Advanced Catalytic Synthesis of 6,6-Dimethyl-3-azabicyclo[3.1.0]hexane for Scalable Antiviral Production
The pharmaceutical industry is constantly seeking more efficient pathways to synthesize critical building blocks for antiviral therapies, particularly for treatments targeting Hepatitis C and emerging coronaviruses. Patent CN114591217A introduces a groundbreaking methodology for the preparation of 6,6-dimethyl-3-azabicyclo[3.1.0]hexane, a pivotal scaffold found in potent protease inhibitors. This technical disclosure outlines a novel four-step sequence that bypasses traditional bottlenecks, utilizing a lactone intermediate to achieve superior atom economy and structural integrity. The significance of this intermediate cannot be overstated, as it serves as the core structural motif for blockbuster drugs such as Boceprevir and the oral antiviral PF-07321332. By shifting the synthetic paradigm from complex natural product derivatives to a concise catalytic assembly, this technology offers a robust solution for securing the supply chain of essential medicines.
![Chemical structures of Boceprevir and PF-07321332 highlighting the 6,6-dimethyl-3-azabicyclo[3.1.0]hexane core](/insights/img/6-6-dimethyl-3-azabicyclo-hexane-synthesis-pharma-supplier-20260313203121-01.webp)
For R&D directors evaluating process viability, the structural fidelity and purity of the final amine are paramount. The patented route ensures high stereocontrol during the initial ring-forming step, which is critical for the biological activity of the downstream API. As a reliable pharmaceutical intermediate supplier, understanding these mechanistic nuances allows us to guarantee the delivery of high-purity 6,6-dimethyl-3-azabicyclo[3.1.0]hexane that meets the rigorous specifications required for clinical and commercial drug substance manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 6,6-dimethyl-3-azabicyclo[3.1.0]hexane has relied heavily on the degradation and modification of natural terpenes or their synthetic analogs, specifically ethyl chrysanthemate or cyhalothric acid. These traditional routes typically involve the conversion of these starting materials into caronic anhydride, which subsequently undergoes amination and reduction to yield the target bicyclic amine. However, this classical approach is fraught with inefficiencies; the synthesis of caronic anhydride itself is a multi-step process with poor atom economy, generating significant stoichiometric waste. Furthermore, the reliance on specific terpene-derived precursors can introduce supply chain volatility, as the availability and price of these natural product derivatives are subject to agricultural and market fluctuations. The lengthy reaction sequence also increases the cumulative loss of material at each stage, resulting in lower overall yields and higher production costs, which is unsustainable for the high-volume demands of modern antiviral drug manufacturing.
The Novel Approach
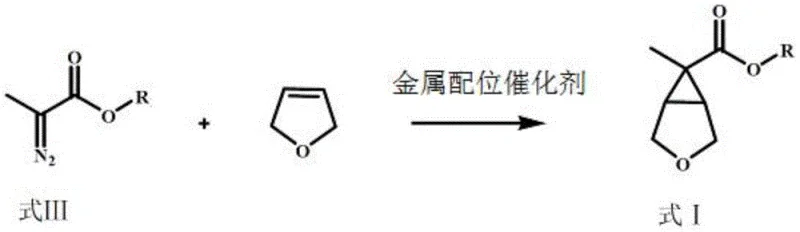
In stark contrast, the methodology disclosed in CN114591217A revolutionizes the synthesis by employing a direct cyclopropanation strategy. The process initiates with the reaction of a readily available 2-diazopropionate compound and 2,5-dihydrofuran under the influence of a metal coordination catalyst. This single step constructs the complex bicyclic lactone framework (Formula I) with remarkable efficiency. Following this key transformation, the lactone is subjected to a streamlined sequence of reduction, dealcoholization, and amination to afford the final amine. This new route drastically reduces the number of operational units and eliminates the need for the cumbersome caronic anhydride intermediate. By leveraging simple, commodity chemicals as starting materials, the process enhances the robustness of the supply chain and significantly lowers the barrier to entry for large-scale production, making it an ideal candidate for cost reduction in API manufacturing.
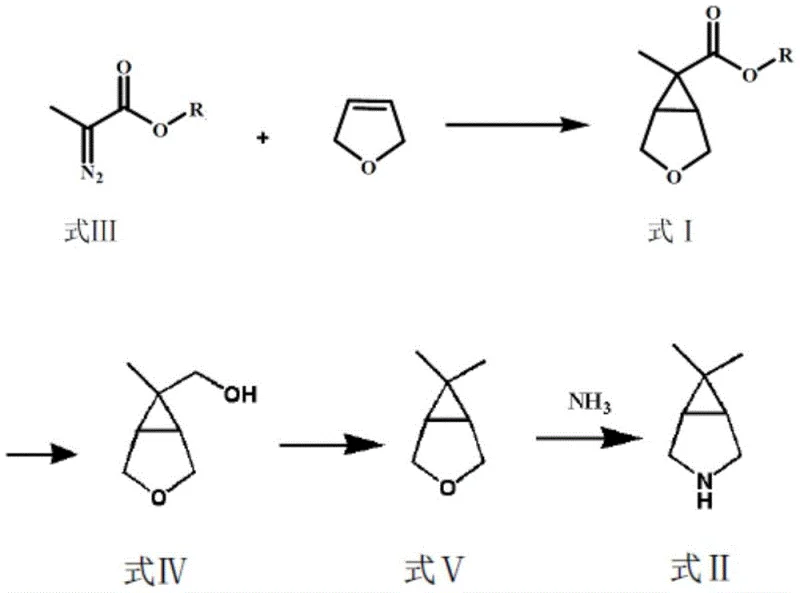
Mechanistic Insights into Metal-Catalyzed Cyclopropanation
The cornerstone of this innovative synthesis is the metal-catalyzed addition reaction between the diazo species and the dihydrofuran derivative. This transformation proceeds through the formation of a reactive metal-carbene intermediate, which subsequently engages in a [2+1] cycloaddition with the electron-rich double bond of the dihydrofuran. The choice of catalyst is critical; the patent specifies the use of metal coordination catalysts prepared from metal compounds containing Group VIII metals, molybdenum, or copper, coordinated with nitrogen-oxygen or nitrogen-nitrogen multidentate ligands. Copper salts, such as copper(I) chloride or copper(II) trifluoromethanesulfonate, have been identified as particularly effective when paired with specific ligands like L1, L2, or L3. These ligands create a chiral or sterically defined environment around the metal center, which not only accelerates the reaction rate but also exerts profound control over the stereoselectivity of the cyclopropanation.
Controlling the cis-to-trans ratio of the lactone intermediate is essential for maximizing the yield of the desired isomer in subsequent steps. The patent data indicates that optimizing the ligand-to-metal ratio and selecting appropriate solvents like dichloroethane or toluene can push the cis-isomer content to over 90%. This high level of stereocontrol minimizes the formation of diastereomeric impurities that would otherwise be difficult to separate later in the synthesis. The mechanism likely involves the coordination of the diazo compound to the metal center, followed by the extrusion of nitrogen gas to form the electrophilic carbene. The dihydrofuran then attacks this carbene species in a concerted manner, closing the three-membered ring while preserving the oxygen heterocycle, setting the stage for the subsequent reductive manipulation of the lactone functionality.
How to Synthesize 6,6-Dimethyl-3-azabicyclo[3.1.0]hexane Efficiently
Implementing this synthesis requires precise control over reaction parameters to ensure safety and reproducibility, particularly given the use of diazo compounds and high-pressure amination. The process begins with the careful addition of the diazo precursor to the catalyst-dihydrofuran mixture at elevated temperatures, typically between 60°C and 80°C, to manage the exotherm and nitrogen evolution. Following the isolation of the lactone, the reduction step utilizes hydride sources such as lithium aluminum hydride or sodium borohydride activated with boron trifluoride to open the lactone ring to the corresponding alcohol. The subsequent dealcoholization converts the hydroxyl group into a leaving group, such as a halide or sulfonate, which is then reductively removed to form the ether bridge. Finally, the oxabicyclic intermediate is subjected to ammonolysis under high pressure and temperature to install the secondary amine. For a detailed breakdown of the specific reagents, molar ratios, and workup procedures, please refer to the standardized guide below.
- Perform a metal-catalyzed addition reaction between a 2-diazopropionate compound and 2,5-dihydrofuran to generate the key lactone intermediate (Formula I).
- Subject the lactone intermediate to a reduction reaction using agents like lithium aluminum hydride or sodium borohydride to obtain the alcohol intermediate (Formula IV).
- Execute dealcoholization and hydroxyl removal on the alcohol intermediate to form the oxabicyclo intermediate (Formula V), followed by high-pressure amination to yield the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this lactone-based route represents a strategic opportunity to de-risk the sourcing of critical antiviral intermediates. The shift away from terpene-derived starting materials towards petrochemical-based feedstocks like 2,5-dihydrofuran and diazo esters ensures a more stable and predictable supply base. These commodity chemicals are produced on a massive global scale, insulating the manufacturing process from the agricultural variances that affect traditional routes. Furthermore, the reduction in the number of synthetic steps directly correlates to a reduction in processing time and facility occupancy, allowing for faster turnaround times and increased production capacity without the need for significant capital expenditure on new equipment.
- Cost Reduction in Manufacturing: The elimination of the caronic anhydride intermediate removes several costly unit operations, including the synthesis and purification of that specific precursor. By shortening the overall synthetic route, the consumption of solvents, reagents, and energy is drastically reduced. Additionally, the high atom economy of the cyclopropanation step means that a greater proportion of the raw material mass is incorporated into the final product, minimizing waste disposal costs. The use of earth-abundant copper catalysts instead of precious metals further contributes to substantial cost savings, making the process economically viable for generic drug production.
- Enhanced Supply Chain Reliability: Diversifying the raw material base to include widely available industrial chemicals mitigates the risk of supply disruptions. Unlike natural product extracts, which can suffer from seasonal shortages or quality inconsistencies, the synthetic precursors used in this route are subject to strict industrial quality controls. This reliability ensures continuous production schedules, which is critical for maintaining the inventory levels required by global pharmaceutical clients. The robustness of the catalytic system also allows for flexibility in sourcing, as various copper salts and ligand variants can be employed without compromising the overall process efficiency.
- Scalability and Environmental Compliance: The process is inherently scalable, with reaction conditions that are compatible with standard batch and continuous flow reactors. The simplified workflow reduces the generation of hazardous waste streams associated with multi-step anhydride syntheses. Moreover, the high selectivity of the catalyst reduces the burden on downstream purification systems, such as chromatography or crystallization, leading to a cleaner manufacturing profile. This alignment with green chemistry principles facilitates easier regulatory approval and supports the sustainability goals of modern pharmaceutical enterprises.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and claims within CN114591217A, providing a factual basis for evaluating the technology's potential impact on your supply chain. Understanding these details is crucial for making informed decisions about process adoption and vendor qualification.
Q: What are the primary advantages of the lactone intermediate route over the traditional caronic anhydride method?
A: The lactone intermediate route significantly shortens the synthetic pathway and improves atom economy compared to the traditional method which relies on ethyl chrysanthemate or cyhalothric acid. This novel approach avoids the complex synthesis of caronic anhydride, leading to a more streamlined process with potentially higher overall yields and reduced waste generation.
Q: Which catalysts are most effective for the initial cyclopropanation step in this process?
A: The patent highlights the use of metal coordination catalysts, particularly copper compounds such as copper(I) chloride or copper(II) trifluoromethanesulfonate, combined with specific nitrogen-oxygen or nitrogen-nitrogen multidentate ligands. These catalysts facilitate the addition reaction between 2-diazopropionate and 2,5-dihydrofuran with high stereoselectivity, favoring the cis-isomer.
Q: Is this synthesis route suitable for large-scale commercial production of antiviral intermediates?
A: Yes, the process is designed with industrial scalability in mind. It utilizes commercially available starting materials like 2,5-dihydrofuran and diazo compounds, and the reaction conditions, including the high-pressure amination step, are consistent with standard chemical engineering practices for API intermediate manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6,6-Dimethyl-3-azabicyclo[3.1.0]hexane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global fight against viral diseases. Our technical team has thoroughly analyzed the pathway described in CN114591217A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are equipped with state-of-the-art high-pressure reactors and rigorous QC labs capable of handling the specific demands of this copper-catalyzed cyclopropanation and subsequent high-temperature amination steps. Our commitment to stringent purity specifications ensures that every batch of 6,6-dimethyl-3-azabicyclo[3.1.0]hexane we deliver meets the exacting standards required for GMP API synthesis, providing you with a secure and compliant source for your drug development programs.
We invite you to collaborate with us to optimize your supply chain for antiviral therapeutics. By leveraging our expertise in process chemistry and scale-up, we can help you realize the full economic potential of this novel synthetic route. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term production goals and ensure the uninterrupted availability of this vital pharmaceutical building block.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
