Optimizing Tucatinib Production: A Novel Pd-Catalyzed Route for High-Purity API Intermediates
Optimizing Tucatinib Production: A Novel Pd-Catalyzed Route for High-Purity API Intermediates
The pharmaceutical industry's demand for potent HER2 inhibitors continues to surge, driving the need for more efficient and scalable manufacturing processes for key intermediates like Tucatinib. Patent CN114230568A introduces a groundbreaking preparation method that addresses critical bottlenecks in the existing synthetic landscape. By innovatively employing a palladium acetylacetonate (Pd(acac)2) catalyst system in conjunction with a specialized ligand L, this technology achieves a highly selective amination reaction that streamlines the production workflow. This technical breakthrough not only enhances the overall yield but also simplifies the downstream processing, making it an attractive option for reliable pharmaceutical intermediate suppliers aiming to optimize their supply chains. The method leverages commercially available starting materials and avoids complex purification techniques, positioning it as a superior alternative for cost reduction in pharmaceutical intermediates manufacturing.
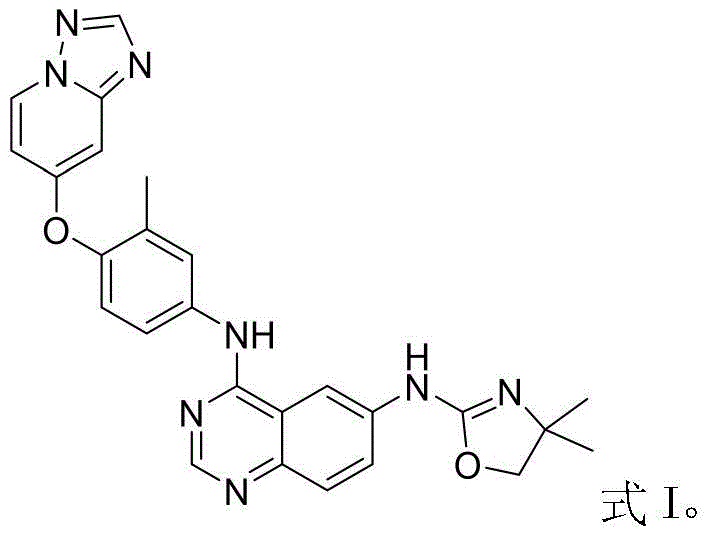
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for Tucatinib often suffer from excessive step counts and inefficient purification protocols that hinder large-scale production. A common conventional strategy involves reducing the nitro group in the precursor to an amino group, followed by a separate halogenation step, and finally a nucleophilic substitution reaction. This multi-step sequence inherently increases the risk of side reactions, particularly when multiple reactive sites are present on the molecular scaffold. Furthermore, the reliance on column chromatography for purification in many legacy methods introduces significant operational costs and solvent waste, creating a bottleneck for commercial scale-up of complex pharmaceutical intermediates. The cumulative yield loss across these additional steps, combined with the logistical burden of handling hazardous halogenating agents, makes conventional routes less economically viable for high-volume manufacturing.
The Novel Approach
In stark contrast, the method disclosed in CN114230568A revolutionizes the final assembly of the Tucatinib molecule through a direct palladium-catalyzed amination. This approach bypasses the unnecessary reduction-halogenation-substitution sequence, effectively condensing the synthesis into a more concise four-step pathway. The use of Pd(acac)2 and Ligand L ensures that the reaction site remains singular and highly specific, minimizing the formation of regioisomers and other impurities. By integrating this catalytic step, the process achieves a remarkable simplification of post-treatment procedures, relying solely on pulping purification rather than chromatography. This strategic shift not only accelerates the production timeline but also aligns perfectly with the goals of reducing lead time for high-purity pharmaceutical intermediates, offering a robust solution for modern API manufacturing.
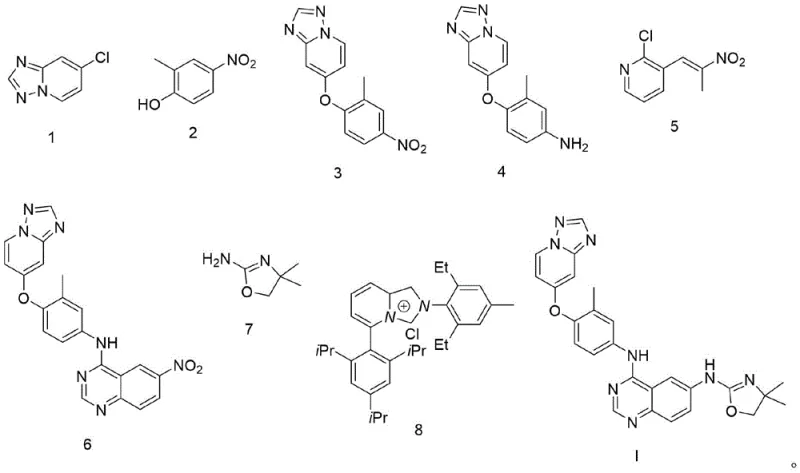
Mechanistic Insights into Pd(acac)2-Catalyzed Amination
The core of this technological advancement lies in the sophisticated catalytic cycle facilitated by the Pd(acac)2 and Ligand L complex. In the critical fourth step, the catalyst activates the aryl halide bond in compound 6, enabling a smooth cross-coupling with the amine component (compound 7). The specific choice of Ligand L is paramount, as it stabilizes the palladium center and modulates its electronic properties to favor the oxidative addition and reductive elimination steps essential for C-N bond formation. This mechanistic precision ensures that the reaction proceeds with high turnover numbers even under relatively mild heating conditions of 90°C in dioxane. The presence of K3PO4·3H2O serves as an effective base to scavenge the acid byproduct, driving the equilibrium towards the desired product without degrading the sensitive functional groups present on the quinazoline core.
From an impurity control perspective, this catalytic system offers distinct advantages over non-catalytic nucleophilic substitutions. The high regioselectivity prevents the amine from attacking unintended positions on the heterocyclic ring, which is a common issue in uncatalyzed thermal reactions. Additionally, the mild reaction conditions prevent the decomposition of the nitro-containing intermediate prior to the intended transformation, although in this specific route, the nitro group is strategically positioned to remain inert until the final stages or is managed through the specific reactivity profile of the catalyst. The result is a crude reaction mixture with exceptionally high purity, often exceeding 99% HPLC purity after simple workup, thereby validating the efficacy of this high-purity pharmaceutical intermediate synthesis strategy.
How to Synthesize Tucatinib Efficiently
The synthesis of Tucatinib via this patented route is designed for operational simplicity and scalability, making it ideal for transfer from laboratory to pilot plant. The process begins with the etherification of commercially available chloro-pyrimidine derivatives, followed by a standard catalytic hydrogenation to install the aniline moiety. The subsequent coupling with the nitro-phenol derivative sets the stage for the final, crucial amination step. Detailed standard operating procedures regarding stoichiometry, temperature ramps, and safety protocols are essential for maximizing the efficiency of the Pd-catalyzed step. For a comprehensive breakdown of the standardized synthesis steps, please refer to the guide below.
- Perform etherification of compound 1 with compound 2 using K2CO3 in DMF at 100°C to obtain compound 3.
- Reduce the nitro group of compound 3 using Pd/C and hydrogen in methanol to yield compound 4.
- React compound 4 with compound 5 using K2CO3 in DMF at 80°C to form the nitro-intermediate compound 6.
- Execute the key amination of compound 6 with compound 7 using Pd(acac)2, Ligand L, and K3PO4 in dioxane at 90°C to finalize Tucatinib.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route translates into tangible operational efficiencies and risk mitigation. The elimination of column chromatography is perhaps the most significant cost driver, as it removes the need for large volumes of high-purity silica gel and expensive elution solvents. This change alone drastically simplifies the waste management profile of the facility, reducing the environmental footprint and associated disposal costs. Furthermore, the high yields reported in each step—consistently hovering around or above 90%—mean that less raw material is required to produce the same amount of final API, directly impacting the cost of goods sold (COGS). The reliance on low-price commercial starting materials further insulates the supply chain from volatility in specialty reagent markets.
- Cost Reduction in Manufacturing: The streamlined four-step sequence inherently lowers labor and utility costs by reducing the total number of unit operations. By avoiding the multi-step reduction-halogenation-detour found in older methods, the process saves significant reactor time and energy consumption. The switch to pulping purification using common solvent mixtures like petroleum ether and ethyl acetate replaces costly chromatographic resins, leading to substantial savings in consumable expenses. These cumulative efficiencies allow for a more competitive pricing structure without compromising on the quality of the pharmaceutical intermediates supplied to downstream partners.
- Enhanced Supply Chain Reliability: The use of robust, commercially available starting materials such as simple chloro-heterocycles and nitro-phenols ensures a stable supply base that is not dependent on custom synthesis of exotic precursors. The high tolerance of the catalytic system to minor variations in reaction conditions adds a layer of process robustness, reducing the likelihood of batch failures that could disrupt delivery schedules. This reliability is crucial for maintaining continuous production lines for critical oncology medications, ensuring that API intermediate availability remains consistent even during periods of high market demand.
- Scalability and Environmental Compliance: The process is inherently designed for scale, utilizing standard solvents like DMF, methanol, and dioxane which are well-understood in industrial settings. The avoidance of hazardous halogenating agents in the final step reduces the safety risks associated with scaling up exothermic reactions. Moreover, the simplified workup generates less hazardous waste, facilitating easier compliance with increasingly stringent environmental regulations. This makes the technology not just economically sound but also sustainable for long-term commercial scale-up initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Tucatinib synthesis method. These insights are derived directly from the experimental data and comparative examples provided in the patent literature, offering clarity on yield expectations, purification strategies, and catalyst performance. Understanding these nuances is vital for technical teams evaluating the feasibility of adopting this route for their own manufacturing portfolios.
Q: What is the key advantage of the Pd(acac)2 catalytic system in Tucatinib synthesis?
A: The use of Pd(acac)2 with Ligand L in the final amination step allows for direct coupling without the need for prior nitro reduction and halogenation, significantly shortening the synthetic route and improving regioselectivity.
Q: How does this method handle purification compared to traditional routes?
A: This process eliminates the need for expensive and time-consuming column chromatography. Instead, it utilizes simple pulping purification with petroleum ether and ethyl acetate, which drastically reduces solvent costs and processing time.
Q: What yields can be expected from this optimized synthetic route?
A: The patent data indicates consistently high yields across all four steps, with the final amination step achieving over 90% yield and HPLC purity exceeding 99.7%, ensuring robust material throughput for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tucatinib Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for life-saving oncology treatments like Tucatinib. Our team of expert chemists has extensively analyzed the Pd-catalyzed amination technology described in CN114230568A and is fully prepared to implement this advanced methodology. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of intermediate we deliver adheres to the highest global standards.
We invite you to collaborate with us to leverage this cutting-edge synthetic route for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and detailed route feasibility assessments to demonstrate how our optimized manufacturing capabilities can enhance your supply chain resilience and drive down your overall production costs.
