Advanced Palladium-Catalyzed Synthesis of 1,2,4-Triazole-3-one Compounds for Commercial Scale-up
Advanced Palladium-Catalyzed Synthesis of 1,2,4-Triazole-3-one Compounds for Commercial Scale-up
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for heterocyclic scaffolds that serve as critical building blocks for bioactive molecules. A significant breakthrough in this domain is detailed in patent CN112538054B, which discloses a highly efficient preparation method for 1,2,4-triazole-3-one compounds. These five-membered nitrogen-containing heterocycles are ubiquitous in medicinal chemistry, exhibiting a broad spectrum of biological activities including antifungal, anti-inflammatory, antitumor, antiviral, and anticonvulsant properties. As illustrated in the structural diversity of known bioactive agents, these cores are essential for developing next-generation therapeutics targeting enzymes like tyrosinase or receptors such as NK1 and AT1. The ability to access these structures reliably is paramount for any reliable pharmaceutical intermediate supplier aiming to support drug discovery pipelines.
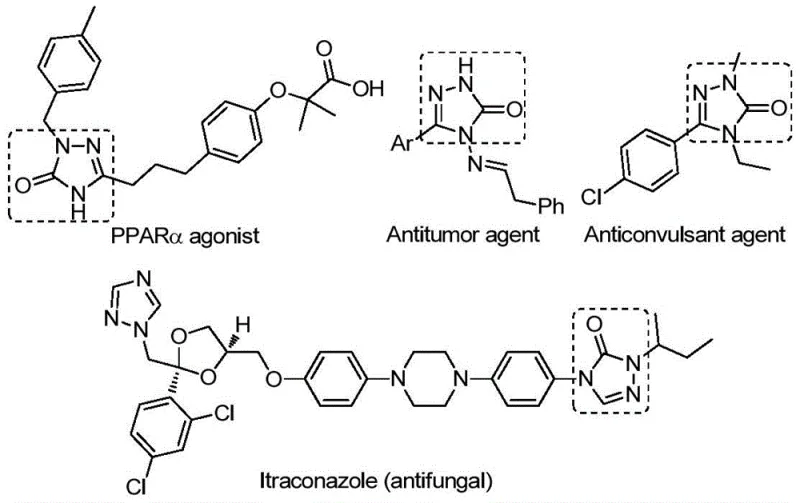
Traditionally, the synthesis of these valuable heterocycles has been fraught with challenges that hinder large-scale production. Conventional methodologies often rely on the cyclization of benzoyl hydrazides with urea under strong basic conditions, or the tandem reaction of hydrazides with isocyanates. These legacy processes frequently suffer from苛刻 reaction conditions, requiring extreme temperatures or pressures that pose safety risks in a manufacturing environment. Furthermore, many traditional routes necessitate the pre-activation of substrates, adding unnecessary steps that reduce overall atom economy and increase waste generation. The narrow substrate scope of these older methods also limits the ability of R&D teams to explore diverse chemical space, often resulting in low yields when attempting to introduce complex functional groups. For procurement managers, these inefficiencies translate directly into higher costs of goods sold (COGS) and extended lead times for critical high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The historical reliance on multi-step syntheses involving unstable intermediates like acyl isocyanates or thioamides creates significant bottlenecks in the supply chain. These reactions often require stringent exclusion of moisture and oxygen, demanding specialized equipment that increases capital expenditure. Additionally, the use of hazardous reagents such as gaseous carbon monoxide in carbonylation reactions presents severe safety liabilities, requiring extensive engineering controls to mitigate exposure risks. The purification of products from these messy reaction mixtures is often difficult, leading to lower isolated yields and higher levels of impurities that must be rigorously controlled to meet regulatory standards. Consequently, the commercial scale-up of complex pharmaceutical intermediates via these traditional pathways is often deemed economically unviable for all but the highest-value APIs.
The Novel Approach
In stark contrast, the methodology described in CN112538054B offers a streamlined, one-pot solution that addresses these痛点 directly. This novel approach utilizes a transition metal palladium-catalyzed carbonylation tandem cyclization reaction, starting from cheap and readily available chlorohydrazones and sodium azide. A key innovation is the use of TFBen (1,3,5-tricarboxylic acid phenol ester) as a solid carbon monoxide substitute, which safely releases CO in situ under heating conditions. This eliminates the need for handling toxic CO gas while maintaining high reaction efficiency. The process operates at a moderate temperature of 100°C in common organic solvents like 1,4-dioxane, making it highly amenable to standard reactor setups. This advancement represents a major step forward in cost reduction in pharmaceutical intermediate manufacturing, as it simplifies the operational workflow and reduces the burden on EHS (Environment, Health, and Safety) departments.

Mechanistic Insights into Pd-Catalyzed Carbonylation Tandem Cyclization
The success of this synthetic route lies in its elegant catalytic cycle, which orchestrates multiple bond-forming events in a single vessel. The reaction initiates with the oxidative addition of the palladium catalyst into the carbon-chlorine bond of the chlorohydrazone substrate, forming a reactive divalent palladium intermediate. Simultaneously, the thermal decomposition of TFBen generates carbon monoxide, which subsequently inserts into the carbon-palladium bond to create an acyl-palladium species. This acyl intermediate then reacts with sodium azide to generate an acyl azide compound in situ. The process continues with a Curtius rearrangement, transforming the acyl azide into a highly reactive isocyanate intermediate. Finally, an intramolecular nucleophilic addition occurs, closing the ring to yield the final 1,2,4-triazole-3-one scaffold. Understanding this mechanism is crucial for R&D directors, as it highlights the precise control over reaction kinetics required to minimize side products and maximize yield.
From an impurity control perspective, this mechanism offers distinct advantages over stepwise approaches. By keeping the reactive isocyanate intermediate within the coordination sphere of the catalyst or in close proximity for immediate cyclization, the potential for intermolecular side reactions, such as polymerization or hydrolysis, is significantly reduced. The use of specific ligands like Xantphos further stabilizes the palladium center, ensuring high turnover numbers and consistent performance across different substrate batches. This level of mechanistic precision allows for the synthesis of derivatives with varied substituents (R1 and R2 groups) without compromising purity. For quality assurance teams, this means a cleaner crude profile, which simplifies downstream purification and ensures that the final high-purity API intermediates meet stringent specifications with minimal effort.
How to Synthesize 1,2,4-Triazole-3-one Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and reaction parameters to achieve optimal results. The patent outlines a robust protocol where chlorohydrazone, sodium azide, the palladium catalyst, and the ligand are combined in an aprotic solvent. The choice of solvent is critical; while polar aprotic solvents like DMSO were tested, 1,4-dioxane was found to provide superior conversion rates, likely due to its ability to solubilize all components effectively while supporting the catalytic cycle. The reaction is typically run for 16 to 30 hours at 100°C, a timeframe that balances complete conversion with operational efficiency. Detailed standardized synthesis steps for this process are provided in the guide below.
- Combine palladium catalyst (Pd2(dba)3), ligand (Xantphos), CO substitute (TFBen), chlorohydrazone, and sodium azide in an organic solvent like 1,4-dioxane.
- Heat the reaction mixture to 100°C and maintain stirring for 16 to 30 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target 1,2,4-triazole-3-one compound.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply continuity, this patented method offers compelling economic and logistical benefits. The shift from hazardous gases to solid reagents and the reduction in synthetic steps directly correlate to lower operational expenditures. By adopting this technology, manufacturers can mitigate risks associated with volatile raw material markets and complex logistics, ensuring a more stable supply of critical heterocyclic building blocks.
- Cost Reduction in Manufacturing: The elimination of high-pressure carbon monoxide gas removes the need for specialized autoclaves and extensive safety monitoring systems, leading to substantial capital and operational cost savings. Furthermore, the use of inexpensive starting materials like sodium azide and chlorohydrazones, which are commodity chemicals, drastically lowers the raw material cost compared to pre-activated substrates used in traditional methods. The high yields reported, reaching up to 96% for certain substrates, mean less waste and higher throughput per batch, effectively driving down the cost per kilogram of the final product.
- Enhanced Supply Chain Reliability: Sourcing solid CO surrogates like TFBen and standard palladium catalysts is far more reliable than managing the supply chain for toxic gases, which often face regulatory shipping restrictions. The robustness of the reaction conditions (100°C, atmospheric pressure equivalent) allows for production in a wider range of facilities, reducing dependency on specialized contract manufacturing organizations. This flexibility ensures reducing lead time for high-purity pharmaceutical intermediates, allowing drug developers to accelerate their timelines from preclinical to clinical stages without supply bottlenecks.
- Scalability and Environmental Compliance: The simplified workup procedure, involving filtration and standard column chromatography, generates less hazardous waste compared to the acidic or basic quenching required in older methods. The high atom economy of the tandem reaction minimizes the formation of byproducts, aligning with green chemistry principles and reducing the environmental footprint of the manufacturing process. This makes the technology highly scalable, capable of transitioning from gram-scale R&D to multi-ton commercial production with minimal process re-engineering, ensuring long-term supply continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity for technical teams evaluating this route for adoption.
Q: What are the safety advantages of using TFBen over carbon monoxide gas?
A: Using TFBen (1,3,5-tricarboxylic acid phenol ester) as a solid carbon monoxide substitute eliminates the need for handling hazardous high-pressure CO gas cylinders, significantly improving operational safety and simplifying reactor requirements for industrial scale-up.
Q: What is the substrate scope for this synthesis method?
A: The method demonstrates excellent compatibility with various functional groups, successfully synthesizing derivatives with aryl, alkyl, naphthyl, and furyl substituents, achieving yields up to 96% for optimized substrates.
Q: How does this method compare to traditional cyclization routes?
A: Unlike traditional methods requiring harsh conditions or pre-activated substrates, this palladium-catalyzed approach operates under milder conditions (100°C) with readily available starting materials like chlorohydrazones and sodium azide, offering higher efficiency and simpler post-processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,4-Triazole-3-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of efficient synthetic methodologies in accelerating drug development. Our team of expert chemists has extensively evaluated the Pd-catalyzed carbonylation route described in CN112538054B and confirmed its potential for robust manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from benchtop to market. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 1,2,4-triazole-3-one intermediate delivered meets the highest international standards.
We invite you to collaborate with us to leverage this advanced technology for your specific API projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of innovation, reliability, and cost-efficiency.
