Advanced Synthesis of Tucatinib: Overcoming Scale-Up Barriers for HER2 Inhibitors
Advanced Synthesis of Tucatinib: Overcoming Scale-Up Barriers for HER2 Inhibitors
The pharmaceutical landscape for HER2-positive breast cancer treatment has been significantly advanced by the development of Tucatinib, a highly specific tyrosine kinase inhibitor. As detailed in patent CN115650977A, a novel synthesis method has emerged that addresses critical bottlenecks in the manufacturing of this vital oncology API. This technical insight report analyzes the strategic shift from traditional coupling methods to a more robust, in-situ ring construction approach. By leveraging stable intermediates and avoiding hazardous triflate reagents, this process offers a compelling value proposition for supply chain stability and cost efficiency. For procurement leaders and R&D directors alike, understanding this pathway is essential for securing a reliable tucatinib supplier capable of meeting global demand.
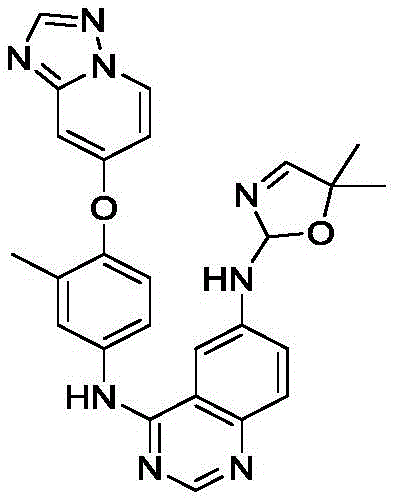
Tucatinib functions as a potent inhibitor with high specificity, often used in combination with trastuzumab and capecitabine. The molecular complexity, featuring a quinazoline core linked to a substituted phenyl and a dimethyl-oxazole ring, presents synthetic challenges. The innovation disclosed in the patent focuses on optimizing the construction of the oxazole heterocycle, a step that has historically plagued manufacturers with low yields and purification difficulties. This report delves into the mechanistic advantages and commercial implications of adopting this refined synthetic strategy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Tucatinib has relied on a convergent strategy that couples a pre-formed oxazole fragment with the quinazoline core. As illustrated in the prior art, this typically involves a seven-step sequence to generate a key amine intermediate, followed by a final coupling reaction with oxazole trifluoromethanesulfonate. This conventional route is fraught with inefficiencies; the total yield is reported to be merely 19 percent, which is economically unsustainable for large-scale production. Furthermore, the use of trifluoromethanesulfonate reagents introduces significant safety hazards and cost burdens due to the expense of the raw materials and the difficulty in handling fluorinated byproducts.
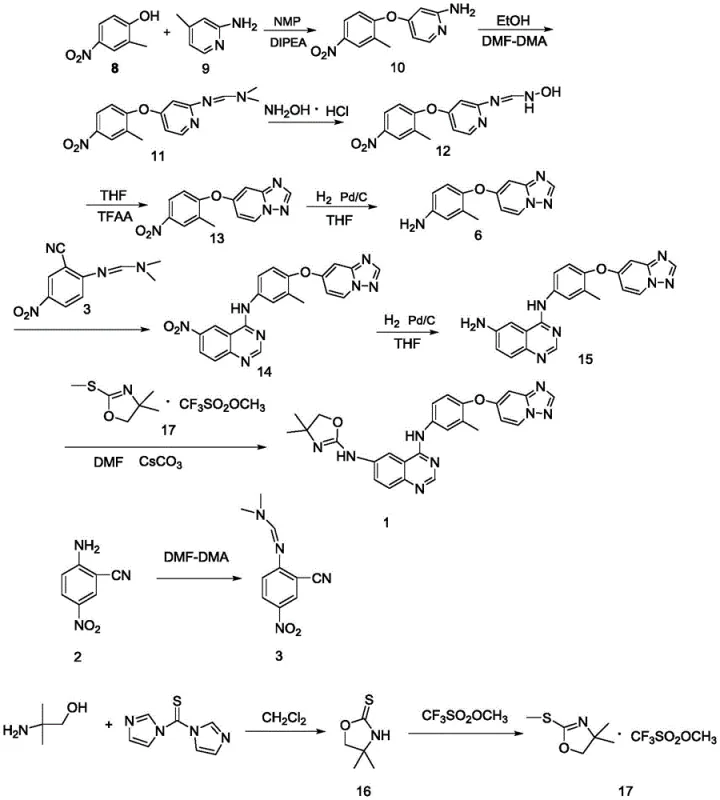
Beyond the economic drawbacks, the final coupling step in the traditional method suffers from poor reproducibility and difficult scale-up characteristics. The reaction conditions are often sensitive, leading to batch-to-batch variability that complicates regulatory validation. For a supply chain head, this translates to unpredictable lead times and potential shortages of high-purity material. The reliance on such a fragile final step creates a single point of failure in the manufacturing process, making it an unattractive option for companies seeking cost reduction in kinase inhibitor manufacturing. The accumulation of impurities through the long linear sequence further exacerbates purification challenges, requiring extensive chromatography that drives up operational costs.
The Novel Approach
In stark contrast, the novel method disclosed in CN115650977A adopts a divergent strategy that constructs the oxazole ring directly on the quinazoline scaffold. Instead of coupling two complex fragments, the process starts with the stable amine intermediate (Compound 15) and builds the heterocycle through a series of high-yielding transformations. This approach eliminates the need for the expensive and hazardous triflate coupling reagent entirely. By breaking the synthesis down into manageable steps involving dithiocarbamate and isothiocyanate intermediates, the process achieves superior control over reaction kinetics and impurity profiles. The result is a streamlined workflow that is inherently safer and more amenable to industrial scale-up.
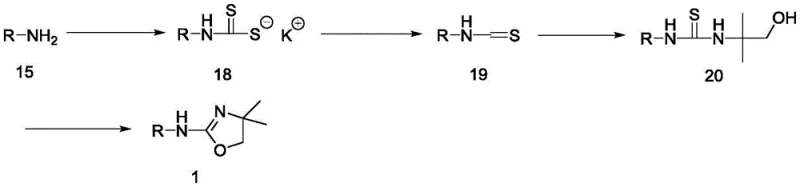
The strategic advantage of this new route lies in its modularity and the stability of its intermediates. The transformation of the amine to an isothiocyanate, followed by reaction with 2-amino-2-methylpropanol (AMP), creates a robust thiourea linkage that serves as a perfect precursor for cyclization. This methodology not only improves the overall reaction yield but also facilitates product purification, as the intermediates can be easily isolated or processed without complex chromatographic separations. For a reliable agrochemical intermediate supplier or pharma partner, this represents a paradigm shift towards more sustainable and economically viable manufacturing processes that reduce waste and energy consumption.
Mechanistic Insights into Thiourea-Mediated Cyclization
The core of this innovation is the clever utilization of sulfur chemistry to construct the oxygen-containing oxazole ring. The mechanism begins with the thionation of the primary amine (R-NH2) using a reagent such as carbon disulfide in the presence of a base like potassium carbonate. This generates a dithiocarbamate salt, which is subsequently cyanated using cyanuric chloride (TCT) to yield the reactive isothiocyanate species. This step is critical as it activates the nitrogen center for nucleophilic attack. The subsequent reaction with AMP proceeds smoothly at mild temperatures (5-25°C) to form the thiourea intermediate (Compound 20). This exothermic process is highly selective, minimizing the formation of side products that often plague direct alkylation methods.
The final cyclization step is a masterpiece of atom economy and functional group tolerance. The thiourea intermediate undergoes intramolecular cyclization upon treatment with p-toluenesulfonyl chloride and a base. The tosyl group acts as an activating agent, facilitating the dehydration and rearrangement required to close the oxazole ring. This transformation effectively replaces the sulfur atom with oxygen in the heterocyclic framework, a non-trivial chemical feat achieved under mild conditions (0-40°C). The use of common bases like sodium hydroxide or triethylamine ensures that the process remains cost-effective. This mechanistic pathway avoids the harsh conditions typically associated with oxazole synthesis, thereby preserving the integrity of the sensitive quinazoline core and ensuring high chemical purity.
How to Synthesize Tucatinib Efficiently
The synthesis of Tucatinib via this novel route is designed for operational simplicity and robustness. The process is divided into three distinct stages: the generation of the isothiocyanate, the formation of the thiourea intermediate, and the final cyclization. Each step utilizes readily available solvents such as DMF, THF, or water, reducing the dependency on exotic or highly regulated chemicals. The reaction parameters are forgiving, with broad temperature windows that allow for flexible manufacturing scheduling. For R&D teams looking to implement this technology, the detailed standardized synthesis steps provided in the patent offer a clear roadmap from gram-scale optimization to multi-ton production.
- Convert the starting amine (Intermediate 15) into an isothiocyanate using a thionating agent and cyanuric chloride.
- React the isothiocyanate with 2-amino-2-methylpropanol (AMP) to form a thiourea intermediate.
- Cyclize the thiourea intermediate using p-toluenesulfonyl chloride and base to form the oxazole ring.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis method offers tangible benefits that extend beyond mere technical feasibility. The primary advantage is the drastic simplification of the supply chain for raw materials. By eliminating the need for specialized triflate reagents, manufacturers can source standard commodity chemicals, thereby reducing exposure to price volatility and supply disruptions. This shift significantly lowers the barrier to entry for production and enhances the overall resilience of the supply network. Furthermore, the improved yield and purification profile mean that less raw material is wasted, directly contributing to cost reduction in pharmaceutical intermediate manufacturing without compromising on quality standards.
- Cost Reduction in Manufacturing: The elimination of expensive coupling reagents and the reduction in purification steps lead to substantial cost savings. The process avoids the use of precious metal catalysts or hazardous fluorinated compounds, which often require specialized waste disposal protocols. By utilizing common reagents like carbon disulfide and tosyl chloride, the variable cost per kilogram of the API is significantly optimized. Additionally, the higher yield means that the fixed costs of production are amortized over a larger output of saleable product, improving the overall margin structure for the manufacturer.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent batch quality, which is crucial for maintaining regulatory compliance and meeting delivery schedules. The use of stable intermediates allows for the potential stocking of key precursors, decoupling the production timeline from the immediate availability of all starting materials. This flexibility enables manufacturers to respond more agilely to market demand spikes. For a reliable tucatinib supplier, this reliability translates into shorter lead times and a stronger commitment to contractual obligations, fostering long-term partnerships with downstream drug developers.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with industrial production in mind. The mild reaction temperatures and ambient pressure requirements simplify the engineering controls needed for large reactors. Moreover, the aqueous workup procedures and the avoidance of heavy metals align with modern green chemistry principles. This reduces the environmental footprint of the manufacturing process and simplifies the permitting process for new production facilities. The ability to scale from pilot plants to commercial volumes without re-engineering the core chemistry is a decisive factor for long-term supply security.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of Tucatinib. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages of this new method. Understanding these details is vital for stakeholders evaluating the feasibility of this technology for their specific supply chain needs. The answers reflect the practical realities of implementing this chemistry in a GMP environment.
Q: How does this new synthesis route improve upon conventional methods?
A: Conventional routes rely on a difficult coupling with oxazole trifluoromethanesulfonate, resulting in low yields (approx. 19%) and scale-up issues. This new method builds the oxazole ring in situ from stable intermediates, significantly improving yield and purification.
Q: What are the critical quality control points in this synthesis?
A: Key control points include the complete conversion of the amine to isothiocyanate in Step S1 and the precise pH adjustment during extraction. The final product consistently achieves HPLC purity greater than 98% after recrystallization.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process utilizes common reagents like carbon disulfide and tosyl chloride, avoiding expensive and hazardous triflate reagents. The mild reaction conditions (0-60°C) and robust workup procedures make it highly scalable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tucatinib Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for life-saving oncology medications. Our team of expert chemists has extensively evaluated the methodology described in CN115650977A and confirmed its potential for high-volume production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Tucatinib meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to leverage this advanced synthesis technology for your pipeline. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments. Let us help you secure a stable, high-quality supply of this essential HER2 inhibitor, enabling you to focus on what matters most: delivering innovative therapies to patients worldwide.
