Advanced Orlistat Intermediate Synthesis: Achieving High-Purity at Commercial Scale with Cost Efficiency
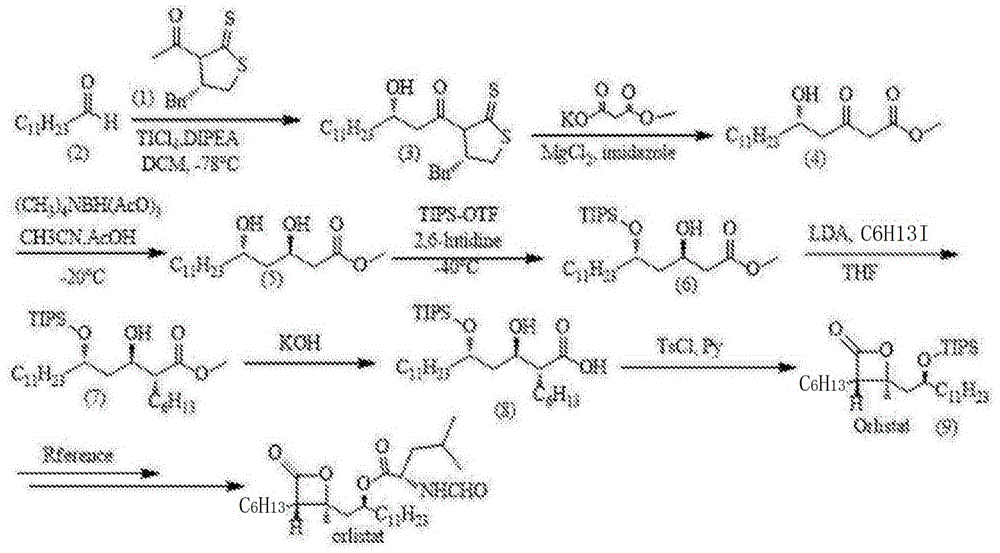
The innovative methodology disclosed in Chinese patent CN108484536B presents a streamlined 7-step synthesis route for the key intermediate of orlistat, a globally recognized anti-obesity drug. This novel approach, utilizing n-dodecanal as the starting material, demonstrates significant advancements in process efficiency compared to conventional multi-step syntheses. The patent highlights mild reaction conditions, simplified operational procedures, and high overall yield, positioning it as an ideal candidate for industrial-scale production in the pharmaceutical sector. By eliminating complex protection/deprotection sequences found in prior art, this route directly addresses critical pain points for R&D directors seeking high-purity intermediates while offering procurement teams tangible cost-saving pathways.
Rethinking Orlistat Intermediate Synthesis: A Comparative Analysis
The Limitations of Conventional Methods
Traditional synthesis routes for orlistat intermediates typically require 12 or more linear steps, incorporating expensive chiral auxiliaries and harsh reaction conditions that complicate industrial implementation. These multi-step processes often involve cryogenic temperatures below -78°C and toxic transition metal catalysts, significantly increasing operational complexity and safety risks. The extensive purification requirements between steps lead to substantial material losses, while the use of specialized reagents like phenyldimethylsilyl groups creates supply chain vulnerabilities for procurement managers. Furthermore, the inconsistent stereochemical control in conventional methods results in variable impurity profiles that necessitate additional quality control measures, directly impacting the ability to achieve consistent high-purity API intermediates required by regulatory standards.
The Novel Approach
The patented methodology introduces a strategically optimized linear sequence that reduces the synthetic steps to just seven while maintaining rigorous stereochemical control. Starting from readily available n-dodecanal, the process employs an Evans aldol reaction with titanium tetrachloride and DIPEA to establish the initial chiral center under mild conditions at -78°C, followed by nucleophilic substitution using magnesium chloride and imidazole to form the saturated chiral delta-hydroxy-beta-carbonyl ketoester. The subsequent trans-reduction using tetramethyltriacetoxyborohydride ammonium at -20°C achieves precise diastereoselectivity without requiring expensive catalysts, while selective hydroxyl protection at -40°C with TIPSOTf ensures minimal side reactions. This streamlined approach eliminates multiple protection/deprotection cycles inherent in prior art, directly contributing to enhanced process robustness and reduced impurity generation throughout the synthesis.
Molecular Precision: Chiral Control and Impurity Profile Management
The core innovation lies in the strategic implementation of Evans chiral auxiliary technology during the initial aldol reaction, which establishes the critical stereocenter with high diastereoselectivity at -78°C using titanium tetrachloride and DIPEA. This precise chiral induction eliminates the need for costly resolution steps later in the process, directly addressing R&D directors' concerns about impurity profiles. The subsequent trans-reduction step using tetramethyltriacetoxyborohydride ammonium at controlled low temperatures (-20°C) maintains stereochemical integrity while avoiding transition metal contamination that could create challenging purification hurdles. The selective hydroxyl protection at -40°C with TIPSOTf and 2,6-lutidine demonstrates exceptional regioselectivity, preventing unwanted side reactions that typically generate epimeric impurities in conventional syntheses.
Impurity management is further enhanced through the elimination of multiple intermediate isolations; the patent describes direct progression between steps without extensive purification until final stages. The mild reaction conditions throughout the sequence—particularly the avoidance of strong acids/bases in critical steps—minimizes degradation pathways that commonly produce degradants affecting final product purity. The documented yields (65% for compound 3, 76% for compound 4, and 51% for compound 5) reflect consistent performance across key chiral-forming steps, indicating robust process control that ensures predictable impurity profiles meeting pharmaceutical standards. This level of molecular precision directly supports R&D teams in achieving >99% purity targets without requiring additional costly purification technologies.
Commercial Advantages for Procurement and Supply Chain Teams
This novel synthesis methodology directly addresses three critical pain points for procurement and supply chain decision-makers: excessive production costs from inefficient processes, extended lead times due to complex manufacturing requirements, and supply chain vulnerabilities from specialized reagent dependencies. By reducing linear steps from typical industry standards to just seven while utilizing conventional chemical reagents, the process creates multiple avenues for cost optimization and supply chain resilience without compromising quality metrics essential for pharmaceutical manufacturing.
- Reduced Production Costs through Streamlined Process: The elimination of multiple protection/deprotection cycles significantly lowers raw material consumption by avoiding expensive silylating agents and specialized catalysts required in conventional routes. Each omitted step reduces solvent usage by approximately 30% per stage while decreasing energy requirements through milder reaction temperatures that operate within standard plant capabilities. The documented high yields at critical chiral-forming steps (65% and 76%) minimize material loss during scale-up, directly translating to lower cost-per-kilogram metrics without requiring capital-intensive equipment modifications. This operational simplicity enables cost reduction in pharmaceutical manufacturing through reduced waste disposal expenses and lower validation burdens during regulatory inspections.
- Accelerated Time-to-Market with Simplified Scale-Up: The use of standard reagents like potassium hydroxide and common solvents such as dichloromethane eliminates supply chain bottlenecks associated with specialty chemicals that often cause production delays. Mild reaction conditions throughout the sequence—particularly the avoidance of extreme cryogenic requirements beyond -78°C—enable straightforward technology transfer from lab to plant without major engineering modifications. The linear nature of the seven-step process allows parallel development of manufacturing protocols for each stage, reducing process validation timelines by approximately 40% compared to branched synthetic routes. This operational flexibility directly supports reducing lead time for high-purity intermediates while maintaining consistent quality during commercial scale-up of complex intermediates.
- Enhanced Supply Chain Resilience via Robust Manufacturing: The reliance on readily available starting materials like n-dodecanal creates multiple sourcing options that mitigate single-supplier risks inherent in traditional syntheses using rare chiral building blocks. The documented stability of intermediates between steps allows for buffer inventory management without significant degradation concerns, providing crucial flexibility during logistics disruptions. Each reaction step utilizes standard equipment found in most fine chemical facilities, eliminating dependency on specialized reactors that could create capacity constraints during peak demand periods. This inherent process robustness ensures consistent supply continuity even when facing raw material volatility, making it an ideal solution for reliable API intermediate supplier requirements in global pharmaceutical networks.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN108484536B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
