Advanced Synthetic Route for Citalopram Intermediates: Enhancing Purity and Commercial Scalability
The pharmaceutical industry continuously seeks robust synthetic pathways that balance high purity with economic efficiency, particularly for high-volume antidepressants like citalopram. Patent CN1161350C discloses a significant methodological advancement in the preparation of citalopram and its key intermediates, specifically focusing on the conversion of a novel aldehyde precursor into the corresponding 5-cyano compound. This technical breakthrough addresses long-standing challenges in impurity control and process safety, offering a viable alternative to traditional methods that often rely on hazardous cyanation reagents or complex transition metal catalysis. For R&D directors and procurement managers alike, understanding the nuances of this patent is critical for optimizing supply chains and ensuring the consistent quality of active pharmaceutical ingredients. The structural integrity of the final molecule, as depicted below, relies heavily on the precision of these intermediate steps.
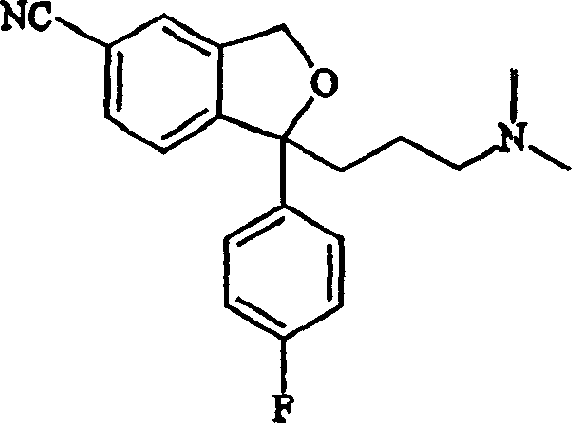
As a reliable pharmaceutical intermediates supplier, analyzing this patent reveals a pathway that not only improves yield but also simplifies the downstream purification processes. The ability to isolate intermediates in high purity directly impacts the cost reduction in API manufacturing, as fewer purification cycles translate to lower solvent consumption and reduced waste disposal costs. Furthermore, the described methodology enhances supply chain reliability by utilizing readily available starting materials and standard reagents, mitigating the risks associated with specialized catalyst shortages. This report delves deep into the mechanistic insights and commercial implications of this technology, providing actionable intelligence for decision-makers in the global fine chemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of citalopram intermediates has been plagued by several technical bottlenecks that hinder efficient commercial production. Traditional routes often involve the introduction of the cyano group using cuprous cyanide, a reagent that poses significant safety hazards and environmental concerns due to the generation of heavy metal waste. The removal of residual copper to meet stringent pharmaceutical specifications requires additional chelating steps and extensive washing, which drastically increases processing time and operational costs. Moreover, conventional methods frequently suffer from poor regioselectivity during the cyclization steps, leading to complex impurity profiles that are difficult to separate. These impurities can persist through subsequent alkylation steps, compromising the quality of the final active pharmaceutical ingredient and necessitating costly recrystallization procedures. The reliance on harsh conditions and toxic reagents also complicates regulatory compliance, making it challenging for manufacturers to maintain consistent quality across large batches.
The Novel Approach
The methodology outlined in CN1161350C presents a transformative solution by reimagining the sequence of functional group transformations. Instead of direct cyanation of a halide, this novel approach constructs the cyano functionality via an aldehyde intermediate, which is subsequently converted to an oxime and dehydrated. This strategic shift eliminates the need for cuprous cyanide entirely, thereby removing the burden of heavy metal clearance from the process workflow. The use of manganese dioxide for the oxidation of the hydroxymethyl group to the aldehyde is a mild and selective transformation that preserves the integrity of the sensitive phthalan ring system. By optimizing the reduction and cyclization steps using phosphoric acid, the process achieves high conversion rates with minimal byproduct formation. This streamlined approach not only enhances the overall yield but also significantly simplifies the workup procedures, making it an ideal candidate for cost reduction in API manufacturing where efficiency is paramount.
Mechanistic Insights into Reductive Cyclization and Oxidation
The core of this synthetic strategy lies in the precise manipulation of oxidation states and ring-closure mechanisms. The process begins with the reduction of a dicarboxylic acid derivative using lithium aluminum hydride, a powerful reducing agent that converts the carboxyl groups into primary alcohols while simultaneously reducing the ketone to a secondary alcohol. This triol intermediate is crucial, as it sets the stage for the subsequent intramolecular etherification. The cyclization is facilitated by acid catalysis, where the protonation of one of the benzylic hydroxyl groups creates a good leaving group, allowing the neighboring phenolic oxygen to attack and close the five-membered phthalan ring. This step is highly sensitive to reaction conditions, but the patent specifies optimized parameters that favor the formation of the desired isomer over potential regioisomers. The resulting hydroxymethyl-phthalan is then selectively oxidized to the aldehyde, a transformation that requires careful control to prevent over-oxidation to the carboxylic acid.
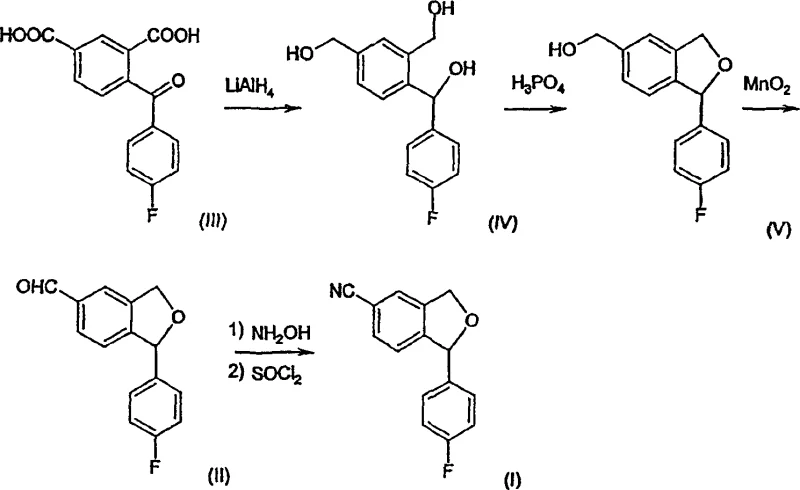
Following the formation of the aldehyde, the introduction of the nitrogen functionality is achieved through a classic oxime formation followed by dehydration. The reaction with hydroxylamine generates the oxime intermediate, which is then treated with thionyl chloride to effect dehydration and yield the nitrile group. This two-step sequence is advantageous because it avoids the use of toxic cyanide salts, aligning with modern green chemistry principles. The mechanism ensures that the cyano group is installed at the correct position on the aromatic ring with high fidelity, minimizing the formation of positional isomers that could act as genotoxic impurities. For R&D teams, understanding this mechanistic pathway is essential for troubleshooting potential scale-up issues and ensuring that the impurity profile remains within acceptable limits. The robustness of this chemistry supports the commercial scale-up of complex pharmaceutical intermediates, providing a stable foundation for large-scale production.
How to Synthesize 1-(4-Fluorophenyl)-1,3-dihydroisobenzofuran-5-carbonitrile Efficiently
Implementing this synthetic route requires careful attention to reagent quality and reaction parameters to maximize efficiency and safety. The process begins with the reduction step, where strict temperature control is necessary to manage the exotherm associated with lithium aluminum hydride. Following the reduction, the cyclization step utilizes phosphoric acid, which offers a balance between acidity and non-oxidizing properties, ensuring the stability of the intermediate. The oxidation step with manganese dioxide is typically performed in dichloromethane, a solvent that facilitates the suspension of the oxidant and easy filtration of the spent reagent. Finally, the conversion to the nitrile involves handling thionyl chloride, requiring appropriate gas scrubbing systems to manage hydrogen chloride evolution. Detailed standard operating procedures are essential to replicate the high yields reported in the patent literature.
- Reduction of 4-fluorobenzophenone-2',4'-dicarboxylic acid using LiAlH4 to form the triol intermediate.
- Acid-catalyzed cyclization using H3PO4 to form the hydroxymethyl-phthalan derivative.
- Oxidation with MnO2 followed by conversion to the nitrile using hydroxylamine and thionyl chloride.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers substantial benefits for procurement managers and supply chain heads looking to optimize their vendor networks. The elimination of cuprous cyanide not only reduces raw material costs but also significantly lowers the environmental compliance burden associated with heavy metal waste disposal. This translates into substantial cost savings over the lifecycle of the product, making the manufacturing process more economically sustainable. Furthermore, the use of common reagents like lithium aluminum hydride and phosphoric acid ensures that supply chain continuity is maintained, as these materials are widely available from multiple global suppliers. This reduces the risk of production delays caused by the shortage of specialized catalysts, enhancing the overall reliability of the supply chain. The simplified purification process also means faster batch turnover times, allowing manufacturers to respond more agilely to market demand fluctuations.
- Cost Reduction in Manufacturing: The removal of expensive transition metal catalysts and the associated clearance steps drastically reduces the operational expenditure per kilogram of product. By avoiding the need for specialized scavengers to remove copper residues, the process simplifies the downstream processing workflow. This efficiency gain allows for a more competitive pricing structure without compromising on quality standards. Additionally, the high yields achieved in the cyclization and oxidation steps minimize raw material waste, further contributing to overall cost effectiveness. These factors combined create a compelling economic case for adopting this technology in large-scale production facilities.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals rather than bespoke catalysts mitigates the risk of supply disruptions. In a volatile global market, having a synthesis route that depends on widely available reagents ensures that production can continue uninterrupted. This stability is crucial for maintaining long-term contracts with pharmaceutical clients who require guaranteed delivery schedules. The robustness of the chemistry also means that technology transfer between manufacturing sites is smoother, as the process does not require highly specialized equipment or unique operational expertise. This flexibility strengthens the resilience of the supply chain against external shocks.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing unit operations that are standard in the fine chemical industry. The absence of highly toxic cyanide salts simplifies the safety protocols required for handling, reducing the regulatory hurdles for plant approval. Waste streams are easier to treat, as they do not contain heavy metals that require specialized precipitation or ion exchange processes. This alignment with environmental regulations facilitates faster permitting and reduces the long-term liability associated with waste management. Consequently, the process supports sustainable manufacturing practices that are increasingly demanded by global pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic pathway. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages for potential partners. Understanding these details is key to evaluating the feasibility of integrating this technology into existing production lines.
Q: What are the key advantages of the new synthetic route described in CN1161350C?
A: The novel route avoids the use of cuprous cyanide for introducing the cyano group, instead utilizing an aldehyde intermediate converted via oxime dehydration. This significantly reduces heavy metal contamination risks and simplifies purification, leading to higher purity profiles suitable for stringent pharmaceutical standards.
Q: How does this process impact the scalability of citalopram production?
A: By utilizing robust reagents like LiAlH4 for reduction and MnO2 for oxidation, the process eliminates sensitive catalytic steps that often hinder scale-up. The crystallization steps described allow for efficient isolation of intermediates, ensuring consistent quality and yield during commercial scale-up of complex pharmaceutical intermediates.
Q: Is this method suitable for producing enantiomerically pure S-citalopram?
A: While the patent describes the racemic synthesis, the high-purity intermediate generated provides an ideal substrate for subsequent chiral resolution or asymmetric synthesis steps. The clean impurity profile minimizes interference during enantiomeric separation, supporting the production of high-purity antidepressants.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Citalopram Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to maintain competitiveness in the global pharmaceutical market. Our technical team has extensively analyzed patent CN1161350C and possesses the expertise to implement this efficient pathway for the production of high-purity citalopram intermediates. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory to plant is seamless and efficient. Our rigorous QC labs and stringent purity specifications guarantee that every batch meets the exacting standards required by top-tier pharmaceutical companies. We are committed to delivering products that not only meet but exceed regulatory expectations.
We invite you to collaborate with us to leverage this technology for your supply chain optimization. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a reliable supply of critical intermediates backed by deep technical expertise and a commitment to continuous improvement. Let us help you secure a stable and cost-effective supply of high-quality pharmaceutical materials.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
