Advanced Carbonylation Strategy for Scalable Isatoic Anhydride Derivative Manufacturing
Introduction to Next-Generation Isatoic Anhydride Synthesis
The chemical landscape for producing heterocyclic intermediates is undergoing a significant transformation driven by the need for safer, more sustainable, and cost-effective methodologies. A pivotal advancement in this domain is detailed in patent CN102863399A, which discloses a novel synthetic route for isatoic anhydride derivatives. This technology leverages a dual-metal catalytic system involving Palladium and Copper, or alternatively Iron and Copper, to facilitate the carbonylation of aniline derivatives. Unlike legacy processes that rely on hazardous reagents, this approach utilizes carbon monoxide as a C1 building block under relatively mild conditions. For R&D directors and process chemists, this represents a critical opportunity to redesign synthetic pathways for key pharmaceutical and agrochemical scaffolds. The ability to construct the benzoxazine-2,4-dione core directly from readily available anilines simplifies the supply chain and reduces the environmental footprint of manufacturing operations.
The significance of isatoic anhydride extends far beyond simple academic interest; it serves as a versatile precursor for a vast array of bioactive molecules, including quinazolinones and benzodiazepines. By adopting the methodology outlined in CN102863399A, manufacturers can achieve high purity profiles essential for regulatory compliance in drug substance production. The reaction operates efficiently at temperatures ranging from 20°C to 100°C, with optimal performance often observed around 60°C, thereby minimizing thermal degradation of sensitive functional groups. This technical breakthrough aligns perfectly with the industry's shift towards green chemistry principles, offering a robust alternative that balances operational safety with high synthetic efficiency.
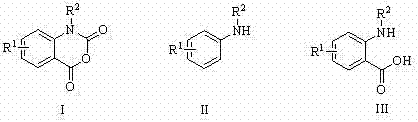
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of isatoic anhydride derivatives has been plagued by significant safety and environmental challenges. The most common traditional route involves the reaction of anthranilic acid derivatives with highly toxic chloroformates or triphosgene. Triphosgene, while a solid substitute for gaseous phosgene, still poses severe risks upon decomposition, releasing phosgene gas which is a potent pulmonary irritant and chemical warfare agent. Furthermore, alternative oxidative methods utilizing chromium trioxide or organic peracids introduce heavy metal waste streams and explosion hazards associated with peroxide handling. These conventional processes often require stringent temperature control, corrosive-resistant equipment, and complex waste treatment protocols to neutralize acidic byproducts and heavy metals. The operational complexity translates directly into higher capital expenditure and increased liability for chemical manufacturers.
The Novel Approach
In stark contrast, the catalytic carbonylation method described in the patent data offers a streamlined and inherently safer pathway. By employing a Pd/Cu or Fe/Cu catalytic system, the reaction bypasses the need for stoichiometric amounts of toxic acylating agents. Instead, it utilizes carbon monoxide, which, while requiring careful handling, is a standard commodity chemical in many fine chemical plants. The reaction proceeds through a catalytic cycle that inserts CO into the amine bond followed by intramolecular cyclization, effectively building the heterocyclic ring in a single step. This atom-economical approach minimizes waste generation and eliminates the formation of stoichiometric salt byproducts typical of phosgene reactions. Moreover, the use of mild solvents like acetonitrile and moderate temperatures ensures that the process is energy-efficient and compatible with a wide range of substrates, including those with sensitive electronic properties.
Mechanistic Insights into Pd/Cu-Catalyzed Carbonylative Cyclization
The mechanistic underpinning of this transformation involves a sophisticated interplay between the palladium center and the copper co-catalyst. The cycle likely initiates with the oxidative addition of the aniline derivative or an in-situ generated aryl halide species to the Pd(0) center, although the exact activation mode of the C-H or C-N bond may vary depending on the specific additives used. Once the organopalladium species is formed, carbon monoxide coordinates to the metal center and undergoes migratory insertion to form an acyl-palladium intermediate. This step is crucial as it introduces the carbonyl carbon necessary for the anhydride structure. The presence of copper salts, such as copper acetate, plays a vital role in facilitating the subsequent cyclization and regeneration of the active catalyst. Copper acts as a Lewis acid or an oxidant, promoting the nucleophilic attack of the adjacent oxygen or nitrogen functionality onto the acyl group, closing the ring to form the isatoic anhydride core.
Furthermore, the inclusion of iodide additives, such as potassium iodide (KI) or tetrabutylammonium iodide, is mechanistically significant. These additives likely assist in the generation of more reactive aryl-iodide intermediates or stabilize the palladium species against aggregation, thereby maintaining high catalytic turnover numbers. The patent data explicitly highlights that reactions without iodide additives suffer from drastically reduced yields, dropping from over 90% to as low as 40% in certain cases. This suggests that the halide ligand environment around the palladium center is critical for optimizing the electronic properties of the catalyst. Understanding these nuances allows process chemists to fine-tune the reaction conditions, ensuring consistent quality and minimizing the formation of side products such as ureas or simple amides which could complicate downstream purification.
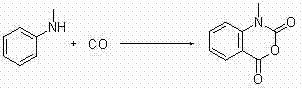
How to Synthesize N-Methylisatoic Anhydride Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and purity. The general protocol involves charging a reactor with the aniline substrate, palladium acetate, copper acetate, and an iodide source in acetonitrile. The system is then pressurized with carbon monoxide, optionally mixed with oxygen to facilitate catalyst re-oxidation if a Pd(0)/Pd(II) cycle is operative. Detailed standard operating procedures regarding stoichiometry, pressure ramping, and work-up protocols are essential for reproducible results on a commercial scale. The following guide outlines the critical steps derived from the patent examples to ensure successful execution of this transformation.
- Combine aniline derivative, palladium catalyst (e.g., Pd(OAc)2), copper salt (e.g., Cu(OAc)2), and iodide additive in acetonitrile.
- Pressurize the reactor with Carbon Monoxide (1-10 atm) or a CO/O2 mixture and heat to 20-100°C.
- Monitor reaction progress via TLC, then filter inorganic salts and purify the crude product via recrystallization or column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic carbonylation technology offers compelling strategic advantages beyond mere technical feasibility. The primary benefit lies in the drastic simplification of the raw material supply chain. By eliminating the dependence on triphosgene or chloroformates, companies can avoid the logistical nightmares and regulatory hurdles associated with transporting and storing Schedule 1 chemical weapons precursors. This shift not only reduces insurance premiums and security costs but also mitigates the risk of supply disruptions caused by strict regulatory crackdowns on toxic reagents. Additionally, the use of commodity chemicals like carbon monoxide and simple metal salts ensures a stable and cost-effective supply base, insulating the manufacturing process from volatile market fluctuations associated with specialized reagents.
- Cost Reduction in Manufacturing: The transition from stoichiometric toxic reagents to a catalytic system fundamentally alters the cost structure of production. While palladium is a precious metal, its usage at catalytic levels (e.g., 5 mol%) combined with inexpensive copper salts results in a significantly lower cost per kilogram of product compared to purchasing expensive phosgene equivalents. Furthermore, the elimination of extensive waste treatment processes for heavy metals and acidic byproducts leads to substantial operational savings. The simplified work-up procedure, often requiring only filtration of inorganic salts followed by crystallization, reduces solvent consumption and labor hours, driving down the overall cost of goods sold (COGS) for these high-value intermediates.
- Enhanced Supply Chain Reliability: Reliability is paramount in the pharmaceutical supply chain, and this method enhances it by utilizing robust and widely available starting materials. Aniline derivatives are commodity chemicals produced on a massive scale globally, ensuring consistent availability and competitive pricing. The mild reaction conditions also reduce the wear and tear on reactor vessels, extending equipment lifespan and minimizing unplanned maintenance downtime. By adopting a process that is less sensitive to extreme conditions, manufacturers can achieve higher batch-to-batch consistency, ensuring that delivery schedules are met without the delays often caused by complex safety interlocks or equipment failures associated with highly corrosive processes.
- Scalability and Environmental Compliance: Scaling chemical processes often exposes hidden inefficiencies, but this carbonylation route is inherently scalable due to its homogeneous nature and manageable exotherm. The ability to run the reaction at near-atmospheric pressures or moderate pressures (1-10 atm) means that standard stainless steel reactors can be utilized without the need for exotic high-pressure autoclaves. From an environmental perspective, the reduction in hazardous waste generation aligns with increasingly stringent global environmental regulations. This proactive compliance reduces the risk of fines and shutdowns, ensuring long-term operational continuity and enhancing the company's reputation as a sustainable manufacturer among eco-conscious clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the safety advantages of this carbonylation method over traditional phosgene routes?
A: Traditional methods utilize highly toxic phosgene or triphosgene, requiring specialized containment and posing severe health risks. This patented carbonylation route utilizes carbon monoxide under controlled pressure with catalytic metal salts, significantly reducing acute toxicity hazards and corrosion issues associated with chloroformates.
Q: Can this synthesis method accommodate diverse functional groups on the aniline ring?
A: Yes, the method demonstrates broad substrate scope. It successfully tolerates various substituents including halogens (Cl, Br, F), nitro groups, alkyl chains, alkoxy groups, and ester functionalities, making it suitable for synthesizing complex pharmaceutical intermediates without extensive protecting group strategies.
Q: What represents the optimal catalytic system for maximum yield?
A: The patent data indicates that a combination of Palladium acetate (5 mol%) and Copper acetate (200 mol%) with Potassium Iodide as an additive in acetonitrile solvent provides superior yields, often exceeding 80% under mild temperatures around 60°C.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isatoic Anhydride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain a competitive edge in the global market. Our technical team has extensively evaluated the carbonylation protocols described in CN102863399A and possesses the expertise to translate these laboratory findings into robust commercial processes. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of isatoic anhydride derivative meets the highest industry standards for pharmaceutical and agrochemical applications.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage potential partners to contact us directly to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing capabilities can drive value and efficiency for your organization. Let us be your trusted partner in navigating the complexities of fine chemical synthesis.
