Revolutionizing Metalloporphyrin Production With Advanced One-Step Catalytic Technology For Global Markets
The chemical industry is constantly evolving, driven by the relentless pursuit of more efficient, cost-effective, and environmentally sustainable synthetic pathways. In this context, patent CN1944447A represents a significant breakthrough in the field of macrocyclic compound synthesis, specifically addressing the longstanding challenges associated with producing metalloporphyrins. This intellectual property introduces a novel one-step methodology that fundamentally deviates from traditional multi-stage protocols, utilizing a sophisticated mixed solvent system composed of C1 to C8 straight-chain fatty acids and nitrobenzene derivatives. By integrating the solvent and catalyst functions into a single phase, this technology not only streamlines the operational workflow but also dramatically enhances the thermodynamic favorability of the reaction. For R&D directors and procurement specialists seeking a reliable metalloporphyrin supplier, understanding the mechanistic underpinnings of this patent is crucial for evaluating its potential impact on large-scale manufacturing and cost reduction in fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of metalloporphyrins has been plagued by inefficiencies inherent in conventional two-step methodologies, which often rely on单一 solvent systems that lack the necessary versatility to optimize reaction conditions dynamically. Traditional approaches typically involve an initial condensation of pyrrole and aromatic aldehydes to form free base porphyrins, followed by a separate metallation step, a process that is not only time-consuming but also prone to significant material loss at each stage. Furthermore, the reliance on single solvents restricts the ability to fine-tune the acidity and polarity of the reaction medium, leading to suboptimal yields that rarely exceed 40% and often result in complex mixtures of by-products that are difficult to separate. The necessity of adding zinc salts as a template center in many legacy processes further complicates the purification workflow, introducing additional costs associated with heavy metal removal and waste disposal that burden the overall economic viability of the production line.
The Novel Approach
In stark contrast to these legacy constraints, the innovative method disclosed in the patent data leverages a synergistic mixed solvent environment to achieve a direct, one-step conversion of raw materials into high-purity metalloporphyrins. By employing a blend of straight-chain fatty acids and nitrobenzene derivatives, the process creates a reaction medium that simultaneously serves as the solvent, the acid catalyst, and the oxidizing agent, thereby eliminating the need for external additives that complicate the chemical landscape. This integrated approach allows for precise control over the reaction parameters, such as pH and boiling point, enabling the synthesis to proceed under milder conditions that minimize thermal degradation and polymerization side reactions. The result is a robust manufacturing protocol that consistently delivers yields ranging from 40% to 60%, surpassing the maximum efficiencies reported in existing literature while significantly reducing the operational complexity and resource consumption required for commercial scale-up of complex catalysts.
Mechanistic Insights into Mixed Solvent Catalytic Cyclization
The core of this technological advancement lies in the unique physicochemical properties of the mixed solvent system, which facilitates a highly efficient cyclization and metallation cascade within a single reactor vessel. The straight-chain fatty acids, typically ranging from C1 to C4, provide the necessary acidic environment to catalyze the condensation of pyrrole and substituted benzaldehyde, while their varying chain lengths allow for the modulation of solvent polarity to match the solubility profiles of different substituents. Simultaneously, the inclusion of nitrobenzene or its derivatives serves a dual purpose: it acts as a potent oxidizing agent to drive the aromatization of the porphyrin ring and enhances the solubility of both reactants and intermediate species, preventing premature precipitation that could halt the reaction progress. This delicate balance of acidity, oxidation potential, and solvation power ensures that the transition metal ions, such as iron, cobalt, or manganese, are effectively incorporated into the porphyrin core without the need for a preliminary zinc template, thus simplifying the coordination chemistry involved.
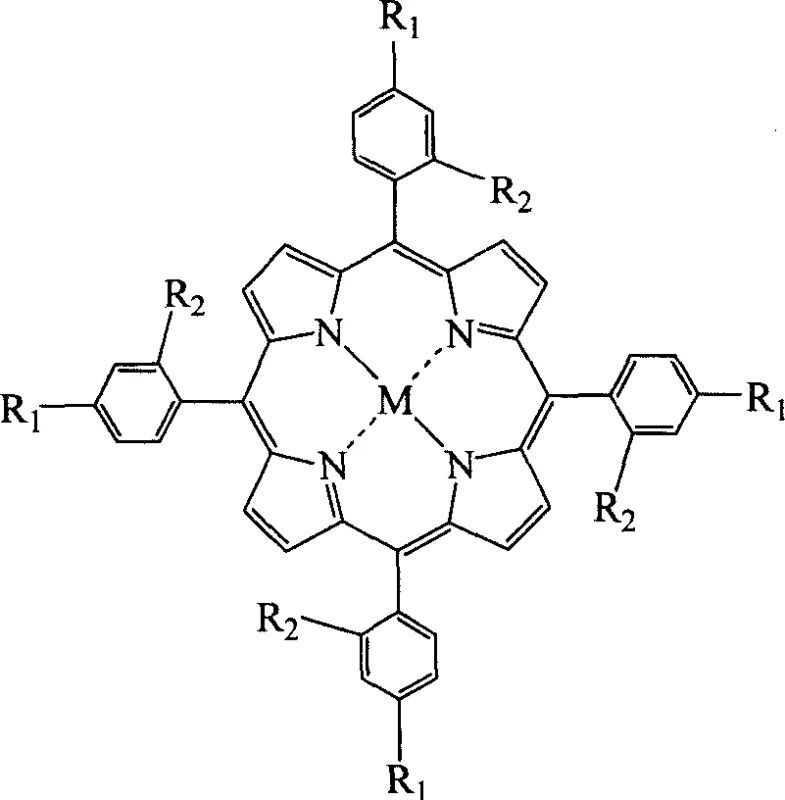
Furthermore, the mechanism inherently supports superior impurity control, which is a critical parameter for R&D directors focused on the purity and impurity profile of advanced materials. The tunable nature of the mixed solvent system allows for the selective dissolution of by-products, keeping them in the solution phase while the desired metalloporphyrin product crystallizes out upon cooling and methanol addition. This self-purifying characteristic reduces the burden on downstream processing units, such as chromatography or extensive recrystallization steps, which are often bottlenecks in the production of high-purity OLED material or pharmaceutical intermediates. By minimizing the formation of high molecular weight polymeric by-products through controlled boiling points and mild reaction temperatures, the process ensures that the final product meets stringent quality specifications with minimal intervention, thereby enhancing the overall reliability of the supply chain for sensitive electronic or medical applications.
How to Synthesize Tetraphenyl Metalloporphyrin Efficiently
Implementing this synthesis route requires a precise understanding of the reagent ratios and thermal conditions to maximize the benefits of the mixed solvent system. The process begins with the preparation of the ternary solvent mixture, followed by the sequential addition of the aldehyde, pyrrole, and metal salt under controlled heating. While the general principles are straightforward, the specific optimization of acid ratios and reflux times is critical to adapting the protocol for different metal centers and substituent patterns. The detailed standardized synthesis steps see the guide below for specific operational parameters.
- Prepare a mixed solvent system comprising C1-C8 straight-chain fatty acids and nitrobenzene derivatives.
- Add substituted benzaldehyde, pyrrole, and soluble transition metal salts in specific molar ratios.
- Heat the mixture to reflux for 0.5 to 1 hour, then cool and purify the resulting metalloporphyrin.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this one-step synthesis technology offers profound advantages that extend beyond mere chemical efficiency to impact the bottom line and supply chain resilience significantly. By consolidating what was traditionally a multi-stage process into a single operational unit, manufacturers can drastically reduce the capital expenditure required for reactor trains and the associated labor costs for monitoring and transferring materials between stages. The elimination of the zinc salt template step not only simplifies the chemical workflow but also removes the logistical and regulatory burdens associated with sourcing, handling, and disposing of heavy metal templates, leading to substantial cost savings in raw material procurement and waste management. Additionally, the shortened reaction time of approximately 0.5 to 1 hour enhances the throughput capacity of existing facilities, allowing for faster turnover rates and improved responsiveness to market demand fluctuations without the need for significant infrastructure expansion.
- Cost Reduction in Manufacturing: The economic benefits of this process are primarily driven by the simplification of the synthetic route and the reduction in auxiliary material consumption. By removing the requirement for additional soluble zinc salts and minimizing the use of complex purification solvents, the overall variable cost per kilogram of product is significantly lowered. The high yield efficiency, reaching up to 60%, means that less raw material is wasted in the form of unreacted starting materials or irrecoverable by-products, directly translating to improved material utilization rates. Furthermore, the energy consumption is optimized due to the shorter reflux times and lower boiling points of the fatty acid solvents compared to traditional high-boiling organic solvents, contributing to a leaner and more cost-effective manufacturing model that enhances competitiveness in the global market.
- Enhanced Supply Chain Reliability: Supply chain stability is greatly improved by the robustness of the mixed solvent system, which utilizes readily available commodity chemicals such as fatty acids and nitrobenzene derivatives that are less susceptible to geopolitical supply disruptions than specialized catalysts. The one-step nature of the reaction reduces the number of critical process parameters that need to be controlled, thereby lowering the risk of batch failures and ensuring consistent product availability for downstream customers. This reliability is crucial for maintaining continuous production schedules in industries where downtime can lead to significant financial penalties, making this technology a preferred choice for securing long-term contracts for high-purity catalysts and intermediates.
- Scalability and Environmental Compliance: Scaling this process from laboratory to industrial production is facilitated by the use of common organic acids which are easier to handle and recycle compared to corrosive mineral acids or toxic chlorinated solvents often used in older methods. The reduced generation of heavy metal waste, owing to the absence of zinc templating, simplifies compliance with increasingly stringent environmental regulations regarding effluent discharge and hazardous waste disposal. This environmental advantage not only mitigates regulatory risk but also aligns with the sustainability goals of modern chemical enterprises, making the technology attractive for green chemistry initiatives and enhancing the corporate social responsibility profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this advanced metalloporphyrin synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages for potential partners and stakeholders. Understanding these details is essential for making informed decisions about integrating this methodology into your existing production frameworks.
Q: What is the primary advantage of the mixed solvent system in metalloporphyrin synthesis?
A: The mixed solvent system acts as both solvent and catalyst, allowing for tunable acidity and polarity which significantly improves yield compared to single solvent methods.
Q: Does this method require a zinc salt template?
A: No, unlike conventional two-step methods, this novel approach eliminates the need for additional soluble zinc salts as a template center.
Q: What is the typical reaction time for this synthesis?
A: The reaction is highly efficient, typically requiring only 0.5 to 1 hour of heating under reflux conditions to achieve completion.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Metalloporphyrin Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this one-step synthesis technology and possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring such innovations to the global market. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to ensure that every batch of metalloporphyrin meets stringent purity specifications, consistently delivering the high-quality standards demanded by the pharmaceutical and electronic materials sectors. We are committed to leveraging our technical expertise to optimize this patented route, ensuring that the theoretical benefits of yield and efficiency are fully realized in practical, large-scale manufacturing environments.
We invite forward-thinking organizations to collaborate with us to explore how this technology can drive value in their specific applications. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can support your supply chain objectives and accelerate your product development timelines.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
