Advanced Catalytic Synthesis of Mono-carboxyfunctionalized Dialkylphosphinic Acids for Industrial Flame Retardants
Advanced Catalytic Synthesis of Mono-carboxyfunctionalized Dialkylphosphinic Acids for Industrial Flame Retardants
The global demand for high-performance, halogen-free flame retardants has necessitated a paradigm shift in the synthesis of organophosphorus intermediates. Patent CN102177166A introduces a groundbreaking multi-step catalytic methodology for producing mono-carboxyfunctionalized dialkylphosphinic acids, esters, and salts. This technology addresses critical bottlenecks in traditional manufacturing, specifically the reliance on hazardous halogenated reagents and the difficulty in controlling exothermic reactions during phosphorus-halogen bond formation. By leveraging transition metal catalysis to couple phosphinic acid sources with olefins and acetylenes, this route offers a cleaner, more controllable pathway to high-value polymer additives. For R&D directors and procurement strategists, understanding this mechanistic evolution is key to securing a sustainable supply chain for next-generation flame-retardant polymers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
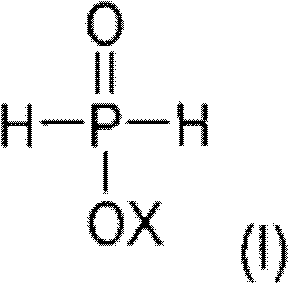
Historically, the industrial preparation of dialkylphosphinic acid esters has relied heavily on the reaction of phosphorus trichloride with haloalkanes in the presence of aluminum chloride. This classical approach is fraught with significant technical and safety challenges. The reaction is highly exothermic and difficult to control on a large scale, posing substantial risks for thermal runaway incidents in commercial plants. Furthermore, the use of phosphorus trichloride and haloalkanes generates stoichiometric amounts of corrosive hydrogen halides and chlorinated byproducts, creating a severe burden on waste treatment facilities and environmental compliance teams. The resulting crude products often contain complex impurity profiles that are difficult to separate, leading to lower yields and compromised purity in the final flame retardant additives. Additionally, the inability to easily prepare halogen-free products limits their application in high-end electronics and automotive sectors where halogen content is strictly regulated.
The Novel Approach
In stark contrast, the method disclosed in CN102177166A utilizes a catalytic addition strategy that fundamentally alters the reaction landscape. Instead of substitution reactions involving halogens, this process employs the addition of a phosphinic acid source to activated olefinic compounds. This shift eliminates the generation of corrosive halogenated waste streams at the source. The process is characterized by four distinct catalytic stages: initial alkylation with olefins, functionalization with acetylenic compounds to introduce vinyl groups, hydrocyanation to introduce nitrile functionality, and final hydrolysis to yield the carboxylic acid. This modular approach allows for precise control over the molecular architecture of the final product. By avoiding the harsh conditions of the Friedel-Crafts type alkylation used in older methods, this novel route enables the synthesis of a broader range of derivatives with higher selectivity and significantly reduced environmental impact.
Mechanistic Insights into Transition Metal Catalyzed Functionalization
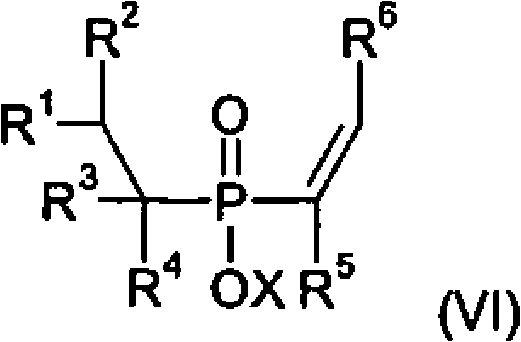
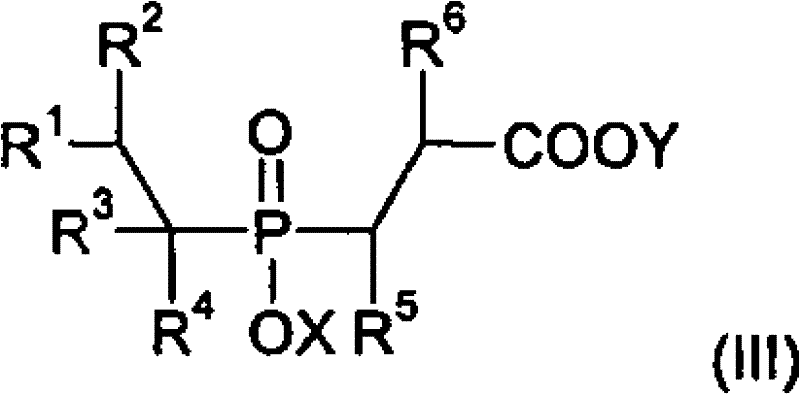
The core innovation of this technology lies in the sophisticated use of transition metal catalysts to drive carbon-phosphorus and carbon-carbon bond formations under mild conditions. The initial step involves the reaction of a phosphinic acid source with olefins in the presence of Catalyst A, typically comprising metals from groups 7, 8, or 9 such as Ruthenium, Rhodium, or Palladium. This catalytic cycle facilitates the anti-Markovnikov or Markovnikov addition of the P-H bond across the double bond of the olefin, generating alkylphosphonic acid derivatives with high regioselectivity. The subsequent step involves the reaction of these intermediates with acetylenic compounds using Catalyst B. This step is crucial for introducing the vinyl functionality required for further chain extension. The catalyst system, often involving phosphine ligands to stabilize the metal center, ensures that the acetylene adds efficiently without polymerizing, a common side reaction in acetylene chemistry. This level of control is essential for maintaining the purity required for high-performance polymer applications.
Following the vinyl functionalization, the process employs a hydrocyanation step using Catalyst C to convert the vinyl group into a nitrile group. This transformation is pivotal as it sets up the molecule for the final conversion to the carboxylic acid. The use of hydrogen cyanide sources in the presence of Nickel or Palladium catalysts allows for the efficient addition of the CN group across the double bond. Finally, Catalyst D, which can be a simple acid or base, mediates the hydrolysis of the nitrile to the carboxylic acid. This final step is robust and scalable, allowing for the production of the free acid or its corresponding metal salts depending on the reaction conditions. The ability to tune the catalyst ligands and metal centers at each stage provides R&D teams with a powerful toolkit for optimizing yield and minimizing impurities, ensuring that the final product meets stringent quality specifications for flame retardancy and thermal stability.
How to Synthesize Mono-carboxyfunctionalized Dialkylphosphinic Acids Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing these high-value intermediates. The process begins with the careful selection of the phosphinic acid source and the olefin substrate, followed by sequential catalytic transformations. Each step requires specific attention to temperature, pressure, and catalyst loading to maximize space-time yield. The flexibility of the solvent system, which can range from water to various organic alcohols and hydrocarbons, allows manufacturers to adapt the process to existing infrastructure. The following guide summarizes the critical operational parameters for implementing this technology effectively.
- React a phosphinic acid source with olefins in the presence of a transition metal catalyst A to generate alkylphosphonic acid derivatives.
- Functionalize the alkylphosphonic acid derivative with acetylenic compounds using catalyst B to form mono-functionalized vinyl derivatives.
- Perform hydrocyanation on the vinyl derivative using a hydrogen cyanide source and catalyst C to introduce the nitrile group.
- Hydrolyze the nitrile derivative under acidic or basic conditions with catalyst D to yield the final mono-carboxyfunctionalized product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic technology offers tangible strategic advantages beyond mere technical feasibility. The shift away from halogenated raw materials mitigates the volatility associated with the supply of chlorine and bromine derivatives, which are often subject to strict regulatory scrutiny and price fluctuations. By utilizing commodity chemicals like ethylene, acetylene, and simple phosphinic acids, manufacturers can secure a more stable and cost-effective raw material base. The elimination of corrosive byproducts also translates to reduced maintenance costs for reactor vessels and piping, extending the lifespan of capital equipment. Furthermore, the simplified purification steps reduce the consumption of solvents and energy required for distillation and crystallization, contributing to a lower overall cost of goods sold (COGS).
- Cost Reduction in Manufacturing: The catalytic nature of the process means that expensive transition metals are used in minute quantities relative to the substrate, and advanced ligand systems allow for high turnover numbers. This drastically reduces the raw material cost per kilogram of product compared to stoichiometric halogenation methods. Additionally, the absence of halogenated waste eliminates the need for expensive neutralization and disposal procedures, further driving down operational expenditures. The high selectivity of the catalysts minimizes the formation of byproducts, leading to higher yields and less material loss during purification, which directly impacts the bottom line.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, such as olefins and acetylenes, are produced on a massive global scale for the petrochemical industry, ensuring a consistent and reliable supply. Unlike specialized halogenated intermediates that may have limited suppliers, these feedstocks are readily available from multiple sources, reducing the risk of supply chain disruptions. The robustness of the catalytic system also allows for flexible production scheduling, as the reactions can be tuned to accommodate variations in feedstock quality without compromising the final product specifications, thereby enhancing overall supply chain resilience.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with continuous flow and large-batch reactor operations in mind. The mild reaction conditions reduce the safety risks associated with high-pressure and high-temperature operations, facilitating easier permitting and regulatory approval for new production lines. From an environmental perspective, the halogen-free nature of the process aligns perfectly with global trends towards green chemistry and sustainability. This compliance not only future-proofs the manufacturing facility against tightening environmental regulations but also enhances the marketability of the final flame retardant products to eco-conscious consumers and OEMs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and chemical versatility of the process for potential partners and licensees.
Q: How does this catalytic method improve upon traditional halogenated synthesis routes?
A: Traditional methods often rely on phosphorus trichloride and haloalkanes, generating toxic byproducts and requiring complex purification. This patented catalytic route utilizes olefins and acetylenes, eliminating halogenated waste streams and significantly simplifying downstream processing.
Q: What represents the primary cost driver in scaling this synthesis?
A: The primary cost drivers are the transition metal catalysts (such as Palladium, Nickel, or Rhodium) and the efficient recovery of solvents. However, the high atom economy of the addition reactions and the ability to recycle catalyst supports mitigate these costs compared to stoichiometric halogenation methods.
Q: Is this process suitable for producing metal salts for polymer compounding?
A: Yes, the process explicitly includes a step for converting the free acid into metal salts (such as Aluminum, Zinc, or Calcium salts) which are directly compatible with thermoplastic compounding processes like extrusion and injection molding.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Mono-carboxyfunctionalized Dialkylphosphinic Acids Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this catalytic technology in the realm of polymer additives. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the nuanced requirements of transition metal catalysis, ensuring that every batch of mono-carboxyfunctionalized dialkylphosphinic acids meets stringent purity specifications. Our rigorous QC labs employ advanced analytical techniques to verify the absence of halogenated impurities and confirm the structural integrity of the phosphorus-carbon backbone, guaranteeing performance consistency for your flame-retardant formulations.
We invite you to collaborate with us to leverage this innovative synthesis route for your specific application needs. Whether you require custom metal salts for polyamide compounding or specific ester derivatives for polyester modification, our technical team is ready to assist. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how partnering with us can optimize your supply chain and accelerate your time to market.
