Advanced Amide Bond Formation Technology for Commercial Scale-Up of Complex Peptide Sequences
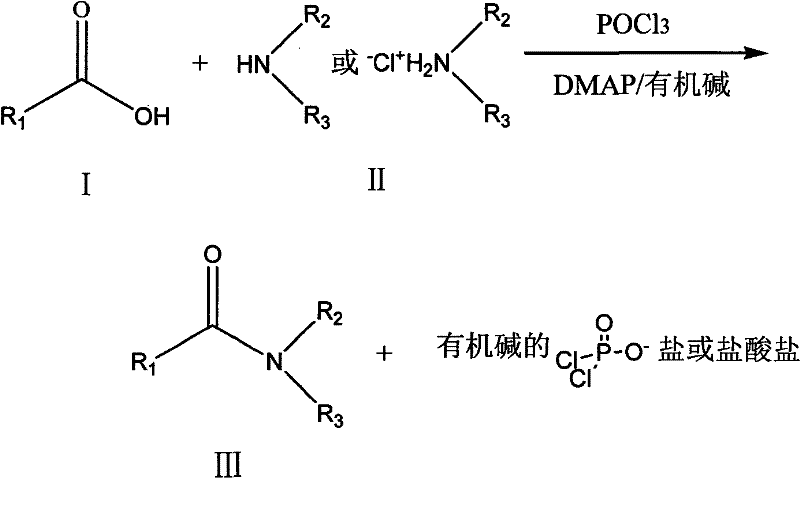
The pharmaceutical and fine chemical industries continuously seek robust methodologies for constructing amide linkages, a fundamental structural motif prevalent in active pharmaceutical ingredients and advanced intermediates. Patent CN101235078B introduces a transformative production method that utilizes phosphoryl halide in the presence of N,N-dimethylpyridine (DMAP) and organic alkali to facilitate the reaction between carboxylic acids and free amines or amine salts. This technical breakthrough addresses longstanding challenges in peptide synthesis and amide formation by offering a pathway characterized by low operational costs, simplified processing steps, and exceptionally high yields without compromising stereochemical integrity. For R&D directors and procurement specialists, understanding the nuances of this technology is critical for optimizing supply chains and reducing the overall cost of goods sold in complex molecule manufacturing. The method's ability to support industrial production while maintaining stringent purity standards positions it as a vital asset for reliable pharmaceutical intermediates suppliers aiming to enhance their competitive edge in the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the formation of amido linkages has relied heavily on a variety of coupling reagents that often present significant logistical and financial burdens for large-scale manufacturing operations. Many existing couplers are prohibitively expensive, driving up the raw material costs for high-purity OLED material or API intermediate production to unsustainable levels. Furthermore, conventional methods frequently encounter solubility issues, requiring complex solvent systems or elevated temperatures that can degrade sensitive functional groups within the molecular structure. A particularly critical drawback in peptide chemistry is the propensity for racemization, where the chiral center adjacent to the carboxyl group loses its optical purity, rendering the final product unsuitable for therapeutic applications. These operational complexities often result in extended reaction times and difficult purification processes, creating bottlenecks that hinder the commercial scale-up of complex polymer additives or specialty chemicals. Consequently, manufacturers face increased waste generation and higher energy consumption, which contradicts modern environmental compliance standards and sustainability goals.
The Novel Approach
In stark contrast to traditional methodologies, the novel approach detailed in the patent leverages phosphoryl halide as a highly efficient and cost-effective coupling agent to drive amide bond formation with remarkable precision. By employing phosphorus oxychloride (POCl3) in conjunction with a catalytic amount of DMAP and an organic base, this method circumvents the need for expensive proprietary coupling reagents while achieving superior reaction kinetics. The process operates effectively across a broad temperature range, typically controlled between -40°C and 50°C, allowing for flexibility in managing exothermic reactions and ensuring safety during scale-up. One of the most significant advantages is the drastic reduction in racemization, with chiral high-performance liquid chromatography detecting rates below 2%, thereby preserving the enantiomeric excess crucial for bioactive molecules. This streamlined protocol simplifies the operational workflow, reducing the number of unit operations required and facilitating easier isolation of the target amide product. Ultimately, this innovation provides a scalable solution that aligns perfectly with the strategic objectives of cost reduction in electronic chemical manufacturing and other high-value sectors.
Mechanistic Insights into POCl3-Catalyzed Amidation
The core mechanism of this reaction involves the activation of the carboxylic acid by the phosphoryl halide to form a highly reactive mixed anhydride or acyl phosphate intermediate in situ. In the presence of the nucleophilic catalyst DMAP, the electrophilicity of the carbonyl carbon is significantly enhanced, facilitating a rapid nucleophilic attack by the amine or amine salt. This catalytic cycle ensures that the reaction proceeds efficiently even with sterically hindered substrates, which are often problematic in standard amidation protocols. The organic base serves a dual purpose by neutralizing the hydrogen halide byproduct generated during the activation step and maintaining the reaction medium's pH to prevent protonation of the amine nucleophile. Detailed analysis of the reaction pathway reveals that the transition state is stabilized by the coordination of the phosphorus species, lowering the activation energy barrier and allowing the reaction to complete within approximately 1 to 2 hours. This mechanistic efficiency is paramount for maintaining high throughput in continuous flow reactors or large batch vessels used in industrial settings.
Controlling impurity profiles is another critical aspect where this mechanism excels, particularly regarding the suppression of side reactions that commonly plague peptide synthesis. The mild reaction conditions and the specific selectivity of the phosphoryl halide activation minimize the formation of N-acylurea byproducts or epimerization at the alpha-carbon of amino acid residues. By carefully selecting the organic solvent, such as dichloromethane or ethyl acetate, the solubility of both reactants and the resulting ammonium salts is optimized, preventing precipitation that could lead to incomplete conversion. The workup procedure involves standard aqueous washing steps to remove the organic base salts and phosphorus-containing byproducts, which are water-soluble and easily separated from the organic phase containing the desired amide. This clean separation profile ensures that the final product meets rigorous quality specifications without the need for extensive chromatographic purification, thereby enhancing the overall process mass intensity. For supply chain heads, this translates to reduced lead time for high-purity pharmaceutical intermediates and a more predictable manufacturing schedule.
How to Synthesize Amide Bonds Efficiently
To implement this synthesis route effectively, operators must adhere to precise stoichiometric ratios and addition sequences to maximize yield and safety. The process begins by dissolving the carboxylic acid and the amine component in a dry organic solvent, followed by the sequential addition of the organic base and the DMAP catalyst under inert atmosphere conditions. It is imperative to control the addition rate of the phosphoryl halide to manage the exotherm and maintain the reaction temperature within the optimal window specified in the technical documentation. Detailed standardized synthesis steps see the guide below for exact parameters regarding molar equivalents and stirring times.
- Dissolve carboxylic acid and free amine or amine salt in an organic solvent such as dichloromethane or ethyl acetate.
- Add an organic base like triethylamine and a catalytic amount of DMAP to the reaction mixture under controlled temperature.
- Slowly add phosphoryl halide (preferably POCl3) to generate the amide bond, followed by standard aqueous workup and purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, adopting this phosphoryl halide-mediated amidation method offers substantial strategic benefits for procurement managers and supply chain leaders focused on margin improvement and operational resilience. The primary driver of value is the replacement of costly, specialized coupling reagents with commodity chemicals like phosphorus oxychloride, which are readily available in the global bulk chemical market at a fraction of the price. This shift in raw material sourcing significantly reduces the direct material costs associated with manufacturing complex intermediates, allowing companies to improve their gross margins without sacrificing product quality. Furthermore, the simplicity of the reaction setup and workup reduces the demand for specialized equipment and minimizes the consumption of utilities such as heating and cooling energy. These operational efficiencies contribute to a leaner manufacturing process that is less susceptible to supply chain disruptions caused by the scarcity of niche reagents. Consequently, organizations can achieve significant cost savings and enhance their ability to respond quickly to fluctuating market demands for key pharmaceutical building blocks.
- Cost Reduction in Manufacturing: The utilization of industrial-grade phosphoryl halides eliminates the dependency on expensive proprietary coupling agents, leading to a drastic simplification of the bill of materials. By removing the need for transition metal catalysts or complex activating esters, the process avoids the costly downstream removal steps often required to meet residual metal specifications in drug substances. This qualitative improvement in process economics allows for a more competitive pricing structure when bidding for long-term supply contracts with major pharmaceutical clients. Additionally, the high yields reported in the patent examples, often exceeding 90%, mean that less raw material is wasted, further driving down the effective cost per kilogram of the final product. The cumulative effect of these factors results in a robust economic model that supports sustainable growth and investment in R&D.
- Enhanced Supply Chain Reliability: Sourcing reliability is greatly improved as the key reagents, such as POCl3 and triethylamine, are produced by multiple manufacturers worldwide, mitigating the risk of single-source supply failures. The stability of these chemicals allows for long-term storage and inventory management without the degradation issues associated with sensitive coupling reagents that require cold chain logistics. This availability ensures that production schedules can be maintained consistently, reducing the lead time for high-purity pharmaceutical intermediates and preventing stockouts that could disrupt downstream drug formulation. Moreover, the robustness of the reaction conditions means that the process can be transferred between different manufacturing sites with minimal re-validation effort, providing flexibility in capacity planning. Such supply chain agility is essential for maintaining continuity of supply in the face of global logistical challenges.
- Scalability and Environmental Compliance: The method is inherently designed for scale-up, with reaction parameters that are easily controlled in large-scale reactors, ensuring consistent product quality from pilot plant to commercial production. The byproducts generated are primarily inorganic salts and phosphorus species that can be treated using standard wastewater treatment protocols, simplifying environmental compliance and reducing the burden on waste management systems. By avoiding the use of heavy metals or toxic organotin reagents, the process aligns with green chemistry principles and regulatory requirements for cleaner production technologies. This environmental advantage not only reduces disposal costs but also enhances the corporate sustainability profile, which is increasingly important for stakeholders and investors. The combination of scalability and environmental stewardship makes this technology a preferred choice for modern chemical manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this amide bond formation technology in industrial settings. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation to ensure accuracy and relevance. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this method into their existing production workflows. The insights provided here aim to clarify the operational benefits and potential applications of this innovative synthetic route.
Q: Does this method cause racemization in chiral amino acids?
A: According to patent data, this method exhibits extremely low racemization rates (less than 2%) even for chiral carboxylic acids linked to the carboxyl group, making it highly suitable for peptide synthesis.
Q: What are the preferred reaction conditions for this coupling?
A: The reaction is preferably conducted in dichloromethane at temperatures ranging from -40°C to 50°C, using POCl3 as the coupling agent and triethylamine as the base.
Q: Is this process scalable for industrial production?
A: Yes, the patent explicitly states the method uses cheap, industrially available reagents and involves simple operations, supporting large-scale industrial production needs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Amide Bond Intermediates Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global fine chemical market. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory concept to full-scale manufacturing. Our commitment to quality is unwavering, with stringent purity specifications enforced through our rigorous QC labs to guarantee that every batch meets the highest industry standards. We understand that the successful commercialization of complex molecules requires not just chemical expertise but also a deep understanding of supply chain dynamics and cost optimization strategies. By leveraging technologies like the phosphoryl halide-mediated amidation method, we help our partners achieve their performance targets while minimizing environmental impact and operational costs.
We invite you to engage with our technical procurement team to discuss how our capabilities can support your specific project requirements and timeline. Request a Customized Cost-Saving Analysis today to explore the potential economic benefits of switching to this efficient synthesis route for your key intermediates. Our experts are ready to provide specific COA data and route feasibility assessments tailored to your unique molecular structures and volume needs. Let us collaborate to build a resilient and cost-effective supply chain that drives your business forward in an increasingly demanding marketplace.
