Advanced Palladium-Catalyzed Synthesis of 1,2,4-Triazole-3-one Compounds for Commercial Scale-up
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes for heterocyclic scaffolds that possess significant biological activity. Patent CN112538054B discloses a groundbreaking preparation method for 1,2,4-triazole-3-one compounds, a privileged structural motif found in numerous bioactive molecules ranging from antifungal agents to antitumor drugs. As illustrated in the structural diversity of known bioactive molecules, these heterocycles serve as critical cores for PPARα agonists, anticonvulsants, and potent antifungal agents like Itraconazole. The innovation presented in this patent addresses the long-standing challenges in synthesizing these valuable intermediates by introducing a transition metal palladium-catalyzed carbonylation tandem cyclization reaction. This method not only streamlines the synthetic pathway but also utilizes inexpensive and commercially available starting materials, marking a significant step forward for reliable pharmaceutical intermediate supplier networks aiming to optimize their production pipelines.
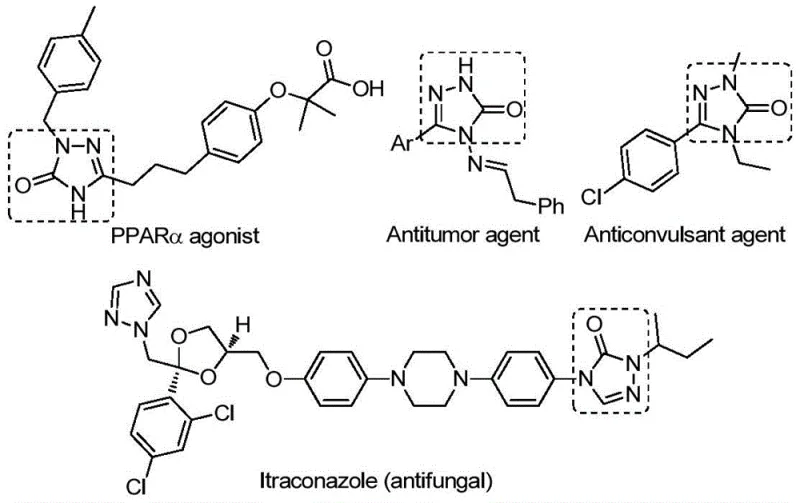
The limitations of conventional methods for constructing the 1,2,4-triazole-3-one ring system have historically hindered efficient large-scale production. Traditional synthetic strategies often rely on the cyclization of benzoyl hydrazide with urea under strong basic conditions, or the tandem cyclization of hydrazides with isocyanates. These legacy processes are frequently plagued by harsh reaction conditions, such as extreme temperatures or highly corrosive reagents, which pose significant safety risks and equipment corrosion issues in a plant setting. Furthermore, many existing routes require the pre-activation of substrates, adding unnecessary synthetic steps that reduce overall atom economy and increase waste generation. The narrow substrate scope of these older methods often fails to accommodate sensitive functional groups, limiting the chemical space available for medicinal chemists during lead optimization phases. Consequently, the industry has faced persistent bottlenecks in cost reduction in pharmaceutical intermediates manufacturing due to low yields and complex purification requirements associated with these outdated techniques.
In stark contrast, the novel approach detailed in the patent utilizes a sophisticated palladium-catalyzed carbonylation strategy that elegantly bypasses these historical hurdles. By employing chlorohydrazones as the primary building blocks and sodium azide as the nitrogen source, the reaction achieves high efficiency through a tandem cyclization mechanism. A key feature of this methodology is the use of TFBen (1,3,5-tricarboxylic acid phenol ester) as a solid carbon monoxide surrogate, which eliminates the need for handling dangerous CO gas cylinders, thereby drastically improving operational safety. The reaction proceeds smoothly in aprotic solvents like 1,4-dioxane at moderate temperatures of 100-120 °C, demonstrating excellent compatibility with a wide array of functional groups including halogens, alkoxys, and alkyl chains. This versatility allows for the rapid generation of diverse libraries of 1,2,4-triazole-3-one derivatives, facilitating the commercial scale-up of complex pharmaceutical intermediates with significantly reduced lead times.
Mechanistic Insights into Pd-Catalyzed Carbonylation Tandem Cyclization
The mechanistic pathway of this transformation offers profound insights into its high efficiency and selectivity, which are critical for R&D directors evaluating process feasibility. The reaction is initiated by the oxidative addition of the palladium catalyst into the carbon-chlorine bond of the chlorohydrazone substrate, forming a reactive divalent palladium intermediate. Simultaneously, the solid CO surrogate, TFBen, undergoes thermal decomposition to release carbon monoxide in situ. This generated CO then inserts into the carbon-palladium bond to create an acyl palladium species, a pivotal step that constructs the carbonyl functionality of the final triazole ring. Subsequently, the acyl palladium intermediate reacts with sodium azide to generate an acyl azide compound, which spontaneously undergoes a Curtius rearrangement to form an isocyanate intermediate. The cycle concludes with an intramolecular nucleophilic addition where the hydrazine nitrogen attacks the isocyanate carbon, closing the ring to yield the stable 1,2,4-triazole-3-one product. This elegant cascade ensures that the reaction proceeds with high atom economy and minimal byproduct formation.

Understanding the impurity profile is essential for maintaining stringent purity specifications in API manufacturing. The mechanism suggests that potential impurities could arise from incomplete oxidative addition or side reactions of the acyl azide intermediate; however, the use of the specific ligand Xantphos stabilizes the palladium center and promotes the desired reductive elimination pathway. The choice of solvent plays a crucial role in suppressing side reactions; aprotic solvents like 1,4-dioxane are preferred over polar aprotic solvents like DMF or DMSO, which were found to inhibit the reaction progress. The protocol specifies a molar ratio of chlorohydrazone to sodium azide of approximately 1:2.5, ensuring that the azide is in excess to drive the formation of the acyl azide intermediate to completion, thus minimizing the accumulation of unreacted starting materials. This precise control over reaction parameters ensures that the final product meets the rigorous quality standards required for downstream pharmaceutical applications, reducing the burden on purification teams.
How to Synthesize 1,2,4-Triazole-3-one Efficiently
The practical implementation of this synthesis route is designed for ease of operation, making it highly attractive for process chemistry teams looking to transfer technology from the lab to the pilot plant. The procedure involves charging a reactor with the palladium catalyst, ligand, CO surrogate, chlorohydrazone, and sodium azide in the chosen organic solvent. The mixture is then heated to the specified temperature range and stirred for a defined period to ensure full conversion. Following the reaction, a straightforward workup involving filtration and silica gel treatment allows for the isolation of the crude product, which can be further purified via standard column chromatography. This simplicity in operation reduces the technical barrier for adoption and minimizes the risk of operator error during scale-up. For detailed procedural specifics regarding reagent quantities and exact timing, please refer to the standardized guide below.
- Combine palladium catalyst (Pd2(dba)3), ligand (Xantphos), CO surrogate (TFBen), chlorohydrazone substrate, and sodium azide in an aprotic organic solvent such as 1,4-dioxane.
- Heat the reaction mixture to a temperature range of 100-120 °C and maintain stirring for a duration of 16 to 30 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify the crude product via column chromatography to isolate the target 1,2,4-triazole-3-one compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this novel synthetic route offers transformative advantages that directly impact the bottom line and operational resilience. The shift towards using chlorohydrazones and solid CO surrogates represents a strategic move away from volatile and hazardous reagents, simplifying logistics and storage requirements. By eliminating the need for high-pressure gas infrastructure and specialized handling equipment for carbon monoxide, facilities can achieve substantial cost savings in capital expenditure and safety compliance. Furthermore, the high reaction efficiency and broad substrate tolerance mean that fewer batches are rejected due to quality issues, leading to a more predictable and reliable supply of critical intermediates. This stability is paramount for maintaining continuous production schedules in the fast-paced pharmaceutical market.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the use of cheap and readily available starting materials. Chlorohydrazones can be rapidly synthesized from common acid chlorides and hydrazines, while sodium azide is a commodity chemical with a stable global supply chain. The replacement of gaseous CO with the solid surrogate TFBen removes the costs associated with gas cylinder rentals, transportation hazards, and specialized scrubbing systems. Additionally, the high yields reported in the patent examples, reaching up to 96% for certain substrates, imply a significant reduction in raw material waste and solvent usage per kilogram of product. This efficiency translates directly into a lower cost of goods sold (COGS), providing a competitive edge in pricing negotiations with downstream API manufacturers.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on niche or hazardous reagents that may face regulatory shipping restrictions. This method mitigates such risks by utilizing reagents that are generally commercially available and easier to transport. The robustness of the reaction conditions, which tolerate a wide range of functional groups, means that the supply of diverse intermediates is less likely to be disrupted by the need for custom-synthesized, exotic starting materials. The ability to source key components from multiple vendors enhances bargaining power and reduces the risk of single-source dependency. Moreover, the simplified post-treatment process reduces the turnaround time for batch release, allowing for faster response to market demand fluctuations and tighter inventory management.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new environmental challenges, but this methodology is inherently greener due to its high atom economy and reduced hazard profile. The use of a solid CO surrogate minimizes the release of toxic gases into the environment, aligning with increasingly stringent global environmental regulations. The reaction operates in standard organic solvents that can be recovered and recycled, further reducing the environmental footprint. The mild thermal conditions (100-120 °C) are energy-efficient compared to high-temperature pyrolysis methods often used in heterocycle synthesis. These factors combined make the process highly scalable from gram to multi-ton quantities without requiring massive re-engineering of waste treatment facilities, ensuring long-term sustainability and regulatory compliance for manufacturing partners.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology, we have compiled answers to common questions based on the detailed experimental data provided in the patent documentation. These insights address concerns regarding safety, scalability, and chemical scope, ensuring that all stakeholders have a clear understanding of the process capabilities. The following responses are derived directly from the validated results of Examples 1 through 15, reflecting the real-world performance of the synthesis method under optimized conditions.
Q: What are the primary advantages of this Pd-catalyzed method over traditional synthesis routes?
A: Unlike conventional methods that often require harsh conditions, pre-activated substrates, or toxic gases, this novel approach utilizes cheap and readily available chlorohydrazones and a solid CO surrogate (TFBen). It operates under relatively mild thermal conditions (100-120 °C) and demonstrates exceptional substrate compatibility, yielding products with high efficiency and purity suitable for pharmaceutical applications.
Q: Is the carbon monoxide source used in this reaction safe for industrial scale-up?
A: Yes, the process utilizes TFBen (1,3,5-tricarboxylic acid phenol ester) as a solid carbon monoxide surrogate rather than hazardous CO gas cylinders. This solid reagent releases CO in situ under heating conditions, significantly enhancing operational safety and simplifying handling protocols for large-scale manufacturing environments.
Q: What is the substrate scope for R1 and R2 groups in this synthesis?
A: The method exhibits broad functional group tolerance. R1 can be varied across C1-C5 alkyls, substituted or unsubstituted aryls (including naphthyl and furyl groups), while R2 accommodates C1-C5 alkyls, benzyl, and various aryl groups. Substituents such as halogens, alkoxys, and trifluoromethyl groups are well-tolerated, allowing for the design of diverse bioactive analogues.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,4-Triazole-3-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and safe synthetic routes in the development of next-generation therapeutics. Our team of expert chemists has thoroughly analyzed the potential of this palladium-catalyzed carbonylation method and is fully prepared to implement it for your specific project needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 1,2,4-triazole-3-one intermediate we deliver meets the highest industry standards for quality and consistency.
We invite you to collaborate with us to leverage this advanced technology for your drug development pipeline. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and target timelines. We encourage you to reach out today to discuss your project specifics,索取 specific COA data for our reference standards, and obtain comprehensive route feasibility assessments. Let us help you accelerate your time-to-market with a supply chain partner dedicated to innovation, quality, and reliability.
