Advanced One-Pot Synthesis of N-Acyl Indoles for High-Value Pharmaceutical Intermediates
Advanced One-Pot Synthesis of N-Acyl Indoles for High-Value Pharmaceutical Intermediates
The structural motif of the indole ring is ubiquitous in medicinal chemistry, serving as the core scaffold for a vast array of bioactive molecules ranging from anti-inflammatory agents like Indomethacin to potent anti-tumor compounds. As depicted in the visual overview of indole-based drugs, the versatility of this heterocyclic system drives continuous demand for efficient synthetic methodologies. 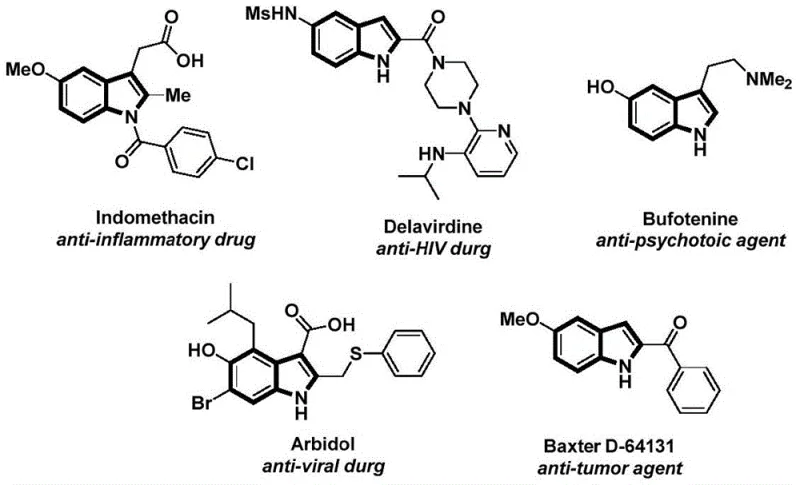 Recent advancements in organic synthesis have sought to streamline the construction of these complex architectures, particularly through carbonylation reactions that introduce essential acyl functionalities. Patent CN112898192B introduces a groundbreaking preparation method for N-acyl indole compounds that addresses critical safety and efficiency bottlenecks inherent in traditional carbonylation processes. This novel approach utilizes a palladium-catalyzed cascade reaction involving 2-alkynyl anilines and aryl iodides, employing a solid carbon monoxide surrogate to achieve high yields under mild conditions. For R&D directors and procurement specialists in the fine chemical sector, this technology represents a significant leap forward in the reliable production of high-purity pharmaceutical intermediates, offering a robust pathway that balances operational simplicity with broad substrate tolerance.
Recent advancements in organic synthesis have sought to streamline the construction of these complex architectures, particularly through carbonylation reactions that introduce essential acyl functionalities. Patent CN112898192B introduces a groundbreaking preparation method for N-acyl indole compounds that addresses critical safety and efficiency bottlenecks inherent in traditional carbonylation processes. This novel approach utilizes a palladium-catalyzed cascade reaction involving 2-alkynyl anilines and aryl iodides, employing a solid carbon monoxide surrogate to achieve high yields under mild conditions. For R&D directors and procurement specialists in the fine chemical sector, this technology represents a significant leap forward in the reliable production of high-purity pharmaceutical intermediates, offering a robust pathway that balances operational simplicity with broad substrate tolerance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-acyl indoles via carbonylation has been plagued by significant operational hazards and logistical complexities. Traditional methods often rely on the direct use of carbon monoxide (CO) gas, which is highly toxic, flammable, and requires specialized high-pressure equipment and rigorous safety protocols to handle safely on an industrial scale. Furthermore, conventional routes frequently involve multi-step sequences where the indole core is constructed first, followed by a separate acylation step, leading to increased waste generation, lower overall atom economy, and higher production costs due to additional purification stages. The reliance on gaseous reagents also introduces variability in reaction kinetics and mass transfer limitations, which can compromise reproducibility and yield consistency in large-scale manufacturing. These factors collectively create substantial barriers for supply chain managers seeking to secure a steady flow of complex intermediates without incurring excessive safety overheads or facing regulatory hurdles associated with hazardous gas usage.
The Novel Approach
The methodology disclosed in patent CN112898192B fundamentally reimagines this synthetic challenge by replacing hazardous CO gas with 1,3,5-tricarboxylic acid phenol ester (TFBen), a stable and easy-to-handle solid carbon monoxide surrogate. This innovation allows the entire transformation to proceed in a convenient one-pot fashion within standard glassware or reactors, drastically simplifying the operational workflow. The process initiates with a palladium-catalyzed coupling between the aryl iodide and the CO surrogate to generate an acyl-palladium intermediate in situ, which then reacts with the 2-alkynyl aniline. Subsequently, the addition of silver oxide triggers an oxidative cyclization that seamlessly closes the indole ring. This tandem strategy not only eliminates the need for high-pressure gas infrastructure but also merges two distinct synthetic steps into a single continuous operation, thereby reducing solvent consumption, minimizing work-up time, and enhancing the overall throughput of the manufacturing process.
Mechanistic Insights into Palladium-Catalyzed Carbonylative Cyclization
The elegance of this synthesis lies in its carefully orchestrated catalytic cycle, which leverages the unique reactivity of palladium complexes to forge multiple bonds in a single vessel. As illustrated in the general reaction scheme, the cycle begins with the oxidative addition of the aryl iodide to the Pd(0) species, forming an aryl-palladium(II) intermediate. 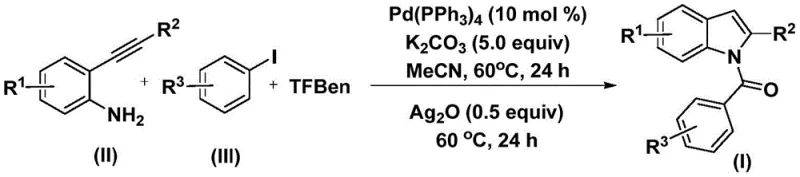 Crucially, the TFBen additive decomposes under the reaction conditions to release carbon monoxide, which immediately inserts into the palladium-carbon bond to generate a reactive acyl-palladium species. This acyl intermediate then undergoes nucleophilic attack by the amino group of the 2-alkynyl aniline, followed by reductive elimination to yield an amide precursor. The second phase of the reaction involves the introduction of silver oxide, which acts as an oxidant to facilitate the intramolecular cyclization of the alkyne moiety onto the nitrogen atom, ultimately delivering the N-acyl indole product with high regioselectivity. This mechanistic pathway ensures that the acyl group is installed precisely at the nitrogen position while simultaneously constructing the five-membered pyrrole ring, showcasing a high degree of chemo- and regio-control.
Crucially, the TFBen additive decomposes under the reaction conditions to release carbon monoxide, which immediately inserts into the palladium-carbon bond to generate a reactive acyl-palladium species. This acyl intermediate then undergoes nucleophilic attack by the amino group of the 2-alkynyl aniline, followed by reductive elimination to yield an amide precursor. The second phase of the reaction involves the introduction of silver oxide, which acts as an oxidant to facilitate the intramolecular cyclization of the alkyne moiety onto the nitrogen atom, ultimately delivering the N-acyl indole product with high regioselectivity. This mechanistic pathway ensures that the acyl group is installed precisely at the nitrogen position while simultaneously constructing the five-membered pyrrole ring, showcasing a high degree of chemo- and regio-control.
From an impurity control perspective, this mechanism offers distinct advantages by minimizing the formation of side products commonly associated with stepwise syntheses. The use of a solid CO source ensures a steady, controlled release of carbon monoxide, preventing the local concentration spikes that can lead to homocoupling of the aryl iodide or other palladium-black precipitation issues. Furthermore, the choice of acetonitrile as the solvent provides an optimal polarity balance that stabilizes the charged intermediates while keeping all organic components in solution, thereby suppressing premature precipitation or aggregation that could trap impurities. The compatibility of this system with a wide range of electronic environments on the aromatic rings—tolerating electron-donating groups like methoxy and methyl as well as electron-withdrawing halogens—demonstrates the robustness of the catalytic cycle against steric and electronic perturbations, ensuring consistent purity profiles across diverse substrate libraries.
How to Synthesize N-Acyl Indole Efficiently
The practical execution of this synthesis is designed for maximum ease of use, requiring only standard laboratory equipment and commercially available reagents. The protocol involves charging a reaction vessel with the palladium catalyst, base, CO surrogate, and substrates, followed by a two-stage heating regimen. Detailed standardized synthesis steps are provided in the guide below to ensure reproducible results.
- Combine palladium catalyst, potassium carbonate, TFBen, 2-alkynyl aniline, and aryl iodide in acetonitrile.
- Heat the mixture at 60°C for 24 hours to facilitate the initial carbonylation and amide formation.
- Add silver oxide and continue heating at 60°C for another 24 hours to complete the cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology translates into tangible strategic benefits that extend far beyond simple yield improvements. By shifting from a gas-phase reagent to a solid surrogate, the process inherently de-risks the supply chain, removing the dependency on specialized gas suppliers and the logistical complexities of transporting and storing pressurized carbon monoxide cylinders. This transition significantly enhances operational continuity, as solid reagents like TFBen have longer shelf lives and are less susceptible to supply disruptions compared to compressed gases. Moreover, the simplification of the reaction setup reduces the capital expenditure required for reactor certification and safety monitoring systems, allowing manufacturing facilities to allocate resources more efficiently towards capacity expansion rather than compliance overhead.
- Cost Reduction in Manufacturing: The elimination of high-pressure equipment and the consolidation of two synthetic steps into a single one-pot operation lead to substantial reductions in both CAPEX and OPEX. Without the need for specialized gas handling infrastructure, facilities can utilize standard glass-lined or stainless steel reactors, lowering the barrier to entry for production. Additionally, the high atom economy of the carbonylation step minimizes raw material waste, while the reduced number of isolation and purification stages lowers solvent consumption and energy usage for distillation. These cumulative efficiencies drive down the cost per kilogram of the final API intermediate, providing a competitive pricing advantage in the global market without compromising on quality standards.
- Enhanced Supply Chain Reliability: The use of readily available starting materials such as 2-alkynyl anilines and aryl iodides ensures a robust upstream supply base, as these commodities are produced by numerous chemical manufacturers worldwide. The stability of the solid CO surrogate further mitigates the risk of production stoppages due to reagent degradation or delivery delays. This reliability is critical for maintaining just-in-time inventory levels and meeting tight delivery schedules for downstream pharmaceutical clients. The method's tolerance for various functional groups also means that a single production line can be flexibly adapted to manufacture a diverse portfolio of N-acyl indole derivatives, enhancing the agility of the supply chain to respond to changing market demands.
- Scalability and Environmental Compliance: Scaling this reaction from gram to ton scale is straightforward due to the absence of gas-liquid mass transfer limitations that typically hinder carbonylation reactions. The homogeneous nature of the reaction mixture in acetonitrile ensures consistent heat and mass transfer even in large vessels, facilitating a smooth technology transfer from R&D to pilot and commercial plants. From an environmental standpoint, the process generates less hazardous waste by avoiding the use of toxic gases and reducing solvent volumes. The simplified work-up procedure, which primarily involves filtration and chromatography, aligns well with green chemistry principles, helping manufacturers meet increasingly stringent environmental regulations and sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis technology. These answers are derived directly from the experimental data and mechanistic studies presented in the patent documentation to provide clarity for potential partners.
Q: What is the primary advantage of using TFBen over carbon monoxide gas?
A: TFBen (1,3,5-tricarboxylic acid phenol ester) acts as a solid carbon monoxide surrogate, eliminating the need for handling toxic and hazardous CO gas cylinders, thereby significantly improving laboratory and plant safety.
Q: What is the role of Silver Oxide (Ag2O) in this reaction?
A: Silver oxide is added in the second stage of the reaction to promote the oxidative cyclization of the intermediate amide, closing the indole ring to form the final N-acyl indole structure.
Q: Can this method tolerate diverse functional groups?
A: Yes, the patent demonstrates excellent substrate compatibility, successfully tolerating various substituents such as halogens (F, Cl, Br), alkyl groups (Me, tBu), and alkoxy groups (OMe) on both the aniline and aryl iodide components.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acyl Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug development timelines. Our team of expert chemists has thoroughly evaluated the potential of this palladium-catalyzed carbonylation route and is fully prepared to leverage it for your specific project needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of N-acyl indole intermediate we deliver meets the highest industry standards for potency and impurity profiles.
We invite you to collaborate with us to unlock the full potential of this innovative chemistry for your pipeline. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to reach out today to discuss your project specifics,索取 specific COA data for our existing catalog, or request detailed route feasibility assessments for novel targets. Let us be your trusted partner in navigating the complexities of fine chemical synthesis and delivering value to your organization.
