduces the dependency on complex distillation columns or chromatography systems, lowering the barrier to entry for contract manufacturing organizations (CMOs) and expanding the pool of potential qualified suppliers. This accessibility fosters a more competitive market environment, driving down costs and ensuring that buyers have multiple sources for these critical building blocks.
- Cost Reduction in Manufacturing: The elimination of n-butyllithium and anhydrous organic solvents represents a massive reduction in raw material costs. Organolithium reagents are notoriously expensive and require specialized logistics for safe delivery, whereas aqueous sodium hydroxide is a commodity chemical available globally at a fraction of the price. Furthermore, the removal of solvent recovery steps significantly lowers energy consumption and utility costs associated with distillation and waste treatment. The dramatic increase in yield from less than 50% to over 90% effectively halves the raw material input required per kilogram of finished product, resulting in substantial cost savings that can be passed down through the supply chain.
- Enhanced Supply Chain Reliability: By utilizing common, non-hazardous reagents, the manufacturing process becomes less susceptible to regulatory bottlenecks and transportation delays. Facilities do not require specialized permits for handling pyrophoric materials, allowing for production in a wider range of geographic locations. This decentralization capability enhances supply security, ensuring that disruptions in one region do not halt global production of the final API. The robustness of the reaction conditions also means that batch-to-batch variability is minimized, providing procurement teams with the confidence of consistent quality and reliable delivery schedules.
- Scalability and Environmental Compliance: The aqueous nature of the reaction medium makes scale-up inherently safer and more predictable. Heat dissipation is more efficient in water than in organic solvents, reducing the risk of thermal runaway during large-scale production. Moreover, the process aligns with increasingly stringent environmental regulations regarding VOC emissions and hazardous waste generation. The reduction in organic waste streams simplifies effluent treatment and lowers the environmental compliance burden, making this route highly attractive for companies aiming to meet sustainability goals and reduce their carbon footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this phase transfer catalysis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential partners and stakeholders.
Q: What are the advantages of using aqueous base over n-butyllithium for this synthesis?
A: Using aqueous base eliminates the need for pyrophoric reagents and cryogenic conditions, significantly improving safety and reducing operational costs compared to traditional organolithium methods.
Q: Which phase transfer catalysts are most effective for this reaction?
A: Quaternary ammonium salts such as tetrabutylammonium bromide (TBAB) or tetrabutylammonium chloride (TBACl) are highly effective, typically used in catalytic amounts of 0.0001 to 0.1 molar equivalents.
Q: Can this process be scaled for commercial production of antidepressant intermediates?
A: Yes, the process operates at mild temperatures (0-60°C) without volatile organic solvents, making it inherently safer and easier to scale for commercial manufacturing of CNS active pharmaceutical ingredients.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted Phenylacetonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable synthetic routes in the modern pharmaceutical landscape. Our technical team has extensively analyzed the methodologies outlined in CN1276913C and possesses the expertise to implement this advanced phase transfer catalysis technology at an industrial scale. We offer extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a steady supply of high-quality intermediates. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch meets the exacting standards required for GMP manufacturing of antidepressant APIs.
We invite global partners to collaborate with us to leverage these process innovations for their supply chains. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage interested parties to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both your product quality and your bottom line.
