Scalable Synthesis of 3,4,5-Trisubstituted 1,2,4-Triazoles: A Metal-Free Route for API Intermediates
Scalable Synthesis of 3,4,5-Trisubstituted 1,2,4-Triazoles: A Metal-Free Route for API Intermediates
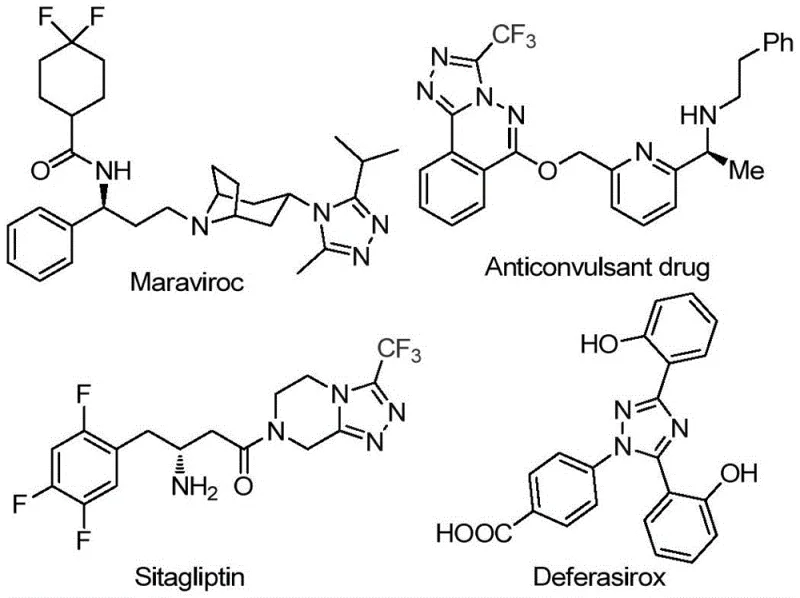
The pharmaceutical industry continuously seeks robust and cost-effective methodologies for constructing nitrogen-containing heterocycles, which serve as critical scaffolds in numerous active pharmaceutical ingredients (APIs). Patent CN113105402B discloses a highly efficient preparation method for 3,4,5-trisubstituted 1,2,4-triazole compounds, addressing key limitations in current synthetic routes. This innovation is particularly relevant given the prevalence of the 1,2,4-triazole motif in blockbuster drugs such as Maraviroc, Sitagliptin, and Deferasirox, as illustrated in the structural diversity of bioactive molecules below.
The disclosed technology leverages a non-metallic iodine-promoted strategy that eliminates the dependency on precious metal catalysts, thereby simplifying the purification process and reducing the environmental footprint. For R&D directors and procurement managers alike, this represents a significant opportunity to optimize the supply chain for high-purity pharmaceutical intermediates. By utilizing cheap and readily available starting materials like aryl ethanones and trifluoroethylimine hydrazides, the method ensures a stable supply of precursors while maintaining high reaction efficiency under relatively mild conditions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic pathways for constructing polysubstituted 1,2,4-triazoles often rely heavily on transition metal catalysis, such as copper or palladium complexes, which introduce significant challenges for large-scale manufacturing. These conventional methods typically require stringent anhydrous and oxygen-free conditions to prevent catalyst deactivation, necessitating specialized equipment and inert gas atmospheres that drive up operational costs. Furthermore, the presence of heavy metal residues in the final product poses a severe regulatory hurdle, requiring extensive and expensive purification steps to meet the strict impurity limits set by pharmacopoeias. The complexity of ligand synthesis and the sensitivity of these catalysts to functional group tolerance further limit the versatility of these older protocols, making them less ideal for the rapid diversification required in modern drug discovery.
The Novel Approach
In stark contrast, the methodology outlined in patent CN113105402B employs a metal-free system driven by elemental iodine and dimethyl sulfoxide (DMSO). This novel approach utilizes an iodine/Kornblum oxidation sequence to generate reactive intermediates in situ, which subsequently undergo tandem cyclization with trifluoroethylimine hydrazides. The general reaction scheme demonstrates the convergence of aryl ethanones and hydrazides to form the target triazole core with high atom economy.
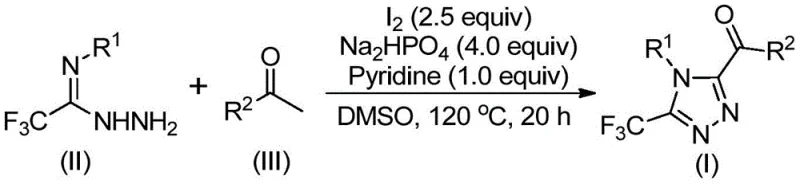
This strategy operates effectively without the need for rigorous exclusion of moisture or oxygen, significantly lowering the barrier for implementation in standard chemical plants. The use of inexpensive reagents like sodium dihydrogen phosphate and pyridine as additives further enhances the economic viability of the process. By avoiding toxic heavy metals entirely, the downstream processing is drastically simplified, allowing for a more streamlined workflow that is highly attractive for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Iodine-Promoted Cyclization
The mechanistic pathway of this transformation is a sophisticated interplay of oxidation and condensation reactions facilitated by the unique properties of the iodine-DMSO system. Initially, the aryl ethanone undergoes alpha-iodination followed by a Kornblum oxidation mediated by DMSO to generate an aryl diketone intermediate. This oxidative step is crucial as it activates the carbonyl carbon for subsequent nucleophilic attack. The generated diketone then reacts with the trifluoroethylimine hydrazide through a dehydration condensation to form a hydrazone intermediate. This specific sequence ensures that the trifluoromethyl group, known for enhancing metabolic stability and lipophilicity in drug candidates, is seamlessly incorporated into the heterocyclic framework.
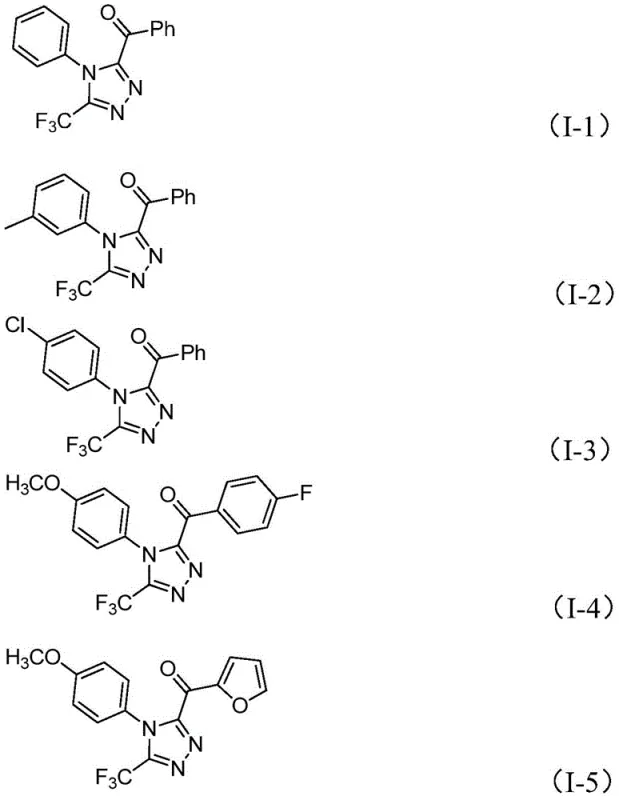
Following the formation of the hydrazone, the reaction proceeds through an intramolecular cyclization promoted by the combined action of iodine and the base system (sodium dihydrogen phosphate/pyridine). This cyclization step closes the five-membered triazole ring, establishing the final 3,4,5-trisubstituted architecture. The tolerance of this mechanism towards various substituents on both the aryl ketone and the hydrazide components is exceptional, accommodating electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups such as chloro and trifluoromethyl. This broad substrate scope is evidenced by the successful synthesis of diverse derivatives, including those with heteroaryl substitutions, confirming the robustness of the catalytic cycle.
How to Synthesize 3,4,5-Trisubstituted 1,2,4-Triazole Efficiently
The practical execution of this synthesis is designed for operational simplicity, making it accessible for both laboratory optimization and pilot plant operations. The process involves a sequential addition of reagents in a single pot, minimizing unit operations and solvent usage. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up.
- Mix aryl ethyl ketone and iodine in DMSO, heating to 90-110°C for 4-6 hours to initiate oxidation.
- Add trifluoroethylimide hydrazide, sodium dihydrogen phosphate, pyridine, and additional iodine to the mixture.
- Heat the reaction to 110-130°C for 12-20 hours, then filter and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iodine-promoted methodology offers tangible strategic advantages over traditional metal-catalyzed routes. The primary benefit lies in the drastic simplification of the raw material portfolio; elemental iodine, DMSO, and simple aryl ketones are commodity chemicals with stable global supply chains, unlike specialized organometallic catalysts that may suffer from geopolitical supply risks or price volatility. This shift to commodity reagents inherently stabilizes the cost structure of the manufacturing process.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and their associated ligands results in substantial direct material cost savings. Moreover, the absence of heavy metals removes the necessity for complex scavenging resins or repeated recrystallization steps dedicated to metal removal, which significantly reduces processing time and waste disposal costs. The use of DMSO, a high-boiling polar aprotic solvent, allows for high-temperature reactions that drive kinetics favorably without requiring high-pressure equipment, further optimizing energy consumption and capital expenditure.
- Enhanced Supply Chain Reliability: Since the starting materials such as aryl ethanones and trifluoroethylimine hydrazides are commercially available and easy to synthesize from bulk chemicals, the risk of supply disruption is minimized. The method does not rely on proprietary or single-source catalysts, granting manufacturers the flexibility to source reagents from multiple vendors. This redundancy is critical for maintaining continuous production schedules and meeting the just-in-time delivery demands of downstream API manufacturers.
- Scalability and Environmental Compliance: The protocol has been validated to scale easily to the gram level and beyond, with reaction conditions that are safe and manageable in large reactors. The avoidance of toxic heavy metals aligns perfectly with increasingly stringent environmental regulations regarding wastewater discharge and residual solvents. This 'green chemistry' aspect not only reduces the environmental compliance burden but also enhances the marketability of the final intermediate to eco-conscious pharmaceutical clients who prioritize sustainable supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation to provide clarity for potential partners.
Q: Does this synthesis require expensive transition metal catalysts?
A: No, the method described in patent CN113105402B utilizes elemental iodine as a promoter, completely avoiding the need for costly and toxic heavy metal catalysts often found in traditional triazole synthesis.
Q: What are the typical reaction conditions for this transformation?
A: The reaction proceeds in dimethyl sulfoxide (DMSO) using a two-stage heating protocol: initially at 90-110°C for oxidation, followed by 110-130°C for cyclization, without requiring strict anhydrous or oxygen-free conditions.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the patent explicitly states that the process is simple to operate, uses cheap and readily available raw materials, and has been successfully demonstrated to scale easily to the gram level, indicating strong potential for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trisubstituted 1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthetic route for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial manufacturing is seamless. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 3,4,5-trisubstituted 1,2,4-triazole meets the highest quality standards required for drug substance synthesis.
We invite you to collaborate with us to leverage this efficient technology for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you secure a reliable supply of these critical heterocyclic building blocks.
