Revolutionizing Flurbiprofen Production: A Deep Dive into Cobalt-Catalyzed Cross-Coupling Technology
The pharmaceutical industry is constantly seeking more efficient pathways to produce non-steroidal anti-inflammatory drugs (NSAIDs), and a recent breakthrough detailed in Chinese patent CN114195634A offers a compelling solution for the synthesis of 2-biphenylyl propionic acid compounds, most notably Flurbiprofen. This patent discloses a novel preparation method that utilizes a cobalt-catalyzed coupling reaction between a specific Grignard reagent and a 2-halopropionate salt. Unlike traditional methods that rely on expensive transition metals or suffer from poor selectivity, this innovation leverages earth-abundant cobalt catalysts in conjunction with amine ligands to achieve exceptional yields and purity. The structural integrity of the target molecule, Flurbiprofen, is critical for its efficacy as a potent COX inhibitor, and the ability to synthesize it with minimal impurities is a major advancement for API manufacturers.
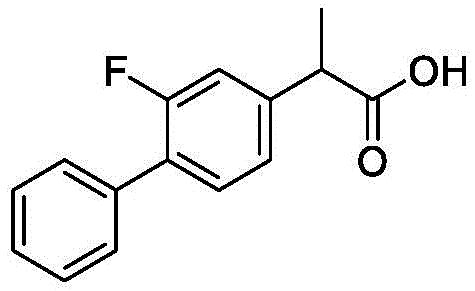
For R&D directors evaluating process chemistry, the significance of this patent lies in its ability to bypass the limitations of prior art. Historically, the synthesis of 2-biphenylyl propionic acid derivatives has been plagued by complex multi-step sequences and the use of scarce starting materials. The new methodology streamlines this by directly coupling a biphenyl Grignard species with a halogenated propionate. This not only shortens the synthetic route but also drastically improves the atom economy of the process. By focusing on the interaction between the cobalt center and the organic substrates, chemists can now access high-purity intermediates that were previously difficult to isolate without extensive chromatographic purification, thereby setting a new standard for process efficiency in NSAID manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the industrial production of Flurbiprofen faced significant hurdles regarding yield and byproduct formation. As highlighted in the background of CN114195634A, earlier methods such as those disclosed in U.S. Pat. No. 4,395,9364A relied on Grignard coupling but suffered from severe side reactions. Specifically, the coupling of 4-bromo-2-fluorobiphenyl Grignard reagents with 2-halopropionates frequently resulted in the formation of di-substituted byproducts. These unwanted species, structurally similar to the target but with altered pharmacological profiles, are notoriously difficult to separate and represent a direct loss of yield. In repeated experiments of these older protocols, side reaction rates often exceeded 30%, capping the overall yield at less than 60%. Furthermore, alternative routes utilizing phenylboron reagents and palladium catalysts, while effective in some contexts, introduce prohibitive costs due to the price of palladium and the complexity of removing trace metal residues to meet regulatory standards.
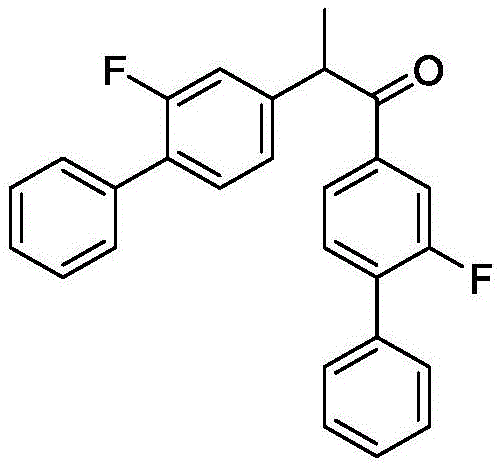
The Novel Approach
The methodology presented in CN114195634A fundamentally alters the reaction landscape by introducing a tailored cobalt catalytic system. Instead of relying on uncatalyzed or palladium-mediated pathways, this approach employs divalent or trivalent cobalt salts—such as CoCl2, CoSO4, or Co(acac)3—complexed with amine ligands like TMEDA or HMTA. This catalytic ensemble facilitates a highly selective cross-coupling reaction that effectively suppresses the formation of the di-substituted byproducts seen in legacy processes. The result is a dramatic improvement in process metrics, with experimental data showing yields consistently above 90% and HPLC purities reaching 99% or higher. This leap in performance is achieved without the need for exotic reagents; the process utilizes readily available sodium or magnesium 2-halopropionates, making the supply chain far more robust and less susceptible to the volatility associated with precious metal markets.
Mechanistic Insights into Cobalt-Catalyzed Cross-Coupling
The core of this technological advancement lies in the unique mechanistic pathway enabled by the cobalt-amine catalyst system. In the proposed mechanism, the cobalt species acts as a mediator for the transmetallation between the Grignard reagent (derived from the biphenyl halide) and the 2-halopropionate electrophile. The presence of the amine ligand is crucial, as it stabilizes the cobalt center in a coordination environment that favors the desired cross-coupling over homocoupling or multiple substitutions. The reaction likely proceeds through a radical or organometallic cycle where the cobalt catalyst lowers the activation energy for the carbon-carbon bond formation. This allows the reaction to proceed efficiently at relatively mild temperatures, typically ranging from -20°C to 80°C, with optimal results observed around -5°C to 0°C during the addition phase. Such control over the reaction kinetics is essential for maintaining the stereochemical integrity and preventing the degradation of sensitive functional groups on the biphenyl ring.
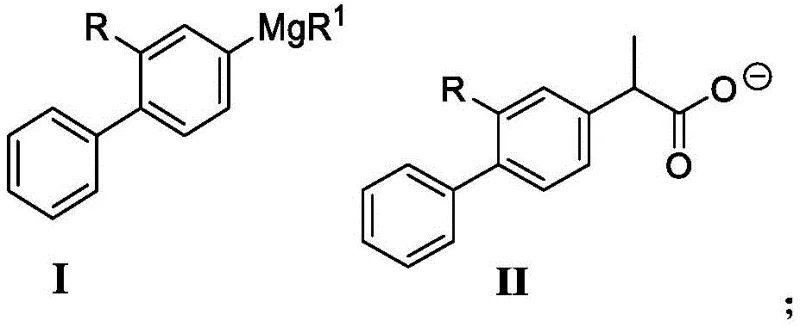
From an impurity control perspective, the choice of catalyst and ligand combination plays a pivotal role in defining the final product quality. The patent data indicates that specific combinations, such as Co(acac)3 with TMEDA, provide superior selectivity compared to other transition metals like iron or copper, which yielded significantly lower conversion rates in comparative examples. The mechanism inherently minimizes the generation of regioisomers and oligomeric side products. Furthermore, the subsequent acidification step, where the 2-biphenylpropionate salt is converted to the free acid using hydrochloric acid, serves as an effective purification checkpoint. By adjusting the pH to 3-4, the product precipitates or partitions cleanly into the organic phase, leaving behind inorganic salts and polar impurities. This inherent ease of purification reduces the reliance on energy-intensive distillation or complex chromatography, aligning perfectly with green chemistry principles and cost-effective manufacturing goals.
How to Synthesize 2-Biphenylyl Propionic Acid Efficiently
The synthesis protocol outlined in the patent is designed for operational simplicity and scalability, making it an attractive candidate for technology transfer. The process begins with the in situ generation of the Grignard reagent, followed immediately by the catalytic coupling step, minimizing the handling of unstable intermediates. The use of common ether solvents like tetrahydrofuran (THF) ensures compatibility with existing reactor infrastructure in most fine chemical plants. Detailed standardized operating procedures for this synthesis, including precise stoichiometric ratios and temperature profiles, are critical for reproducing the high yields reported in the patent examples. For process engineers looking to implement this route, understanding the nuances of the exothermic Grignard formation and the controlled addition of the coupling partners is key to success.
- Preparation of the Grignard Reagent: React 4-bromo-2-fluorobiphenyl with magnesium chips in an ether solvent (preferably THF) with a catalytic amount of iodine at 55-65°C.
- Cobalt-Catalyzed Coupling: Add the Grignard solution to a mixture of 2-halopropionate (e.g., sodium 2-bromopropionate), a cobalt catalyst (e.g., Co(acac)3), and an amine ligand (e.g., TMEDA) at -5 to 0°C.
- Acidification and Isolation: Quench the reaction with hydrochloric acid to pH 3-4, extract with ethyl acetate, and recrystallize from methanol/water to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift to this cobalt-catalyzed methodology represents a strategic opportunity to optimize both cost structures and supply reliability. The elimination of palladium catalysts removes a significant variable cost component, as palladium prices are historically volatile and subject to geopolitical supply constraints. By substituting this with cobalt salts, which are more abundant and stable in price, manufacturers can achieve substantial cost savings in raw material procurement. Additionally, the use of simple alkali metal salts like sodium 2-bromopropionate replaces more complex and expensive coupling partners like phenylboronic acids. This simplification of the bill of materials (BOM) not only lowers direct material costs but also reduces the logistical burden of sourcing specialized reagents from limited suppliers.
- Cost Reduction in Manufacturing: The economic benefits of this process extend beyond raw material savings. The high yield (>90%) and high purity (>99%) significantly reduce the cost of goods sold (COGS) by minimizing waste and the need for reprocessing. In traditional low-yield processes, a significant portion of the budget is consumed by the disposal of failed batches and the solvents required for extensive purification. By achieving near-quantitative conversion with minimal byproducts, this method maximizes the output per batch, effectively lowering the unit cost of production. Furthermore, the mild reaction conditions reduce energy consumption associated with heating and cooling, contributing to overall operational efficiency and a smaller carbon footprint for the manufacturing facility.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commodity chemicals. The starting materials, such as 4-bromo-2-fluorobiphenyl and magnesium, are widely produced and available from multiple global vendors, reducing the risk of single-source dependency. The robustness of the reaction also means that minor variations in raw material quality are less likely to cause batch failures, ensuring consistent production schedules. This reliability is crucial for meeting the strict delivery timelines demanded by downstream API manufacturers and pharmaceutical companies. The ability to scale this process from kilogram to multi-ton quantities without changing the fundamental chemistry provides a clear path for long-term supply security.
- Scalability and Environmental Compliance: From an environmental and safety standpoint, this process offers distinct advantages for large-scale operations. The avoidance of heavy metal contaminants like palladium simplifies the waste treatment process and ensures that the final API meets stringent residual metal specifications without additional scavenging steps. The solvents used, primarily THF and ethyl acetate, are well-understood and can be efficiently recovered and recycled, further enhancing the sustainability profile of the operation. The simplified work-up procedure, involving straightforward extraction and crystallization, reduces the volume of hazardous waste generated. This alignment with environmental regulations facilitates smoother regulatory approvals and reduces the long-term liability associated with chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cobalt-catalyzed synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in patent CN114195634A, offering a clear picture of the technology's capabilities and limitations. Understanding these details is essential for stakeholders evaluating the feasibility of adopting this new method for commercial production.
Q: How does the cobalt-catalyzed method improve yield compared to traditional palladium routes?
A: Traditional methods often suffer from di-substitution byproducts and low yields (<60%). The novel cobalt-catalyzed route described in CN114195634A suppresses these side reactions, achieving yields exceeding 90% with HPLC purity >99%.
Q: What are the key cost drivers eliminated in this new synthesis pathway?
A: This method eliminates the need for expensive palladium catalysts and complex phenylboron reagents. It utilizes readily available cobalt salts and simple alkali metal 2-halopropionates, significantly reducing raw material costs.
Q: Is this process scalable for industrial production of Flurbiprofen?
A: Yes, the patent explicitly highlights industrial suitability. The reaction operates under mild temperatures (-20 to 80°C) and uses common solvents like THF, making it highly adaptable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Flurbiprofen Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the cobalt-catalyzed synthesis route for 2-biphenylyl propionic acid compounds. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this patented laboratory methodology into robust, commercial-scale manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high yields and purity levels demonstrated in the patent are maintained at an industrial level. We operate stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of intermediate meets the exacting standards required for pharmaceutical applications, providing our clients with peace of mind regarding product quality and regulatory compliance.
We invite pharmaceutical companies and API manufacturers to collaborate with us to leverage this advanced technology for their Flurbiprofen supply chains. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you optimize your manufacturing strategy, reduce costs, and secure a reliable supply of high-quality pharmaceutical intermediates for the global market.
