Optimizing Cefdinir Production: A Technical Breakthrough for Commercial Scale-up of Complex Antibiotics
Optimizing Cefdinir Production: A Technical Breakthrough for Commercial Scale-up of Complex Antibiotics
The pharmaceutical industry constantly seeks more efficient pathways for producing critical antibiotics, and the recent disclosure in patent CN115260213A offers a compelling solution for the synthesis of Cefdinir. This third-generation cephalosporin is vital for treating a broad spectrum of bacterial infections, yet its manufacturing has historically been plagued by complex multi-step sequences and environmental concerns. The patented methodology introduces a novel active intermediate derived from the reaction of noraminothiazolyloximate and N-ethynyl-N,4-dimethylbenzenesulfonamide, fundamentally altering the condensation landscape with 7-AVNA. By shifting away from traditional thioester activation, this process achieves a one-pot preparation under remarkably mild conditions, significantly enhancing product quality while reducing energy consumption. For R&D directors and supply chain leaders, this represents a pivotal opportunity to streamline the production of high-purity pharmaceutical intermediates.
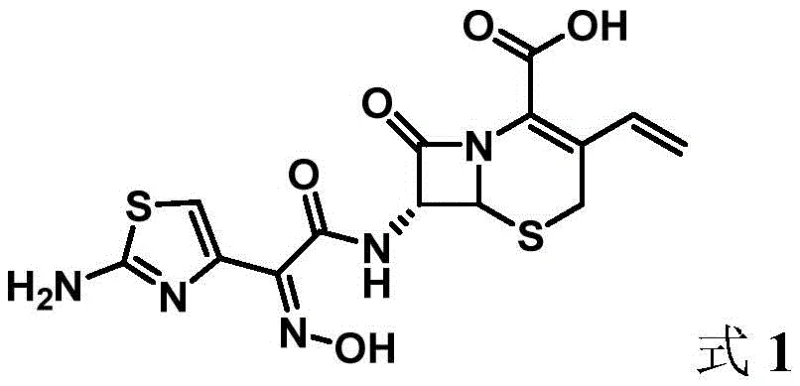
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Cefdinir has relied heavily on the introduction of the side chain via activated thioesters, such as MICA-activated thioesters or Dinis-activated thioesters. These conventional routes necessitate the use of organophosphorus reagents to activate the corresponding carboxylic acids, introducing significant complexity and cost into the manufacturing process. The reliance on phosphorus chemistry often leads to the generation of sulfide by-products that are notoriously difficult to separate from the final API, creating bottlenecks in downstream processing and purification. Furthermore, the traditional workflow typically involves at least two distinct steps for side chain introduction, each requiring isolation and purification operations that cumulatively erode the overall reaction yield. This multi-step nature not only increases the consumption of raw materials and solvents but also amplifies the environmental footprint due to the generation of hazardous waste streams associated with phosphorus and sulfur residues.
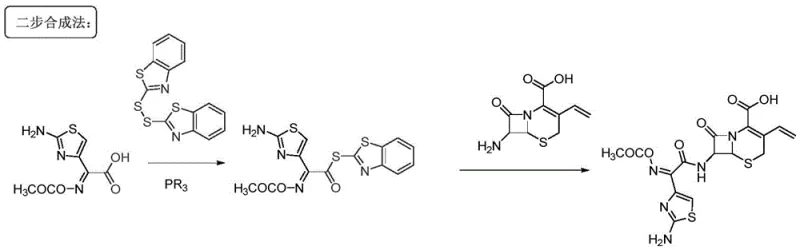
The Novel Approach
In stark contrast to the legacy thioester pathways, the innovative strategy outlined in CN115260213A utilizes a unique alkyne-based activation mechanism to facilitate the coupling reaction. By reacting noraminothiazolyloximate with N-ethynyl-N,4-dimethylbenzenesulfonamide, the process generates a highly reactive intermediate in situ that readily undergoes nucleophilic substitution with 7-AVNA. This approach effectively bypasses the need for organophosphorus activators, thereby eliminating the associated toxicological and separation challenges. The reaction proceeds efficiently in common organic solvents like dichloromethane at ambient temperatures, demonstrating exceptional compatibility with large-scale manufacturing equipment. The ability to perform this transformation in a one-pot fashion without isolating the intermediate drastically reduces the operational timeline and minimizes the potential for product degradation during handling. This streamlined protocol not only improves the atom economy of the synthesis but also aligns perfectly with modern green chemistry principles by reducing waste and energy usage.
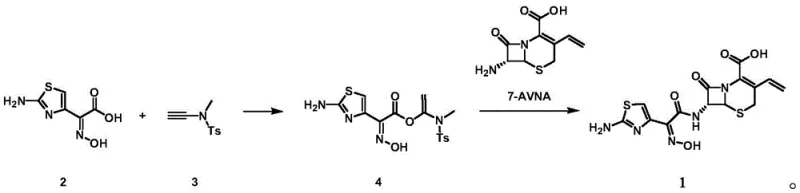
Mechanistic Insights into Alkyne-Mediated Activation and Coupling
The core of this technological advancement lies in the specific reactivity of the N-ethynyl-N,4-dimethylbenzenesulfonamide reagent, which acts as a superior electrophilic activator compared to traditional carbodiimides or phosphonium salts. Upon interaction with the oxime acid moiety of the thiazole side chain, the alkyne group facilitates an addition reaction that creates a transient, highly activated ester-like species. This intermediate possesses enhanced electrophilicity at the carbonyl carbon, making it exceptionally susceptible to nucleophilic attack by the amino group of the 7-AVNA beta-lactam nucleus. The mechanistic pathway avoids the formation of stable phosphorous-oxygen bonds that characterize older methods, instead relying on the cleavage of the sulfonamide-alkyne bond to drive the reaction forward. This subtle shift in chemical logic ensures that the driving force for amide bond formation is maintained without generating persistent, hard-to-remove by-products that could contaminate the final antibiotic substance.
From an impurity control perspective, this mechanism offers profound advantages for ensuring the safety and efficacy of the final drug product. The absence of organophosphorus reagents means there is no risk of phosphine oxide residues, which are often genotoxic impurities of concern in regulatory filings. Additionally, the mild reaction conditions, typically ranging from 0°C to 50°C with an optimum around 25°C, prevent the thermal degradation of the sensitive beta-lactam ring, a common issue in harsher activation protocols. The high conversion rates observed in the initial addition step ensure that very little unreacted starting material carries over into the second stage, simplifying the purification profile. Consequently, the final crude product exhibits a much cleaner HPLC profile, requiring less aggressive chromatographic conditions to achieve the stringent purity specifications demanded by global pharmacopoeias for oral antibiotics.
How to Synthesize Cefdinir Efficiently
The practical implementation of this synthesis route is designed for seamless integration into existing pilot and commercial facilities, requiring only standard reactor configurations and common solvent systems. The protocol emphasizes simplicity, utilizing a molar ratio of approximately 1.0:1.0:1.2 for the oxime acid, alkyne activator, and 7-AVNA respectively, to maximize efficiency while minimizing excess reagent costs. Operators can monitor the reaction progress using standard TLC or HPLC techniques, allowing for precise endpoint determination before proceeding to the workup phase. The detailed standardized synthesis steps below outline the precise conditions and parameters required to replicate the high yields reported in the patent examples, ensuring consistent batch-to-batch quality.
- Perform an addition reaction between noraminothiazolyloximate and N-ethynyl-N,4-dimethylbenzenesulfonamide in dichloromethane at 25°C to form the active intermediate.
- Without isolating the intermediate, directly add 7-AVNA to the reaction mixture for nucleophilic substitution.
- Concentrate the reaction mixture and purify via silica gel column chromatography to obtain high-purity Cefdinir.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route translates into tangible strategic benefits that extend far beyond simple yield improvements. By eliminating the dependency on specialized and often volatile organophosphorus reagents, manufacturers can significantly reduce their exposure to supply chain disruptions caused by the scarcity of these specific chemicals. The simplification of the process from a multi-step sequence with isolations to a streamlined one-pot operation inherently lowers the labor intensity and equipment occupancy time, leading to substantial cost savings in manufacturing overheads. Furthermore, the reduction in hazardous waste generation simplifies compliance with increasingly strict environmental regulations, mitigating the risk of production halts due to waste disposal limitations. This robust and scalable methodology ensures a more reliable supply of high-purity pharmaceutical intermediates, safeguarding the continuity of antibiotic production lines against market fluctuations.
- Cost Reduction in Manufacturing: The elimination of expensive organophosphorus activators and the removal of intermediate isolation steps directly lower the bill of materials and processing costs. By avoiding the need for complex purification to remove phosphorus and sulfur by-products, the facility saves significantly on solvent consumption and chromatography media, driving down the overall cost of goods sold. The higher reaction yields mean that less raw material is wasted per kilogram of finished product, optimizing the utilization of valuable starting materials like 7-AVNA. These cumulative efficiencies create a leaner manufacturing model that enhances competitiveness in the global generic antibiotic market without compromising on quality standards.
- Enhanced Supply Chain Reliability: Utilizing commercially available and stable reagents like N-ethynyl-N,4-dimethylbenzenesulfonamide reduces the risk of supply bottlenecks associated with specialty activators. The mild reaction conditions allow for the use of standard glass-lined or stainless steel reactors without the need for exotic materials resistant to highly corrosive phosphorus species. This flexibility enables faster turnaround times between batches and allows for more agile response to sudden spikes in demand for respiratory infection treatments. The simplified workflow also reduces the likelihood of operator error during complex transfer and isolation steps, ensuring a more consistent and predictable production schedule for long-term supply contracts.
- Scalability and Environmental Compliance: The one-pot nature of this synthesis drastically reduces the volume of wastewater and solid waste generated per unit of production, aligning with sustainable manufacturing goals. The absence of toxic phosphorus residues simplifies the effluent treatment process, lowering the operational costs associated with environmental compliance and waste disposal. This greener profile makes the facility more attractive to international partners who prioritize ESG (Environmental, Social, and Governance) criteria in their supplier selection processes. The proven scalability of the reaction in solvents like dichloromethane and toluene ensures that the process can be safely expanded from kilogram-scale development to multi-ton commercial production with minimal re-optimization.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced Cefdinir synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on process robustness and quality outcomes. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the value proposition of this new manufacturing route.
Q: What are the primary advantages of this new Cefdinir synthesis route over traditional thioester methods?
A: The new route eliminates the need for expensive organophosphorus reagents and avoids the formation of difficult-to-separate sulfide by-products, resulting in a cleaner reaction profile and higher overall yield.
Q: Which solvent system provides the optimal yield for this one-pot condensation?
A: Experimental data indicates that dichloromethane (DCM) provides the highest yield (91.1%) and purity (99.2%), outperforming solvents like THF, toluene, and ethyl acetate.
Q: Is purification of the intermediate required between reaction steps?
A: No, the process is designed as a true one-pot method where the intermediate is not purified; 7-AVNA is added directly to the reaction mixture after the first step is complete.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefdinir Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this patented synthesis route in delivering high-quality Cefdinir to the global market. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this one-pot method are fully realized in practice. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of API intermediate adheres to the highest international standards. We are committed to leveraging this innovative chemistry to provide our clients with a secure, cost-effective, and environmentally responsible supply of critical antibiotic ingredients.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can optimize your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic impact of switching to this phosphorus-free route. We encourage potential partners to contact us for specific COA data and route feasibility assessments tailored to your production volumes, ensuring a seamless transition to this superior manufacturing technology.
