Optimizing Docetaxel Production: A Novel Protection Strategy for High-Purity API Intermediates
Optimizing Docetaxel Production: A Novel Protection Strategy for High-Purity API Intermediates
The global demand for next-generation antineoplastic agents continues to drive innovation in the synthesis of complex taxanes, specifically Docetaxel. Patent CN101353334A introduces a transformative methodology for preparing Docetaxel that addresses longstanding challenges in yield, purity, and process safety. This technical insight report analyzes the proprietary synthetic route which utilizes a novel C-7 and C-10 protection strategy involving substituted benzyl chloroformates, coupled with a unique 1-ethoxyethyl side-chain protection. For R&D directors and supply chain leaders, understanding this shift from traditional trichloroethyl or organolithium-based methods is crucial for securing a reliable pharmaceutical intermediates supplier capable of delivering high-purity materials at scale.
The significance of this patent lies in its ability to bypass the formation of persistent impurities that plague conventional syntheses. By replacing harsh reagents with catalytic esterification and mild hydrogenolysis, the process not only enhances the chemical profile of the intermediate but also aligns with modern green chemistry principles. This report details the mechanistic advantages and commercial implications of adopting this refined pathway for the commercial scale-up of complex API intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
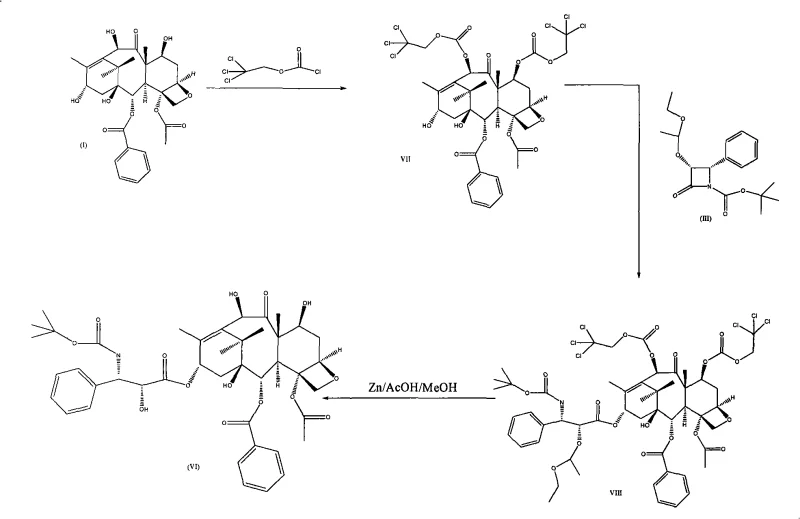
Historically, the synthesis of Docetaxel from 10-deacetyl baccatin III has been hindered by the use of 2,2,2-trichloroethyl chloroformate for protecting the C-7 and C-10 hydroxyl groups. As illustrated in the prior art, this approach frequently leads to the formation of problematic byproducts, specifically compound IX, which possesses a trichloroethyl carbonate moiety at the C-13 position. The appearance of byproduct IX not only drastically influences reaction yield but also renders the purification of the desired intermediate extremely difficult, often requiring extensive chromatography that is impractical for ton-scale manufacturing. Furthermore, the deprotection of trichloroethyl groups typically requires violent reaction conditions, such as zinc dust in acidic media, which can induce degradation of the sensitive oxetane ring and other functional groups on the taxane core.
Alternative methods, such as those described by Nicholas J. (US 5688977), utilize n-butyllithium at cryogenic temperatures (-78°C) to achieve selectivity. While chemically feasible, this approach suffers from significant operational drawbacks, including massive energy consumption for cooling and poor reaction selectivity that limits yields to approximately 80%. The extreme alkalinity of n-butyllithium also poses safety risks and compatibility issues with other functional groups, making it a suboptimal choice for cost reduction in pharmaceutical intermediates manufacturing. These conventional bottlenecks necessitate a more robust and selective synthetic strategy.
The Novel Approach
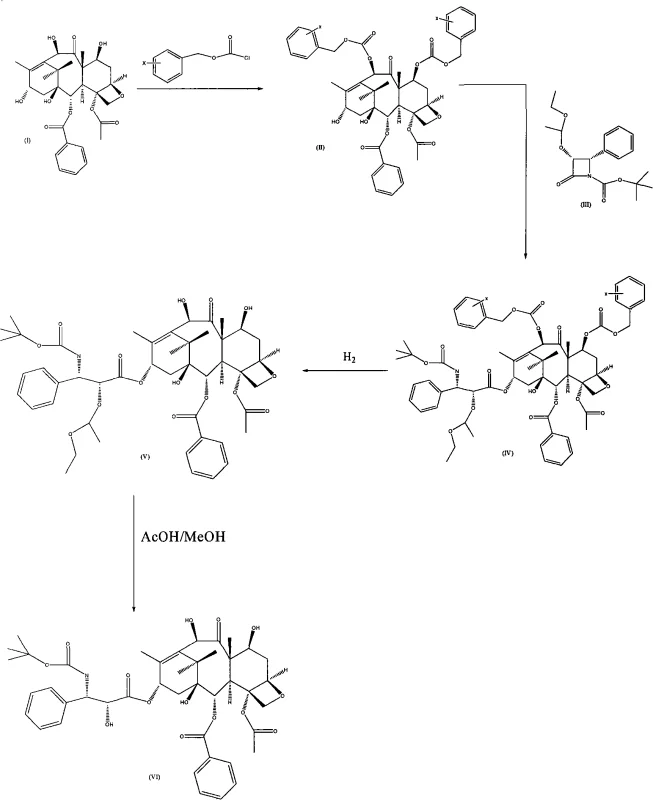
The methodology disclosed in CN101353334A offers a superior alternative by employing substituted benzyl chloroformates (X-CBZ-Cl) for the initial protection step. This reaction proceeds under mild conditions using 4-dimethylaminopyridine (DMAP) or N-lutidine as a catalyst in anhydrous tetrahydrofuran. Crucially, this esterification is highly chemoselective for the C-7 and C-10 hydroxyls, leaving the C-13 hydroxyl untouched, thereby preventing the formation of the troublesome C-13 protected byproducts seen in older methods. The resulting intermediate, C-7, C-10-di-X-CBZ-10-deacetyl baccatin III, is obtained with high purity (up to 97%) and excellent yield (85%), establishing a clean foundation for subsequent coupling reactions.
Furthermore, the novel approach integrates a side-chain precursor containing a 1-ethoxyethyl protecting group. This specific protecting group is designed to be removed under exceptionally mild acidic conditions, such as acetic acid in methanol, rather than the harsh acidic or reductive conditions required by other protecting groups. This gentleness preserves the stereochemical integrity of the molecule and minimizes the generation of degradation impurities. The combination of stable benzyl-type protection on the core and the labile ethoxyethyl protection on the side chain creates a orthogonal protection strategy that streamlines the entire synthesis, facilitating the production of high-purity Docetaxel suitable for clinical applications.
Mechanistic Insights into Selective Esterification and Deprotection
The core of this technological advancement lies in the precise control of nucleophilicity and steric hindrance during the esterification phases. In the first step, the use of DMAP acts as a potent nucleophilic catalyst, activating the benzyl chloroformate towards the secondary hydroxyls at C-7 and C-10. The reaction temperature is carefully controlled between -30°C and 50°C, with an optimal range of -5°C to 30°C, to balance reaction rate and selectivity. The stoichiometry is also critical; using 5 to 10 equivalents of DMAP and 5 to 80 equivalents of the acylating agent ensures complete conversion without promoting over-acylation at the C-13 position. This mechanistic precision eliminates the need for cryogenic cooling, representing a significant energy saving compared to organolithium routes.
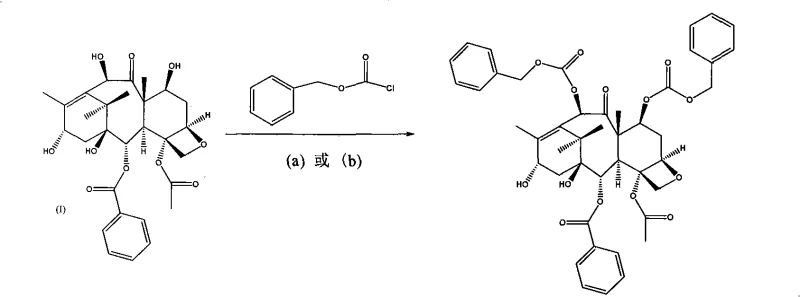
The deprotection sequence is equally sophisticated, leveraging orthogonal reactivity to strip protecting groups without affecting the core structure. The removal of the C-7 and C-10 benzyl carbamate groups is achieved via catalytic hydrogenolysis using palladium on carbon (Pd/C) at room temperature and normal pressure. This method is vastly superior to zinc-mediated reduction as it produces no heavy metal waste and operates under neutral conditions. Subsequently, the 1-ethoxyethyl group on the side chain is cleaved using a mixture of methanol and acetic acid. The acid-catalyzed hydrolysis proceeds rapidly at room temperature or with mild heating (45-55°C), releasing the free hydroxyl group at the C-2' position. This two-stage deprotection ensures that the final product, Docetaxel, is obtained with minimal epimerization or degradation, addressing the purity concerns that often plague generic taxane synthesis.
How to Synthesize Docetaxel Efficiently
The synthesis of Docetaxel via this novel route involves a sequence of highly optimized steps designed for reproducibility and scalability. The process begins with the selective protection of the baccatin core, followed by the coupling of the modified side chain, and concludes with a gentle deprotection sequence. Each stage has been refined to maximize yield while minimizing the formation of hard-to-remove impurities. For process chemists looking to implement this technology, strict adherence to the solvent ratios, catalyst loading, and temperature profiles described in the patent embodiments is essential to replicate the high purity results. The detailed standardized synthesis steps for implementing this pathway are provided in the guide below.
- Perform the first esterification of 10-deacetyl baccatin III with substituted benzyl chloroformate using DMAP as a catalyst in anhydrous THF to form the C-7, C-10 protected intermediate.
- Conduct the second esterification by reacting the protected intermediate with 1-boc-(3R,4S)-4-phenyl azetidine-2-ketone derivative using NaH and sodium bis(trimethylsilyl)amide.
- Execute selective deprotection via hydrogenolysis to remove C-7/C-10 protecting groups, followed by acidic hydrolysis to remove the 1-ethoxyethyl group, yielding pure Docetaxel.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible benefits beyond mere chemical elegance. The elimination of cryogenic conditions and violent deprotection steps translates directly into reduced operational expenditures (OPEX) and lower capital requirements for specialized equipment. By avoiding the use of n-butyllithium and zinc dust, facilities can operate with standard reactor configurations, significantly enhancing the commercial scale-up of complex pharmaceutical intermediates. Furthermore, the high selectivity of the reaction reduces the burden on downstream purification, leading to substantial cost savings in solvent usage and chromatography media.
- Cost Reduction in Manufacturing: The novel process eliminates the need for expensive and hazardous reagents like n-butyllithium and trichloroethyl chloroformate. By operating at ambient or mildly elevated temperatures, the energy consumption for heating and cooling is drastically reduced compared to conventional methods. Additionally, the high selectivity of the DMAP-catalyzed esterification minimizes the formation of byproducts, which reduces the loss of valuable starting materials and lowers the overall cost of goods sold (COGS) through improved material efficiency.
- Enhanced Supply Chain Reliability: The reliance on readily available reagents such as substituted benzyl chloroformates and common solvents like THF and methanol ensures a robust supply chain. Unlike methods dependent on specialized organometallics that may have long lead times or stability issues, this process utilizes stable chemicals that are easily sourced from multiple vendors. This diversification of raw material sources mitigates the risk of supply disruptions, ensuring consistent production schedules and reducing lead time for high-purity API intermediates.
- Scalability and Environmental Compliance: The replacement of zinc-mediated deprotection with catalytic hydrogenolysis significantly reduces the generation of heavy metal waste, simplifying wastewater treatment and environmental compliance. The mild acidic deprotection step further minimizes the corrosive load on equipment and the generation of hazardous effluents. These factors make the process inherently safer and more environmentally friendly, facilitating easier regulatory approval and smoother technology transfer to large-scale manufacturing sites.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Docetaxel synthesis method. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on purity profiles, reaction conditions, and scalability potential. Understanding these details is vital for technical teams evaluating the feasibility of adopting this route for commercial production.
Q: What are the primary advantages of this novel Docetaxel synthesis method over conventional trichloroethyl protection?
A: The novel method avoids the formation of difficult-to-remove byproducts associated with trichloroethyl chloroformate, such as compound IX. It utilizes milder deprotection conditions that prevent side reactions and significantly improve the purity of the final API intermediate.
Q: How does the new process impact the scalability of Docetaxel manufacturing?
A: By eliminating the need for extremely low temperatures (e.g., -78°C) required by n-butyllithium methods and avoiding violent deprotection steps, this process is far more amenable to large-scale industrial production, reducing energy consumption and operational complexity.
Q: What represents the critical quality attribute in the final deprotection step?
A: The critical quality attribute is the complete removal of the 1-ethoxyethyl protecting group without degrading the sensitive taxane core. The patented acidic conditions (AcOH/MeOH) ensure high conversion rates while maintaining the structural integrity of the molecule.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Docetaxel Supplier
The technological advancements detailed in patent CN101353334A represent a significant leap forward in the efficient production of Docetaxel, yet translating laboratory success to commercial reality requires a partner with deep process engineering expertise. NINGBO INNO PHARMCHEM stands at the forefront of this transition, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of taxane synthesis, including stringent purity specifications and rigorous QC labs that ensure every batch meets the highest international standards for oncology drugs.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to optimize their supply chains. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, ensuring that your transition to this advanced synthesis method is seamless, compliant, and economically advantageous.
