Advanced Synthetic Route for Citalopram Intermediates Enhancing Commercial Scalability
Advanced Synthetic Route for Citalopram Intermediates Enhancing Commercial Scalability
The pharmaceutical industry continuously seeks robust and safe manufacturing processes for high-volume antidepressants like Citalopram. Patent CN1138768C discloses a groundbreaking preparation method for cyanophthalofluoroaniline, a critical intermediate in the synthesis of this selective serotonin reuptake inhibitor. Unlike legacy processes that rely on hazardous heavy metal catalysts, this invention leverages a sophisticated sequence of Grignard reactions on functionalized benzo[c]furanone derivatives. By shifting the synthetic strategy towards nucleophilic additions on esters or amides, the technology significantly mitigates environmental risks while maintaining high structural fidelity. This report analyzes the technical merits of this approach, offering strategic insights for R&D directors and supply chain leaders aiming to optimize their API intermediate sourcing strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cyanophthalide derivatives has been plagued by reliance on toxic reagents and complex multi-step sequences that hinder efficient scale-up. Prior art, such as that described in US4136193, typically involves the reaction of 5-bromo derivatives with copper cyanide to introduce the essential nitrile functionality. This approach presents severe drawbacks, including the generation of heavy metal waste streams that require expensive remediation and strict regulatory compliance measures. Furthermore, the use of copper cyanide often necessitates harsh reaction conditions that can compromise the integrity of sensitive functional groups, leading to lower overall yields and difficult purification profiles. The reliance on brominated starting materials also introduces cost volatility, as halogenated precursors are generally more expensive and less atom-economical than their carboxylic acid counterparts.
The Novel Approach
The methodology outlined in CN1138768C represents a paradigm shift by utilizing 5-carboxy-2-benzo[c]furanone derivatives as the foundational building blocks. Instead of introducing the cyano group via substitution, this route constructs the complex molecular architecture through sequential carbon-carbon bond formations using Grignard reagents. This strategy allows for the use of safer, more abundant starting materials and avoids the immediate handling of cyanide sources until the final dehydration steps, if even required in that specific form. The process enables the formation of the quaternary carbon center and the installation of the fluorophenyl and dimethylaminopropyl side chains in a controlled manner. By focusing on ester or amide intermediates, the chemistry becomes more amenable to standard organic processing equipment, reducing the need for specialized containment facilities required for cyanide handling.
![General structure of Formula IV starting material showing the benzo[c]furanone core with ester or amide functionality](/insights/img/citalopram-intermediate-grignard-synthesis-pharma-supplier-20260315094732-01.png)
Mechanistic Insights into Sequential Grignard Addition and Cyclization
The core innovation lies in the dual Grignard addition strategy performed on the lactone or ester functionality of the benzo[c]furanone core. Initially, the compound of Formula IV, where R1 is a C1-6 alkyl group and X is oxygen or nitrogen, undergoes nucleophilic attack by a 4-halofluorophenyl magnesium halide. This reaction opens the lactone ring or attacks the ester carbonyl to form a ketone-alcohol intermediate, effectively installing the crucial 4-fluorophenyl moiety. The reaction conditions are meticulously controlled, typically maintained below 5°C in anhydrous THF, to prevent side reactions such as over-addition or decomposition of the sensitive organometallic species. This step is critical for establishing the stereochemical environment around the future quaternary center.
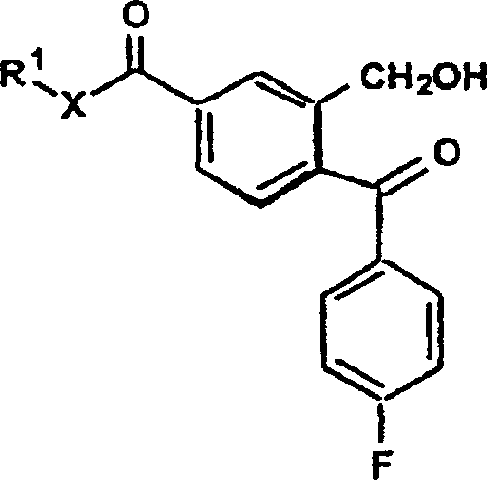
Following the first addition, the reaction mixture is treated with a second Grignard reagent, specifically 3-halo-N,N-dimethylpropylamine magnesium halide. This second nucleophilic attack targets the remaining carbonyl or ketone functionality, resulting in the formation of a diol or amino-alcohol intermediate (Formula V). The subsequent ring closure is achieved through acid-catalyzed dehydration or via the formation of labile esters like mesylates followed by base treatment. This cyclization step reconstructs the isobenzofuran ring system, locking the side chains into the correct spatial arrangement. Finally, if the starting material was an amide or ester, a dehydration step using agents like thionyl chloride converts the carbonyl-derived group into the requisite nitrile, yielding the target cyanophthalofluoroaniline.
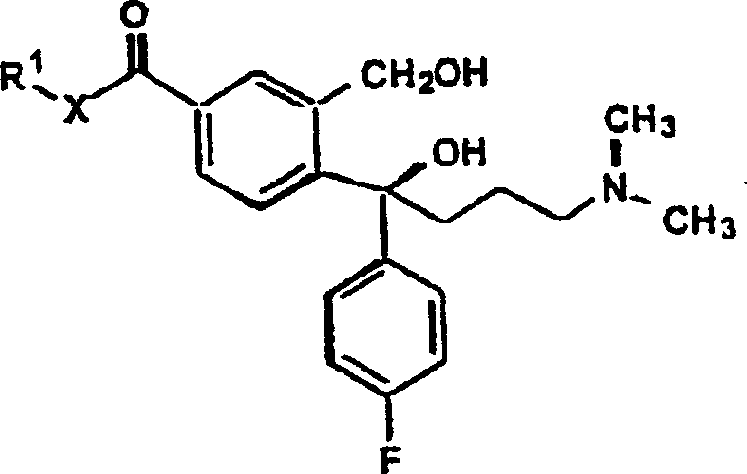
How to Synthesize Cyanophthalofluoroaniline Efficiently
The synthesis of this high-value antidepressant intermediate requires precise control over stoichiometry and temperature to ensure high purity and yield. The patent details a robust protocol starting from commercially available 5-carboxy-2-benzo[c]furanone, which is first converted into a stable ester or amide derivative. The detailed standardized synthesis steps are provided in the guide below.
- Prepare the starting benzo[c]furanone derivative (Formula IV) by esterification or amidation of 5-carboxy-2-benzo[c]furanone.
- Perform sequential Grignard reactions: first with 4-halofluorophenyl magnesium halide, then with 3-halo-N,N-dimethylpropylamine magnesium halide.
- Effect ring closure under acidic or basic conditions to form the isobenzofuran core, followed by dehydration to install the cyano group.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this Grignard-based synthesis offers tangible logistical and financial benefits beyond mere chemical elegance. By eliminating the dependency on copper cyanide, manufacturers can drastically reduce the costs associated with hazardous waste disposal and environmental compliance auditing. The use of common solvents like THF and standard reagents like thionyl chloride simplifies the supply chain, as these materials are globally sourced with high availability, reducing the risk of production stoppages due to raw material shortages. Furthermore, the potential for telescoping the Grignard steps into a one-pot process significantly reduces reactor occupancy time and solvent consumption, leading to substantial operational cost savings.
- Cost Reduction in Manufacturing: The elimination of expensive and toxic copper cyanide reagents removes a major cost driver from the bill of materials. Additionally, the avoidance of heavy metal catalysts means that downstream purification does not require expensive scavenging resins or complex filtration steps to meet residual metal specifications. This streamlining of the workup procedure reduces solvent usage and labor hours, directly translating to a lower cost of goods sold (COGS) for the final API intermediate without compromising quality standards.
- Enhanced Supply Chain Reliability: The starting materials for this route, particularly 5-carboxy-2-benzo[c]furanone and simple alkyl halides, are commodity chemicals with stable global supply networks. Unlike specialized brominated intermediates which may have limited suppliers, the precursors for this method are produced by multiple vendors worldwide. This diversification of the supply base mitigates the risk of single-source bottlenecks and ensures consistent delivery schedules, which is critical for maintaining continuous API production lines in a regulated pharmaceutical environment.
- Scalability and Environmental Compliance: The reaction conditions described, primarily involving low-temperature Grignard additions in THF, are well-understood and easily scalable in standard stainless steel reactors. The process avoids the generation of heavy metal sludge, simplifying effluent treatment and aligning with increasingly stringent green chemistry regulations. This environmental compatibility facilitates faster regulatory approvals for new manufacturing sites and reduces the long-term liability associated with hazardous waste storage and transport.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic route. These answers are derived directly from the experimental data and claims within the patent documentation to ensure accuracy for technical decision-makers.
Q: How does this new method improve safety compared to traditional cyanophthalide synthesis?
A: Traditional methods often utilize copper cyanide for cyanation, which poses significant toxicity and waste disposal challenges. This novel route utilizes sequential Grignard additions on safe ester or amide precursors, eliminating the need for heavy metal cyanide salts and reducing hazardous waste generation.
Q: What are the key impurities controlled in this Grignard-based process?
A: The process controls impurities through precise temperature management during the exothermic Grignard additions (keeping below 5°C to 10°C) and by utilizing specific dehydration agents like thionyl chloride for the final cyanation step, ensuring high chemical purity suitable for pharmaceutical applications.
Q: Is this synthetic route suitable for large-scale commercial production?
A: Yes, the route is designed for scalability. It uses commercially available starting materials like 5-carboxy-2-benzo[c]furanone and allows for potential one-pot processing of the Grignard steps, which simplifies operations and enhances throughput for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Citalopram Intermediate Supplier
The synthetic pathway detailed in CN1138768C demonstrates significant potential for producing high-purity cyanophthalide intermediates with improved safety and efficiency profiles. As a leading CDMO partner, NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with state-of-the-art cryogenic reactors capable of handling the low-temperature Grignard steps described, ensuring that the stringent purity specifications required for antidepressant APIs are consistently met. Our rigorous QC labs employ advanced analytical techniques to monitor impurity profiles throughout the synthesis, guaranteeing product quality that exceeds industry standards.
We invite global pharmaceutical partners to collaborate with us on optimizing their supply chains for Citalopram and related compounds. By leveraging our technical expertise, you can achieve significant process improvements and cost efficiencies. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to support your regulatory filings and commercial planning.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
