Advanced Docetaxel Synthesis: Enhancing Commercial Viability Through Selective Protection
Advanced Docetaxel Synthesis: Enhancing Commercial Viability Through Selective Protection
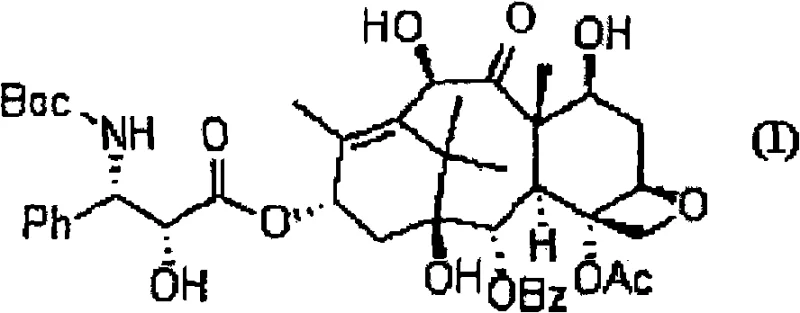
The pharmaceutical industry continuously seeks robust synthetic routes for high-value oncology agents, and patent CN101558051A presents a transformative approach to the preparation of docetaxel. This technology addresses the longstanding bottleneck in taxane synthesis: the selective protection of the 7- and 10-hydroxyl groups on the 10-deacetylbaccatin III core. Traditional methods often suffer from poor regioselectivity, leading to complex mixtures that are difficult and expensive to purify. By introducing a novel protection strategy using nitro-substituted benzoyl halides, specifically 3,5-dinitrobenzoyl chloride, this invention achieves unprecedented selectivity and yield. For R&D directors and procurement managers, this represents a critical opportunity to optimize the cost reduction in API manufacturing while securing a more reliable supply chain for this essential chemotherapy agent.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
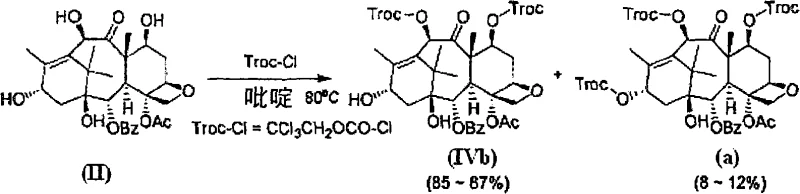
Historically, the semi-synthesis of docetaxel has been plagued by inefficiencies in the initial protection steps. As illustrated in prior art schemes, such as the use of 2,2,2-trichloroethyl chloroformate (Troc-Cl), the reaction lacks sufficient specificity. Data from European Patent No. 0253738 indicates that reacting 10-deacetylbaccatin III with Troc-Cl yields the desired 7,10-protected product in only 85-87% yield, while generating 8-12% of the unwanted 7,10,13-tri-protected byproduct. This impurity profile necessitates rigorous purification, typically involving column chromatography, which is notoriously difficult to scale and results in significant material loss. Furthermore, alternative methods using dichloroacetyl or trichloroacetyl groups perform even worse, with yields dropping to 40-45% and creating complex impurity profiles that compromise the overall process economics and timeline.
The Novel Approach
In stark contrast, the methodology disclosed in CN101558051A utilizes electron-deficient benzoyl halides to achieve exceptional regiocontrol. By employing 3,5-dinitrobenzoyl chloride, the process selectively targets the 7- and 10-hydroxyl positions while leaving the 13-hydroxyl group largely untouched. Experimental data from the patent demonstrates a remarkable 97.1% yield of the desired 7,10-bis-protected intermediate, with the tri-protected byproduct reduced to a negligible 0.9%. Crucially, this high-purity intermediate can be isolated through simple recrystallization from methanol, completely bypassing the need for chromatographic separation. This shift from chromatography to crystallization is a game-changer for commercial scale-up of complex pharmaceutical intermediates, drastically simplifying the workflow and enhancing throughput capabilities for large-scale production facilities.
Mechanistic Insights into Nitro-Benzoyl Selective Protection
The success of this synthetic route lies in the electronic properties of the nitro-substituted benzoyl group. The strong electron-withdrawing nature of the nitro groups increases the electrophilicity of the carbonyl carbon in the benzoyl chloride, facilitating a faster acylation rate. However, the steric bulk combined with the specific electronic environment of the baccatin III core dictates the regioselectivity. The 7- and 10-hydroxyl groups are more accessible and reactive under these specific conditions compared to the sterically hindered 13-hydroxyl group. This kinetic differentiation allows for the preferential formation of the 7,10-diester. Following protection, the coupling with the oxazolidine side chain proceeds efficiently using standard condensing agents like dicyclohexylcarbodiimide (DCC). The subsequent acid-catalyzed ring opening of the oxazolidine moiety releases the active side chain without disturbing the sensitive taxane core, ensuring the structural integrity required for biological activity.
Impurity control is inherently built into this mechanism. Because the formation of the tri-protected byproduct is suppressed to less than 1%, the downstream burden on purification units is minimized. In conventional processes, the tri-protected species often co-elutes or requires multiple passes to remove, dragging down the overall yield. Here, the crystallization step acts as a powerful purification engine, leveraging the solubility differences between the desired di-protected species and the minor impurities. This results in a final intermediate purity of 98.5% or higher before the coupling step even begins. For quality assurance teams, this means a much cleaner impurity profile entering the final stages of synthesis, reducing the risk of carryover contaminants in the final high-purity pharmaceutical intermediates and ensuring compliance with stringent regulatory standards.
How to Synthesize Docetaxel Efficiently
The synthesis of docetaxel via this patented route involves a streamlined four-step sequence that prioritizes yield and operational simplicity. The process begins with the selective acylation of 10-deacetylbaccatin III, followed by coupling with a specialized oxazolidine derivative, ring opening, and final deprotection. Each step has been optimized to maximize conversion and minimize waste, making it an ideal candidate for industrial adoption. The detailed standardized synthesis steps, including specific reagent ratios, temperature controls, and workup procedures, are outlined in the guide below to assist technical teams in replicating this high-efficiency pathway.
- React 10-deacetylbaccatin III with 3,5-dinitrobenzoyl chloride to selectively protect 7- and 10-hydroxyl groups.
- Couple the protected intermediate with an oxazolidine derivative using a condensing agent like DCC.
- Perform acid-catalyzed ring opening of the oxazolidine side chain followed by base-mediated deprotection.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic method offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the purification process. By eliminating column chromatography in favor of crystallization, manufacturers can significantly reduce solvent consumption, waste generation, and processing time. This directly translates to lower operating expenditures and a smaller environmental footprint, aligning with modern green chemistry initiatives. Furthermore, the high yield of the key intermediate ensures that expensive starting materials like 10-deacetylbaccatin III are utilized with maximum efficiency, reducing the cost per kilogram of the final active pharmaceutical ingredient.
- Cost Reduction in Manufacturing: The elimination of chromatographic purification steps removes a major cost driver associated with silica gel, large volumes of elution solvents, and extended processing times. Additionally, the high selectivity of the 3,5-dinitrobenzoyl protection minimizes the loss of valuable taxane core material to byproducts. This efficiency gain means that less raw material is required to produce the same amount of final product, leading to substantial cost savings in the overall manufacturing budget without compromising on quality or safety standards.
- Enhanced Supply Chain Reliability: Processes that rely on crystallization are inherently more robust and scalable than those dependent on chromatography. Chromatography columns can channel, clog, or vary in performance, leading to batch-to-batch inconsistencies. In contrast, crystallization is a unit operation that scales linearly and predictably. This reliability ensures a steady flow of intermediates, reducing lead time for high-purity APIs and mitigating the risk of supply disruptions. Suppliers adopting this method can offer more consistent delivery schedules, which is critical for maintaining uninterrupted production of life-saving oncology medications.
- Scalability and Environmental Compliance: The reduced solvent load and waste generation associated with this method simplify waste treatment protocols. Handling large volumes of chromatographic eluents poses significant logistical and environmental challenges at the multi-ton scale. By switching to a crystallization-based workflow, facilities can operate with smaller reactor footprints and reduced solvent recovery loads. This ease of scale-up facilitates the rapid transition from pilot plant to commercial production, allowing companies to respond quickly to market demand while maintaining strict adherence to environmental regulations and sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this docetaxel synthesis method. These insights are derived directly from the experimental data and process descriptions within the patent, providing a clear understanding of the operational advantages and chemical principles involved. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing infrastructure.
Q: Why is 3,5-dinitrobenzoyl chloride preferred over Troc chloride for docetaxel synthesis?
A: 3,5-dinitrobenzoyl chloride offers significantly higher selectivity (97.1% yield) for 7,10-protection compared to Troc chloride (85-87%), minimizing difficult-to-remove tri-protected byproducts.
Q: How does this method improve purification efficiency?
A: The novel intermediate can be purified via simple recrystallization from methanol, eliminating the need for costly and time-consuming column chromatography required in conventional methods.
Q: What are the scalability benefits of this synthetic route?
A: By avoiding chromatographic purification and utilizing robust crystallization steps, the process is highly amenable to commercial scale-up, ensuring consistent supply continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Docetaxel Intermediate Supplier
The technological advancements detailed in patent CN101558051A underscore the potential for more efficient and cost-effective production of docetaxel. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to leverage such innovative synthetic routes for our clients. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory successes are seamlessly translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of intermediate meets the highest global standards, providing our partners with the confidence they need to advance their drug development pipelines.
We invite you to collaborate with us to explore how this optimized synthesis can benefit your specific project requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can support your long-term supply chain objectives.
