Advanced Pd-Catalyzed Multicomponent Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Commercial Scale-Up
Advanced Pd-Catalyzed Multicomponent Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and safe methodologies for constructing complex heterocyclic scaffolds, which serve as critical backbones for bioactive molecules. A significant breakthrough in this domain is detailed in Chinese Patent CN115353511A, which discloses a novel multi-component method for synthesizing carbonyl-bridged biheterocyclic compounds. This technology represents a paradigm shift from traditional, hazardous carbonylation techniques to a safer, more efficient cascade reaction protocol. By leveraging a palladium-catalyzed system that generates carbon monoxide in-situ, this invention eliminates the need for handling toxic CO gas while maintaining high reaction efficiency and broad substrate tolerance. For R&D directors and procurement managers alike, this development offers a compelling pathway to access high-purity indolinone-imidazole derivatives, which are valuable intermediates in drug discovery and functional material science.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of carbonyl-bridged biheterocyclic systems has relied heavily on direct coupling reactions or transition metal-catalyzed processes that often necessitate the use of gaseous carbon monoxide. The reliance on external CO sources presents severe logistical and safety challenges, including the requirement for specialized high-pressure equipment, rigorous leak detection systems, and extensive safety training for personnel. Furthermore, traditional methods frequently suffer from poor atom economy, harsh reaction conditions involving high temperatures and pressures, and limited functional group compatibility, which can lead to significant impurity profiles and difficult downstream purification processes. These factors collectively drive up the cost of goods sold (COGS) and extend the lead time for high-purity pharmaceutical intermediates, creating bottlenecks in the supply chain for complex API manufacturing.
The Novel Approach
In stark contrast, the methodology described in patent CN115353511A introduces a streamlined, one-pot multicomponent reaction that elegantly circumvents these traditional pitfalls. By utilizing a mixture of formic acid and acetic anhydride as a safe, liquid carbon monoxide surrogate, the process allows for the generation of CO directly within the reaction medium under mild conditions. This innovation not only drastically simplifies the reactor setup by removing the need for gas cylinders but also enables the reaction to proceed at a remarkably low temperature of 30°C. The use of readily available starting materials—trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivatives—further enhances the economic viability of the process. This approach ensures that the commercial scale-up of complex pharmaceutical intermediates can be achieved with reduced capital expenditure on safety infrastructure and lower operational risks.
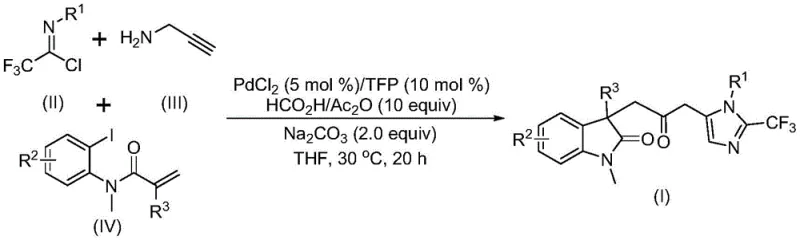
The core of this technological advancement lies in its ability to construct multiple chemical bonds simultaneously in a single operation. As illustrated in the reaction scheme, the convergence of three distinct building blocks leads to the formation of the target carbonyl-bridged biheterocyclic structure (Formula I) with high precision. This telescoped synthesis minimizes the number of isolation steps, thereby reducing solvent consumption and waste generation, which aligns perfectly with modern green chemistry principles and environmental compliance standards required by global regulatory bodies.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
From a mechanistic perspective, this transformation is a sophisticated example of palladium-catalyzed cascade chemistry that integrates Heck-type cyclization with carbonylation. The catalytic cycle likely initiates with the oxidative addition of a zero-valent palladium species into the carbon-iodine bond of the acrylamide substrate. This is followed by an intramolecular Heck reaction, which forms a divalent alkyl-palladium intermediate and establishes the initial cyclic framework. Subsequently, the carbon monoxide generated from the decomposition of the formic acid/acetic anhydride mixture inserts into the palladium-carbon bond, yielding a crucial acyl-palladium intermediate. This step is pivotal as it introduces the carbonyl bridge that defines the final molecular architecture.
Concurrently, the trifluoroethylimidoyl chloride and propargylamine undergo a base-promoted intermolecular reaction to form a trifluoroacetamidine species, which subsequently isomerizes. The acyl-palladium intermediate then activates this trifluoroacetamidine compound, catalyzing an intramolecular cyclization that finalizes the formation of the imidazole ring fused to the indolinone core. This intricate dance of bond formations occurs with remarkable chemoselectivity, tolerating a wide range of substituents on the aromatic rings, including halogens, alkyl groups, and electron-withdrawing trifluoromethyl moieties. Such mechanistic robustness ensures that the impurity profile remains manageable, a critical factor for R&D teams focused on purity specifications and regulatory filing requirements.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
Implementing this synthesis route requires careful attention to reagent stoichiometry and reaction parameters to maximize yield and minimize side products. The patent outlines a generalized procedure where the catalyst system, consisting of palladium chloride and trifurylphosphine, is combined with sodium carbonate as the base in an aprotic solvent like tetrahydrofuran. The specific molar ratios are optimized to ensure complete conversion, typically employing a slight excess of the amine and acrylamide components relative to the imidoyl chloride. The reaction is maintained at a constant mild temperature to facilitate the slow release of carbon monoxide, preventing pressure buildup while ensuring steady progression of the carbonylation step.
- Combine palladium chloride catalyst, trifurylphosphine ligand, sodium carbonate base, and the formic acid/acetic anhydride CO source mixture in an organic solvent such as THF.
- Add the three key substrates: trifluoroethylimidoyl chloride, propargylamine, and the specific acrylamide derivative to the reaction vessel under stirring.
- Maintain the reaction mixture at a mild temperature of 30°C for 12 to 20 hours, followed by filtration and column chromatography purification to isolate the target biheterocycle.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers tangible strategic advantages beyond mere chemical elegance. The elimination of toxic carbon monoxide gas from the supply chain removes a significant hazard classification from the manufacturing process, which translates directly into reduced insurance premiums and simplified logistics for raw material transport. Furthermore, the use of commodity chemicals such as formic acid and acetic anhydride as CO surrogates ensures a stable and cost-effective supply of reagents, mitigating the risk of shortages associated with specialized gases. This stability is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of downstream pharmaceutical clients.
- Cost Reduction in Manufacturing: The economic benefits of this process are multifaceted, stemming primarily from the simplification of the reaction infrastructure. By obviating the need for high-pressure autoclaves and gas handling systems, the capital investment required for setting up production lines is significantly lowered. Additionally, the mild reaction temperature of 30°C results in substantial energy savings compared to traditional reflux conditions, reducing the overall utility costs per kilogram of product. The high atom economy of the multicomponent reaction also means less waste disposal cost, contributing to a leaner and more profitable manufacturing model for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The robustness of this synthetic method against variations in substrate electronics ensures consistent batch-to-batch quality, which is vital for long-term supply contracts. Since the starting materials are commercially available and the reaction conditions are forgiving, the risk of batch failure due to minor fluctuations in raw material quality is minimized. This reliability allows supply chain planners to optimize inventory levels with greater confidence, reducing the need for excessive safety stock and freeing up working capital. Moreover, the scalability demonstrated in the patent suggests that transitioning from pilot plant to full commercial production can be achieved with minimal process re-engineering.
- Scalability and Environmental Compliance: As environmental regulations become increasingly stringent globally, the ability to demonstrate a greener synthesis route is a competitive differentiator. This method's reliance on in-situ CO generation and its operation in standard organic solvents facilitates easier waste treatment and solvent recovery. The process avoids the generation of heavy metal waste streams often associated with stoichiometric carbonylation reagents, simplifying the effluent treatment process. Consequently, manufacturers can achieve compliance with environmental standards more easily, avoiding potential fines and reputational damage while positioning themselves as sustainable partners for eco-conscious pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential adopters.
Q: What is the primary safety advantage of this synthesis method compared to traditional carbonylation?
A: Unlike conventional methods that require handling toxic and hazardous carbon monoxide gas cylinders, this patented process utilizes a formic acid and acetic anhydride mixture to generate carbon monoxide in-situ, significantly enhancing operational safety and reducing regulatory compliance burdens.
Q: What are the typical reaction conditions required for this transformation?
A: The reaction proceeds efficiently under mild conditions, specifically at a temperature of 30°C in tetrahydrofuran (THF) solvent, utilizing a PdCl2/TFP catalyst system over a period of 12 to 20 hours.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the patent explicitly demonstrates the scalability of this method to gram-level synthesis with high yields and excellent substrate compatibility, indicating strong potential for commercial scale-up in pharmaceutical intermediate manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN115353511A for accelerating drug discovery pipelines. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are seamlessly translated into industrial reality. Our state-of-the-art facilities are equipped to handle sensitive palladium-catalyzed reactions with stringent purity specifications, supported by rigorous QC labs that guarantee every batch meets the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this efficient route can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a reliable supply of these critical biheterocyclic building blocks for your next generation of therapeutics.
