Advanced Palladium-Catalyzed Synthesis of 1,2,4-Triazole-3-one Intermediates for Commercial Scale-up
Advanced Palladium-Catalyzed Synthesis of 1,2,4-Triazole-3-one Intermediates for Commercial Scale-up
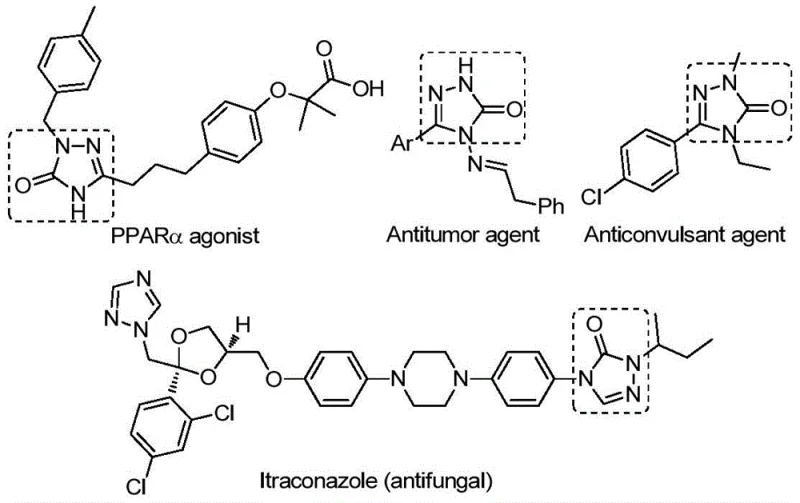
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing nitrogen-containing heterocycles, particularly those with proven biological significance. Patent CN112538054B introduces a groundbreaking preparation method for 1,2,4-triazole-3-one compounds, a privileged scaffold found in numerous bioactive molecules ranging from antifungal agents to antitumor drugs. This innovation addresses critical bottlenecks in traditional synthesis by employing a transition metal palladium-catalyzed carbonylation tandem cyclization strategy. By utilizing cheap and readily available chlorohydrazones and sodium azide as starting materials, coupled with a solid carbon monoxide substitute, this process offers a safer and more efficient pathway for producing high-purity pharmaceutical intermediates. The technology represents a significant leap forward for reliable pharmaceutical intermediate suppliers aiming to optimize their production pipelines while maintaining stringent quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,2,4-triazole-3-one derivatives has been plagued by significant operational challenges that hinder large-scale manufacturing efficiency. Conventional literature reports typically rely on methods such as the cyclization of benzoyl hydrazide with urea under strong basic conditions like potassium hydroxide, or the tandem cyclization of hydrazides with isocyanates. These traditional routes often suffer from harsh reaction conditions that require extreme temperatures or corrosive reagents, posing safety risks and increasing equipment maintenance costs. Furthermore, many existing protocols necessitate the pre-activation of substrates, adding unnecessary synthetic steps that reduce overall atom economy and increase waste generation. The narrow substrate scope of these older methods frequently results in low yields when attempting to introduce diverse functional groups, limiting the chemical space available for drug discovery teams exploring structure-activity relationships.
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent utilizes a sophisticated palladium-catalyzed system that dramatically simplifies the synthetic workflow while expanding substrate tolerance. By employing tridibenzylideneacetone dipalladium (Pd2(dba)3) alongside the Xantphos ligand, the reaction achieves high efficiency through a carbonylation tandem cyclization mechanism that proceeds smoothly at moderate temperatures. A key innovation is the use of TFBen (1,3,5-tricarboxylic acid phenol ester) as a safe, solid carbon monoxide substitute, which eliminates the hazards associated with handling high-pressure CO gas cylinders in a production environment. This approach allows for the direct conversion of chlorohydrazones and sodium azide into the target heterocycles with excellent yields, as demonstrated by the successful synthesis of various substituted derivatives including those with bulky tert-butyl or electron-deficient fluoro groups. The operational simplicity and broad compatibility make this an ideal solution for cost reduction in API manufacturing where process robustness is paramount.

Mechanistic Insights into Pd-Catalyzed Carbonylation Cyclization
The mechanistic pathway of this transformation involves a intricate sequence of organometallic steps that ensure high selectivity and conversion rates. The reaction is initiated by the oxidative addition of the palladium catalyst into the carbon-chlorine bond of the chlorohydrazone substrate, forming a reactive divalent palladium intermediate. Subsequently, TFBen decomposes under the heating conditions to release carbon monoxide in situ, which then inserts into the carbon-palladium bond to generate an acyl palladium species. This acyl intermediate reacts with sodium azide to form an acyl azide compound, which undergoes a thermally induced Curtius rearrangement to produce a highly reactive isocyanate intermediate. Finally, an intramolecular nucleophilic addition occurs where the nitrogen atom attacks the electrophilic carbon of the isocyanate, closing the ring to yield the final 1,2,4-triazole-3-one structure. Understanding this detailed catalytic cycle is crucial for R&D directors aiming to troubleshoot potential impurities or optimize reaction parameters for specific substrate classes.
Impurity control in this system is inherently managed by the specificity of the palladium catalyst and the mild nature of the reagents used. Unlike strong base-mediated cyclizations that can lead to hydrolysis or polymerization side products, this neutral to mildly acidic environment preserves sensitive functional groups such as halogens and ethers. The use of sodium azide in slight excess ensures complete consumption of the chlorohydrazone starting material, minimizing the presence of unreacted precursors in the crude mixture. Furthermore, the choice of 1,4-dioxane as the preferred solvent enhances the solubility of all reactants and stabilizes the transition states, leading to cleaner reaction profiles that simplify downstream purification. For procurement managers, this means that the resulting crude product requires less intensive purification efforts, directly translating to reduced solvent consumption and lower waste disposal costs during the commercial scale-up of complex heterocycles.
How to Synthesize 1,2,4-Triazole-3-one Efficiently
The practical execution of this synthesis protocol is designed to be straightforward and adaptable to standard laboratory or pilot plant equipment without requiring specialized high-pressure reactors. The process begins by charging a reaction vessel with the palladium catalyst, ligand, carbon monoxide substitute, chlorohydrazone, and sodium azide in an appropriate organic solvent. The mixture is then heated to a temperature range of 100 to 120 degrees Celsius and maintained under stirring for a duration of 16 to 30 hours to ensure full conversion. Detailed standardized synthesis steps see the guide below.
- Combine Pd2(dba)3 catalyst, Xantphos ligand, TFBen, chlorohydrazone, and sodium azide in an organic solvent like 1,4-dioxane.
- Heat the reaction mixture to 100-120°C and maintain stirring for 16 to 30 hours to ensure complete conversion.
- Perform post-treatment including filtration and silica gel mixing, followed by column chromatography purification to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, this patented method offers transformative benefits that address common pain points in the sourcing and production of specialty chemical intermediates. The reliance on commercially available and inexpensive starting materials such as sodium azide and chlorohydrazones ensures a stable supply chain that is not vulnerable to the fluctuations often seen with exotic or proprietary reagents. The elimination of high-pressure carbon monoxide gas removes a major safety barrier and regulatory hurdle, allowing for faster permitting and easier integration into existing multipurpose manufacturing facilities. Additionally, the robustness of the reaction conditions means that batch-to-batch variability is minimized, providing supply chain heads with the predictability needed to plan long-term production schedules and meet tight delivery deadlines for global clients.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the substitution of hazardous gaseous reagents with safe solids and the use of earth-abundant palladium catalysts that operate at low loadings. By avoiding the need for specialized high-pressure autoclaves and the associated safety infrastructure, capital expenditure for new production lines is significantly lowered. The high reaction efficiency and yield reduce the amount of raw material required per kilogram of product, while the simplified workup procedure involving filtration and column chromatography minimizes labor and solvent costs. These factors combine to create a leaner manufacturing process that delivers substantial cost savings without compromising on the quality or purity of the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: The use of widely available commodity chemicals as starting materials mitigates the risk of supply disruptions that can occur with niche reagents. Sodium azide and various substituted hydrazones are produced by multiple global suppliers, ensuring competitive pricing and consistent availability even during market volatility. The flexibility of the substrate scope allows manufacturers to pivot quickly between different analogues based on customer demand without needing to retool or requalify entirely new synthetic routes. This agility is critical for reducing lead time for high-purity pharmaceutical intermediates, enabling partners to respond rapidly to changes in the drug development pipeline or market needs.
- Scalability and Environmental Compliance: Scaling this reaction from gram to tonnage levels is facilitated by the homogeneous nature of the catalytic system and the absence of exothermic hazards associated with gas handling. The process generates minimal hazardous waste compared to traditional methods that utilize stoichiometric amounts of toxic reagents or produce large volumes of saline wastewater. The ability to recycle the organic solvent and the potential for catalyst recovery further enhance the environmental profile of the manufacturing process. For companies committed to green chemistry principles, adopting this methodology supports sustainability goals while ensuring compliance with increasingly stringent environmental regulations regarding volatile organic compounds and heavy metal discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology in an industrial setting. These answers are derived directly from the experimental data and specifications outlined in the patent documentation to provide clarity on feasibility and performance. Understanding these details helps stakeholders make informed decisions about integrating this route into their existing portfolios.
Q: What are the advantages of using TFBen over gaseous CO in this synthesis?
A: TFBen acts as a solid carbon monoxide substitute, eliminating the need for high-pressure gas equipment and significantly enhancing operational safety and scalability in industrial settings.
Q: What is the substrate scope for R1 and R2 groups in this reaction?
A: The method demonstrates excellent compatibility with various groups including C1-C5 alkyls, substituted aryls (with halogens, alkoxy, or trifluoromethyl groups), benzyl, and naphthyl groups.
Q: How does this method compare to traditional cyclization routes?
A: Unlike traditional methods requiring harsh bases or high temperatures, this Pd-catalyzed route operates under milder conditions with higher yields and simpler post-processing steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,4-Triazole-3-one Supplier
As a leader in the fine chemical sector, NINGBO INNO PHARMCHEM possesses the technical expertise and infrastructure to translate this innovative patent into commercial reality for our global partners. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to manufacturing plant is seamless and efficient. We operate stringent purity specifications and utilize rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 1,2,4-triazole-3-one meets the highest industry standards. Our commitment to quality assurance means that clients can rely on us for consistent supply of critical intermediates needed for the development of next-generation therapeutics.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis method can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this palladium-catalyzed route for your specific target molecules. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that will demonstrate the viability of this technology for your supply chain.
