Advanced Synthesis of Triazolinethione Derivatives for Commercial Scale-Up of Complex Agrochemical Intermediates
Advanced Synthesis of Triazolinethione Derivatives for Commercial Scale-Up of Complex Agrochemical Intermediates
The global demand for high-efficacy fungicides continues to drive innovation in the synthesis of heterocyclic intermediates, particularly those containing triazole and thiocarbonyl motifs. Patent CN1187343C introduces a transformative methodology for the production of 2-(1-chloro-cycloprop-1-yl)-1-(2-chloro-phenyl)-3-(4,5-dihydro-1,2,4-triazol-5-thiocarbon-1-yl)-propan-2-ol, a potent active compound with significant microbicidal properties. This technical disclosure addresses critical bottlenecks in existing manufacturing protocols by replacing unstable intermediates with robust hydrochloride salts and substituting harsh oxidation conditions with a controlled iron(III) chloride catalytic system. For R&D directors and procurement strategists, this patent represents a pivotal shift towards more reliable agrochemical intermediate supplier capabilities, offering a pathway to higher purity profiles and reduced operational risks in multi-kilogram production campaigns.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those described in WO 99-18 087, rely on a synthetic sequence that introduces significant instability early in the process. The conventional route typically involves reacting a chlorohydrin precursor with hydrazine hydrate to generate a free hydrazine intermediate. As illustrated in the structural comparison of legacy intermediates, this free base form is chemically fragile and susceptible to degradation, leading to the formation of complex impurity profiles that are difficult to purge in downstream processing. Furthermore, the final oxidation step in traditional methods often employs elemental sulfur and oxygen in the presence of strong bases like potassium hydroxide. This aggressive environment frequently results in interfering overoxidation reactions, causing the dissociation of the critical sulfur atom from the triazole ring and drastically lowering the final assay of the active pharmaceutical ingredient.
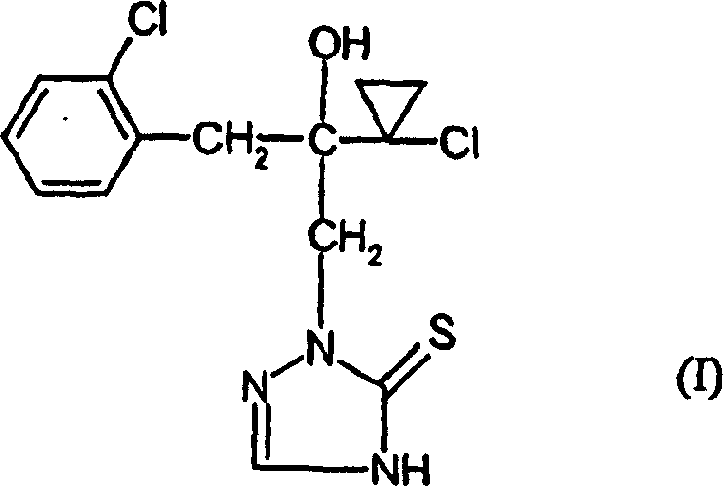
The Novel Approach
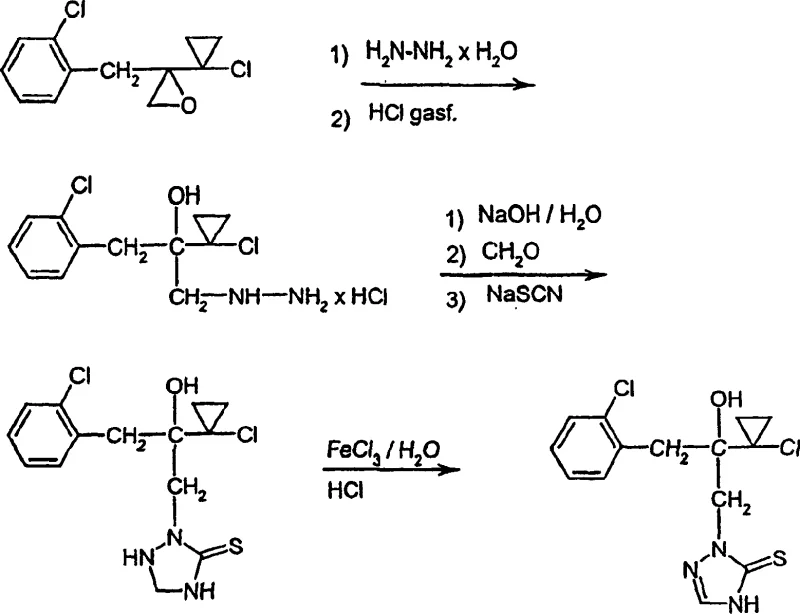
In stark contrast, the methodology disclosed in CN1187343C engineers stability directly into the molecular backbone of the intermediate. By reacting the oxirane precursor (Formula II) with hydrazine hydrate and immediately converting the product into a hydrazine hydrochloride salt (Formula III), the process eliminates the handling risks associated with free hydrazines. This salt form is crystalline, stable, and easily purified, serving as a robust pivot point for the subsequent cyclization. The complete reaction landscape demonstrates how this stable salt is seamlessly converted into the triazolidine ring system and finally oxidized to the target thione. This strategic modification not only simplifies the isolation procedures between steps but also creates a buffer against variability, ensuring that the supply chain remains uninterrupted even during scale-up phases where thermal gradients might otherwise degrade sensitive species.
Mechanistic Insights into FeCl3-Catalyzed Cyclization and Oxidation
The cornerstone of this improved synthetic route lies in the mechanistic elegance of the final oxidative cyclization step. Unlike the radical-based oxidation mechanisms of prior art which rely on atmospheric oxygen and sulfur powder, the present invention utilizes iron(III) chloride in an aqueous hydrochloric acid medium. This Lewis acid-mediated pathway facilitates a controlled dehydrogenation of the triazolidine ring (Formula V) to form the aromatic triazolinethione system (Formula I). The presence of ferric ions likely coordinates with the sulfur and nitrogen atoms, lowering the activation energy required for the formation of the double bond within the heterocycle while simultaneously protecting the sulfur moiety from excessive oxidation to sulfones or sulfoxides. This precise control over the redox potential is critical for maintaining the biological activity of the final fungicide, as the thione tautomer is essential for binding to the target enzymatic sites in fungal pathogens.
From an impurity control perspective, the use of FeCl3 offers a distinct advantage by minimizing side reactions that plague basic oxidation conditions. In alkaline environments, the cyclopropyl ring adjacent to the reaction center is prone to ring-opening or rearrangement due to the strain energy inherent in the three-membered ring. However, the acidic conditions employed in this novel protocol preserve the integrity of the 1-chloro-cyclopropyl group, ensuring that the final product retains its specific stereochemical and structural features. Additionally, the aqueous workup following the iron-catalyzed step allows for the efficient removal of inorganic iron salts through simple phase separation, thereby reducing the burden on chromatographic purification and enabling the production of high-purity agrochemical intermediates that meet stringent regulatory specifications for residue limits.
How to Synthesize Triazolinethione Derivative Efficiently
The implementation of this synthesis requires careful attention to solvent selection and stoichiometry to maximize the benefits of the stabilized intermediate pathway. The process begins with the ring-opening of the epoxide in aromatic hydrocarbons, followed by a telescoped neutralization and cyclization sequence that leverages the solubility differences between the salt and the free base. Detailed standardized operating procedures for temperature control, reagent addition rates, and phase separation techniques are critical for reproducing the high yields reported in the patent examples. For process chemists looking to adopt this technology, understanding the nuances of the hydrazine salt isolation is key to unlocking the full potential of this route.
- React 2-(1-chloro-cycloprop-1-yl)-2-(2'-chloro-benzyl)-oxirane (II) with hydrazine hydrate in aromatic hydrocarbons, followed by HCl treatment to form stable hydrazine hydrochloride (III).
- Treat hydrazine hydrochloride (III) with alkali metal hydroxides, formaldehyde, and thiocyanate to cyclize into the triazolidine derivative (V).
- Oxidize the triazolidine derivative (V) using iron(III) chloride in aqueous hydrochloric acid to obtain the final triazolinethione product (I).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers compelling economic and logistical benefits that extend beyond simple yield improvements. The stabilization of the hydrazine intermediate fundamentally changes the inventory management profile of the production campaign. Because the hydrazine hydrochloride salt is stable and non-hazardous compared to free hydrazines, it can be stockpiled or transported with greater ease, reducing the need for just-in-time delivery of sensitive reagents and mitigating the risk of production stoppages due to raw material degradation. This enhanced stability translates directly into a more resilient supply chain, allowing manufacturers to respond more flexibly to market fluctuations in demand for fungicide active ingredients without compromising on quality or safety standards.
- Cost Reduction in Manufacturing: The elimination of unstable intermediates and the avoidance of harsh oxidation conditions lead to substantial cost savings in raw material consumption and waste disposal. By achieving yields exceeding 95% in the initial steps and nearly quantitative conversion in the final oxidation, the process minimizes the volume of mother liquors and by-products that require treatment. Furthermore, the replacement of specialized oxidation reagents with commodity chemicals like iron(III) chloride reduces the direct cost of goods sold, while the simplified workup procedures decrease the consumption of solvents and energy required for purification, driving down the overall manufacturing cost per kilogram of the high-purity agrochemical intermediate.
- Enhanced Supply Chain Reliability: The robustness of the hydrazine hydrochloride intermediate ensures consistent quality output regardless of minor variations in reaction conditions, which is vital for maintaining long-term supply contracts. The use of common, commercially available solvents such as toluene, ethyl acetate, and ethanol further secures the supply chain against disruptions caused by the scarcity of exotic reagents. This reliability allows procurement teams to negotiate better terms with vendors and ensures that production schedules for reducing lead time for high-purity agrochemical intermediates can be met with high confidence, even in volatile market environments.
- Scalability and Environmental Compliance: The process is inherently scalable due to its operation at moderate temperatures and atmospheric pressure, removing the need for expensive high-pressure reactors or cryogenic cooling systems. From an environmental standpoint, the avoidance of sulfur powder and strong bases in the final step significantly reduces the generation of hazardous sulfide wastes and saline effluents. This aligns with modern green chemistry principles and facilitates easier compliance with increasingly strict environmental regulations, positioning the manufacturer as a responsible partner in the sustainable production of crop protection agents.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on the practical advantages of the FeCl3-mediated pathway over traditional methods.
Q: Why is the hydrazine hydrochloride salt preferred over the free base in this synthesis?
A: The free hydrazine compound formed in conventional methods is relatively unstable and prone to forming undesired by-products. Converting it immediately to the hydrochloride salt (Formula III) ensures stability during storage and handling, significantly improving overall process safety and yield consistency.
Q: What is the advantage of using FeCl3 for the final oxidation step?
A: Traditional methods utilize oxygen and sulfur in the presence of potassium hydroxide, which carries a high risk of interfering overoxidation leading to sulfur dissociation. The FeCl3-catalyzed pathway operates under milder conditions, preventing sulfur loss and ensuring the structural integrity of the thione group.
Q: Can this process be scaled for industrial production?
A: Yes, the process utilizes common solvents like toluene and ethyl acetate and avoids extreme pressures. The high yields reported in the patent examples (exceeding 95% in key steps) indicate excellent atom economy and reduced waste generation, making it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triazolinethione Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced crop protection agents depends on the seamless translation of laboratory patents into industrial reality. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate details of the hydrazine salt stabilization and iron-catalyzed oxidation are perfectly managed at scale. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify that every batch of triazolinethione derivative meets the exacting standards required for global agrochemical registration and formulation.
We invite you to collaborate with us to leverage this innovative synthesis for your next product launch. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your supply chain efficiency and reduce your total cost of ownership for high-value fungicide intermediates.
