Scalable Palladium-Catalyzed Synthesis of 1,2,4-Triazole-3-one Compounds for Advanced Drug Discovery
Scalable Palladium-Catalyzed Synthesis of 1,2,4-Triazole-3-one Compounds for Advanced Drug Discovery
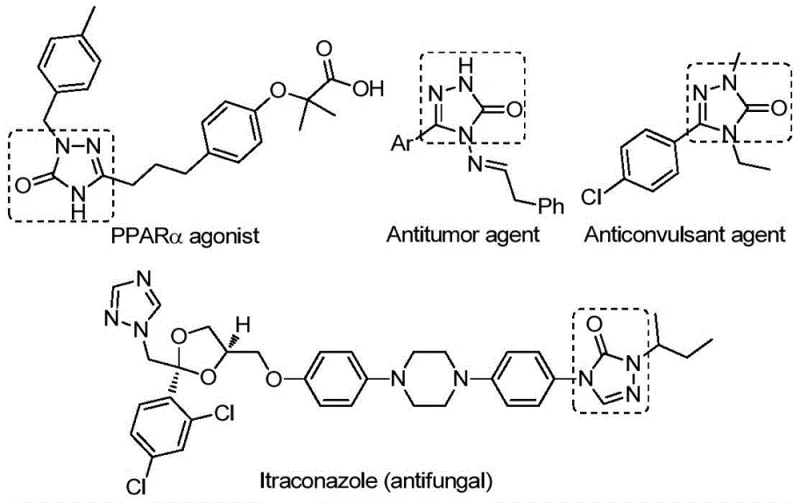
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for nitrogen-containing heterocycles due to their prevalence in bioactive small molecules. A significant breakthrough in this domain is detailed in patent CN112538054B, which discloses a highly efficient preparation method for 1,2,4-triazole-3-one compounds. These scaffolds are critical structural motifs found in a wide array of therapeutic agents, exhibiting potent antifungal, anti-inflammatory, antitumor, antiviral, and anticonvulsant activities. As illustrated in the biological context below, these cores serve as key pharmacophores in tyrosinase inhibitors, CB receptor modulators, and angiotensin AT1 receptor antagonists. For R&D directors and procurement specialists, accessing a reliable pharmaceutical intermediate supplier capable of delivering these complex structures with high purity is essential for accelerating drug discovery pipelines.
The strategic value of this patent lies not only in the biological potential of the products but also in the operational simplicity of the synthesis. Traditional methods for constructing the 1,2,4-triazole-3-one ring often suffer from significant limitations, including the requirement for harsh reaction conditions, multi-step sequences, and the need for pre-activated substrates that drive up costs and waste. In contrast, the novel approach described in CN112538054B utilizes a transition metal palladium-catalyzed carbonylation tandem cyclization reaction. This method employs readily available chlorohydrazones and sodium azide as starting materials, bypassing the need for dangerous gaseous carbon monoxide by using a solid CO surrogate. This shift represents a paradigm change in cost reduction in API manufacturing, allowing for safer and more economically viable production scales.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,2,4-triazol-3-one compounds has been constrained by inefficient methodologies that struggle to meet modern green chemistry standards. Common legacy routes include the cyclization of benzoyl hydrazide with urea under strong basic conditions, or the tandem cyclization of hydrazides with isocyanates. These processes frequently demand extreme temperatures, utilize corrosive reagents, and exhibit poor atom economy. Furthermore, methods involving thioamides or acyl isocyanates often require tedious pre-activation steps and result in low yields with narrow substrate scopes. For supply chain heads, these inefficiencies translate into unpredictable lead times and higher raw material costs, creating bottlenecks in the production of high-purity pharmaceutical intermediates. The inability to tolerate diverse functional groups further limits the chemical space accessible for medicinal chemistry optimization.
The Novel Approach
The innovative methodology presented in the patent overcomes these historical barriers through a streamlined palladium-catalyzed carbonylation strategy. By reacting chlorohydrazones with sodium azide in the presence of a palladium catalyst and a ligand system, the process achieves a tandem cyclization that constructs the triazole ring and installs the ketone functionality in a single operational step. The use of TFBen as a carbon monoxide substitute is particularly advantageous, as it eliminates the safety hazards associated with handling high-pressure CO gas. This approach demonstrates exceptional substrate compatibility, successfully synthesizing derivatives with various alkyl, aryl, and heteroaryl substituents. The reaction efficiency is markedly improved, with yields reaching as high as 96% for certain substrates, thereby providing a reliable agrochemical intermediate supplier or pharma partner with a distinct competitive advantage in terms of output and purity.
Mechanistic Insights into Pd-Catalyzed Carbonylation Tandem Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to optimize the process for specific analogs. The reaction is believed to initiate with the oxidative addition of the palladium(0) catalyst into the carbon-chlorine bond of the chlorohydrazone substrate, generating a reactive divalent palladium intermediate. Simultaneously, the CO surrogate, TFBen, undergoes thermal decomposition to release carbon monoxide in situ. This generated CO then inserts into the carbon-palladium bond, forming a key acyl-palladium species. Subsequent interaction with sodium azide leads to the formation of an acyl azide compound, which spontaneously undergoes a Curtius rearrangement to generate an isocyanate intermediate. Finally, an intramolecular nucleophilic addition occurs, closing the ring to afford the final 1,2,4-triazole-3-one product. This elegant cascade minimizes side reactions and maximizes atom utilization.

From an impurity control perspective, this mechanism offers significant benefits. The use of a well-defined catalytic cycle involving Pd2(dba)3 and Xantphos ensures high selectivity, reducing the formation of by-products common in non-catalytic thermal cyclizations. The mild reaction temperature of 100°C prevents the degradation of sensitive functional groups that might occur under more vigorous conditions. Moreover, the stoichiometric use of sodium azide acts as both a reactant and a driving force for the rearrangement, ensuring complete consumption of the intermediate species. For quality control laboratories, this translates to a cleaner crude reaction profile, simplifying downstream purification and ensuring that stringent purity specifications are met with less effort. The ability to tune the R1 and R2 groups allows for precise modulation of the electronic and steric properties of the final molecule without compromising the integrity of the core scaffold.
How to Synthesize 1,2,4-Triazole-3-one Efficiently
The practical execution of this synthesis is designed to be straightforward, facilitating easy technology transfer from the laboratory to pilot and commercial plants. The protocol involves charging a reactor with the palladium catalyst, ligand, CO surrogate, chlorohydrazone, and sodium azide in a suitable aprotic solvent such as 1,4-dioxane. The mixture is heated to 100°C for a duration of 16 to 30 hours, depending on the specific substrate reactivity. Post-reaction workup is minimal, typically involving filtration to remove inorganic salts followed by standard silica gel chromatography. This operational simplicity is a key factor in reducing lead time for high-purity pharmaceutical intermediates, as it requires less specialized equipment and operator training compared to high-pressure carbonylation techniques. The detailed standardized synthesis steps are outlined in the guide below.
- Combine Pd2(dba)3, Xantphos ligand, TFBen, chlorohydrazone substrate, and sodium azide in an organic solvent such as 1,4-dioxane.
- Heat the reaction mixture to 100°C and maintain stirring for 16 to 30 hours to ensure complete conversion.
- Upon completion, filter the mixture, adsorb onto silica gel, and purify via column chromatography to isolate the target 1,2,4-triazole-3-one.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this patented method offers tangible economic and logistical benefits that extend beyond simple yield improvements. The shift towards this catalytic protocol addresses several critical pain points in the sourcing of complex heterocyclic building blocks. By leveraging cheap and commercially available starting materials like chlorohydrazones and sodium azide, manufacturers can significantly decouple production costs from volatile specialty reagent markets. The elimination of hazardous gas handling infrastructure further reduces capital expenditure requirements for production facilities. Additionally, the robustness of the reaction conditions ensures consistent batch-to-batch quality, which is vital for maintaining supply continuity in regulated pharmaceutical markets.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the replacement of expensive or hazardous reagents with commodity chemicals. The use of TFBen as a solid CO source eliminates the need for specialized high-pressure reactors and safety systems required for gaseous carbon monoxide, leading to substantial cost savings in facility operations and maintenance. Furthermore, the high catalytic efficiency means that lower loadings of precious metal catalysts can be utilized while maintaining high turnover, directly reducing the raw material cost per kilogram of the active pharmaceutical ingredient. The simplified workup procedure also lowers labor and solvent consumption costs associated with extensive purification steps.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available raw materials that are not subject to the same geopolitical or logistical constraints as exotic reagents. Chlorohydrazones and sodium azide are produced at scale globally, ensuring a stable supply base. The mild reaction conditions reduce the risk of batch failures due to thermal runaway or equipment malfunction, thereby improving on-time delivery performance. This reliability allows procurement teams to negotiate better terms and maintain leaner inventory levels, knowing that the production process is robust and predictable. The method's scalability ensures that supply can be rapidly ramped up to meet sudden increases in demand without compromising quality.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this method aligns well with modern green chemistry principles. The avoidance of toxic gases and the use of recyclable solvents like dioxane minimize the environmental footprint of the manufacturing process. The high atom economy of the tandem cyclization reduces the volume of chemical waste generated, lowering disposal costs and simplifying compliance with increasingly strict environmental regulations. The process is inherently scalable, having been demonstrated to work efficiently from milligram to gram scales, which suggests a smooth path to ton-scale commercial production. This scalability ensures that the technology can support the entire lifecycle of a drug candidate, from early discovery to late-stage clinical trials and commercial launch.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology for their specific applications, we have compiled answers to common inquiries regarding the process parameters and scope. These insights are derived directly from the experimental data and technical disclosures within the patent documentation. Understanding these nuances is critical for assessing the feasibility of integrating this synthetic route into existing manufacturing workflows. The following questions address substrate scope, safety considerations, and purification strategies.
Q: What are the key advantages of this Pd-catalyzed method over traditional synthesis?
A: Unlike traditional methods requiring harsh conditions or pre-activated substrates, this protocol utilizes cheap chlorohydrazones and a solid CO surrogate (TFBen), offering milder conditions (100°C), broader substrate tolerance, and significantly higher yields up to 96%.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method is designed for scalability. It uses commercially available reagents, avoids hazardous gaseous carbon monoxide by using TFBen, and demonstrates excellent functional group compatibility, making it ideal for commercial scale-up of complex pharmaceutical intermediates.
Q: What is the role of TFBen in this reaction mechanism?
A: TFBen (1,3,5-tricarboxylic acid phenol ester) acts as a safe and efficient carbon monoxide surrogate. Under heating conditions, it releases CO in situ, which inserts into the palladium-carbon bond to form the necessary acyl-palladium intermediate for cyclization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,4-Triazole-3-one Supplier
As the global demand for advanced heterocyclic intermediates continues to grow, partnering with an experienced CDMO is essential for navigating the complexities of process development and scale-up. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the bench to the plant. Our team is equipped with rigorous QC labs and state-of-the-art analytical instrumentation to guarantee stringent purity specifications for every batch of 1,2,4-triazole-3-one derivatives we produce. We understand the critical nature of timeline and quality in the pharmaceutical sector and are committed to delivering excellence.
We invite you to collaborate with us to leverage this cutting-edge synthetic technology for your next drug discovery program. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. Please contact us today to request specific COA data and route feasibility assessments for your target molecules. Let us help you optimize your supply chain and accelerate your path to market with our superior manufacturing capabilities.
